Label: ANTIFLU DES COUGH AND FLU- acetaminophen phenylephrine hcl chlorpheniramine maleate capsule
- NDC Code(s): 82688-001-12
- Packager: Productos Farmaceuticos, S.A. de C.V.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 18, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
-
Warnings
Liver warning This product contains acetaminophen. Severe liver damage may occur if you take ▪More than 4,000 mg in 24 hours, which is the maximum daily amount ▪ With other drugs containing acetaminophen ▪ 3 or more alcoholic drinks every day while using this product.
Allergy alert: Acetaminophen may cause severe skin or severe allergic reactions. Symptoms can include▪ skin reddening ▪ blisters ▪ rash ▪ hives ▪ facial swelling ▪ asthma (wheezing) ▪ shock. If a skin or any allergic reaction occurs, stop use and seek medical attention right away.
Sore throat warning: Severe or persistent sore throat lasting more than 2 days or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious. Consult a Doctor promptly.
-
Do not use
▪ With any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains Acetaminophen, ask a Doctor. If you are now taking a prescription Monoamine Oxidase Inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or if you have taken these products 2 weeks previously. If you do not know if your prescription drug contains an MAOI, ask a Doctor before taking this product
- Ask a Doctor before use if you have
- Ask a Doctor before use if you are
-
When using this product
▪ Do not exceed recommended dosage. If nervousness, dizziness, or sleeplessness occur, discontinue use and consult a Doctor. ▪ Do not mix with alcohol, sedatives and tranquilizers as they may increase drowsiness ▪ Driving a motor vehicle or operating machinery could be affected ▪ Excitability may occur especially in children(12-18 y) ▪ if symptoms do not improve within 7 days or are accompanied by fever, consult a Doctor
- Stop use and ask a Doctor if
-
Keep out of reach of children
If pregnant or breast-feeding, ask a health professional before use .
As with all medical products keep out of reach of children.
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away. Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
- Directions
- Other information
- Inactive Ingredients
- Questions and Comments?
- Package Label
-
INGREDIENTS AND APPEARANCE
ANTIFLU DES COUGH AND FLU
acetaminophen phenylephrine hcl chlorpheniramine maleate capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82688-001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 4 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MAGNESIUM STEARATE (UNII: 70097M6I30) GELATIN (UNII: 2G86QN327L) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 28 (UNII: 767IP0Y5NH) D&C RED NO. 33 (UNII: 9DBA0SBB0L) POVIDONE (UNII: FZ989GH94E) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SHELLAC (UNII: 46N107B71O) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color gray (gray and orange) Score no score Shape CAPSULE (immediate release hard gelatin) Size 24mm Flavor Imprint Code ANTIDESFLU Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82688-001-12 1 in 1 CARTON 04/15/2022 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 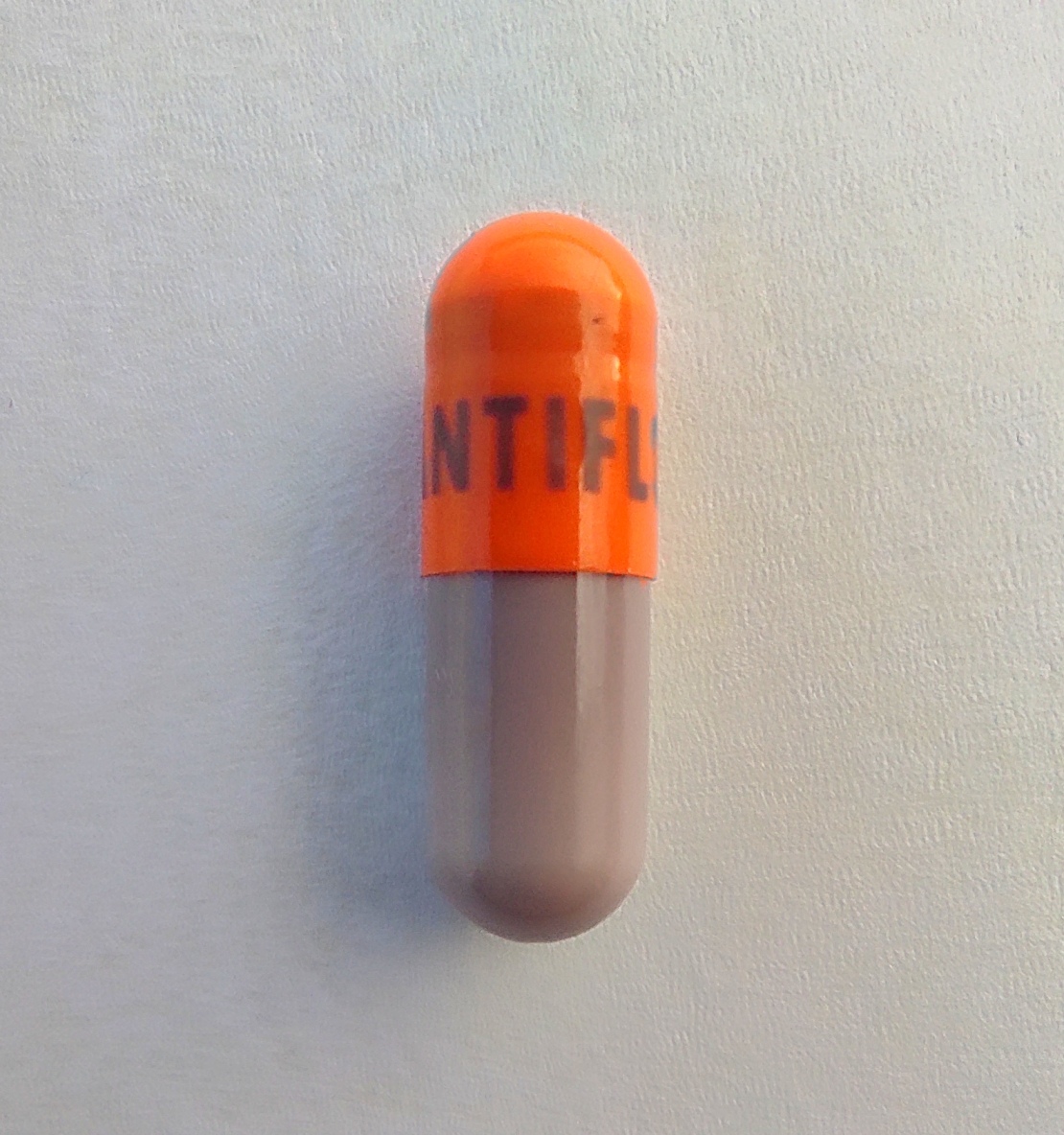
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 04/15/2022 Labeler - Productos Farmaceuticos, S.A. de C.V. (815664461) Establishment Name Address ID/FEI Business Operations Productos Farmaceuticos, S.A. de C.V. 815664461 manufacture(82688-001)


