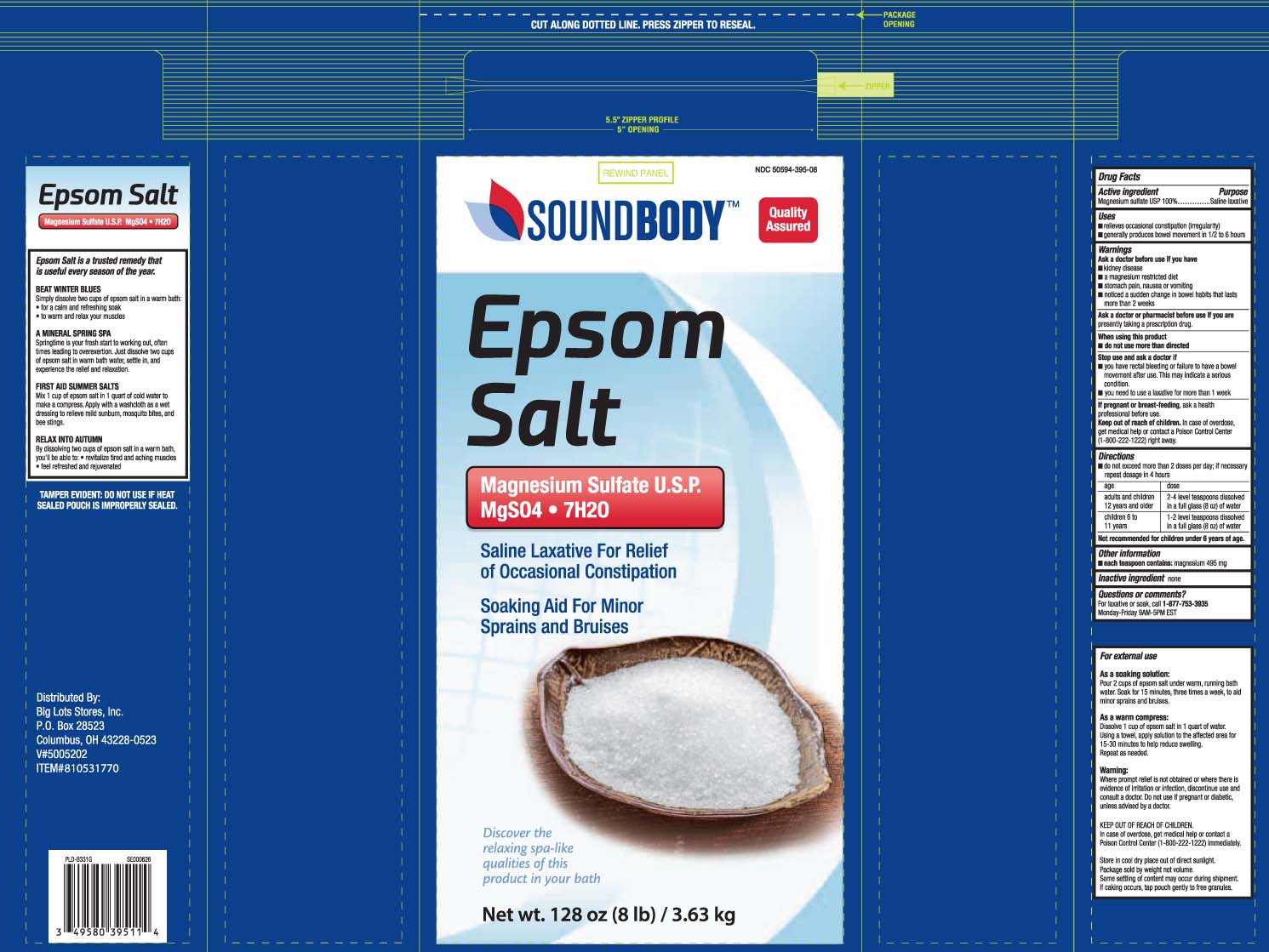
Label: EPSOM SALT granule
- NDC Code(s): 50594-395-08
- Packager: Big Lots
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- kidney disease
- a magnesium restricted diet
- stomach pain, nausea or vomiting
- noticed a sudden change in bowel habits that lasts more than 2 weeks
-
Directions
- do not exceed more than 2 doses per day; if necessary repeat dosage in 4 hours
age dose adults and children 12 years and older 2-4 level teaspoons dissolved in a full glass (8oz) of water children 6 to 11 years 1-2 level teaspoons dissolved in a full glass (8oz) of water
Not recommended for children under 6 years of age. - Other information
- Inactive ingredient
- Questions or comments?
- Principal Display Panel
- Package Label
-
INGREDIENTS AND APPEARANCE
EPSOM SALT
epsom salt granuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50594-395 Route of Administration ORAL, TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM SULFATE HEPTAHYDRATE (UNII: SK47B8698T) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM SULFATE HEPTAHYDRATE 1 g in 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50594-395-08 3629 g in 1 POUCH; Type 0: Not a Combination Product 04/30/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 04/30/2021 Labeler - Big Lots (017885351)