Label: ACNE FREE SEVERE ACNE 24 HR ACNE CLEARING SYSTEM- benzoyl peroxide kit
- NDC Code(s): 80861-004-01, 80861-010-01, 80861-011-01
- Packager: AcneFree, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
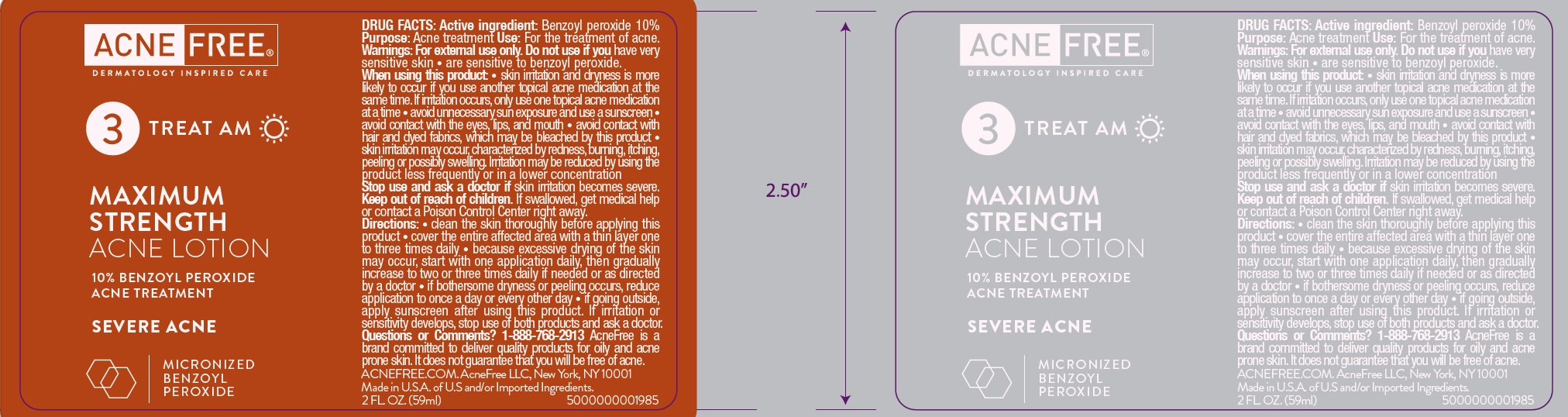
- Active ingredient
- Use
-
Warnings
For external use only
When using this product
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with the eyes, lips, and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
- skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
-
Directions
- use every morning and evening
- apply a dime-size amount to damp skin and gently massage, avoiding the eye area
- rinse well
- use wash on entire affected area one to two times daily
- because excessive drying of the skin may occur, start with washing once daily, then gradually increase to two times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce cleansing to once a day or every other day
- if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor.
- may be used on chest or back
-
Questions or comments?
water, glycerin, propylene glycol, cocamidopropyl hydroxysultaine, sodium C14-16 olefin sulfonate, xanthan gum, citric acid, potassium hydroxide, methyl methacrylate/glycol dimethacrylate crosspolymer, sodium lauroyl lactylate, glycolic acid, ceramide NP, ceramide AP, phytosphingosine, cholesterol, carbomer, ceramide EOP
- Questions or comments?
- Drug Facts
- Active ingredient
- Use
-
Warnings
For external use only
When using this product
• skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
• avoid unnecessary sun exposure and use a sunscreen
• avoid contact with the eyes, lips, and mouth
• avoid contact with hair and dyed fabrics, which may be bleached by this product
• skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration. -
Directions
• clean the skin thoroughly before applying this product
• cover the entire affected area with a thin layer one to three times daily
• because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
• if bothersome dryness or peeling occurs, reduce application to once a day or every other day
• if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor. -
Inactive ingredients
water, butylene glycol, C12-15 alkyl benzoate, glycerin, glyceryl stearate, PEG-100 stearate, cetearyl alcohol, phenoxyethanol, hydroxypropyl starch phosphate, polysorbate 60, dimethicone, xanthan gum, cetyl alcohol, bisabolol, tetrasodium EDTA, ethylhexylglycerin, methylisothiazolinone, benzalkonium chloride, sea whip extract, zingiber officinale (ginger) root extract, chamomilla recutita (matricaria) flower extract, alcohol, leuconostoc/radish root ferment filtrate, methylparaben, disodium EDTA, ethylparaben, methyl methacrylate/glycol dimethacrylate crosspolymer
- Questions or comments?
- KIT 80861-010-01
- 80861-004-01
- 80861-011-01
-
INGREDIENTS AND APPEARANCE
ACNE FREE SEVERE ACNE 24 HR ACNE CLEARING SYSTEM
benzoyl peroxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80861-010 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80861-010-01 1 in 1 KIT 12/01/2017 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 1 BOTTLE 59 mL Part 1 of 2 OIL FREE ACNE CLEANSER
benzoyl peroxide gelProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) COCAMIDOPROPYL HYDROXYSULTAINE (UNII: 62V75NI93W) SODIUM C14-16 OLEFIN SULFONATE (UNII: O9W3D3YF5U) XANTHAN GUM (UNII: TTV12P4NEE) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) METHYL METHACRYLATE (UNII: 196OC77688) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) GLYCOLIC ACID (UNII: 0WT12SX38S) CERAMIDE NP (UNII: 4370DF050B) CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) CHOLESTEROL (UNII: 97C5T2UQ7J) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) CERAMIDE 1 (UNII: 5THT33P7X7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80861-004-01 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 12/01/2017 Part 2 of 2 MAXIMUM STRENGTH ACNE
benzoyl peroxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLYSORBATE 60 (UNII: CAL22UVI4M) DIMETHICONE (UNII: 92RU3N3Y1O) XANTHAN GUM (UNII: TTV12P4NEE) CETYL ALCOHOL (UNII: 936JST6JCN) LEVOMENOL (UNII: 24WE03BX2T) EDETATE SODIUM (UNII: MP1J8420LU) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) PSEUDOPTEROGORGIA ELISABETHAE (UNII: UDY3H1OUX5) GINGER (UNII: C5529G5JPQ) CHAMOMILE (UNII: FGL3685T2X) ALCOHOL (UNII: 3K9958V90M) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) METHYLPARABEN (UNII: A2I8C7HI9T) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) ETHYLPARABEN (UNII: 14255EXE39) METHYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: EG97988M5Q) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80861-011-01 59 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 12/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 12/01/2017 Labeler - AcneFree, LLC (122237140)