Label: ADVANTUS- imidacloprid tablet, chewable
-
NDC Code(s):
58198-0017-1,
58198-0017-2,
58198-0017-3,
58198-0018-1, view more58198-0018-2, 58198-0018-3
- Packager: Elanco US Inc.
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated April 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients:
- Use:
- Warnings:
- Side Effects:
-
Directions:
You should weigh your dog before giving this medication to make sure you are using the right size for your dog. Do not give to puppies younger than 10 weeks of age or to dogs weighing less than 4 pounds. Do not give more than one tablet a day.
Use the table to find the right dose for your dog:
*If your dog becomes re-infested, you can safely repeat treatment as often as once a day. Dosing Table
Body
WeightDose*
Soft Chew Strength
4-22 lbs
One Soft Chew
7.5 mg imidacloprid
23-110
lbsOne Soft Chew
37.5 mg imidacloprid
To give advantus, place the soft chew directly in your dog's mouth or allow your dog to take the soft chew from your hand. Watch closely to ensure your dog swallows the soft chew.
A single dose kills adult fleas on your dog. Because advantus does not have an effect on fleas in the environment, your dog may become re-infested with fleas. If your dog becomes re-infested, you can safely repeat treatment as often as once a day. Contact your veterinarian if you have any questions about flea infestations on your dog.
Treat all pets in your household with a flea protection product. advantus should only be given to dogs.
-
Questions/Comments?
For product questions or to report side effects, call Elanco at 888-545-5973. For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae
-
Other Information:
- •
- Contact your veterinarian if your dog ingests more than the recommended dose of advantus.
- •
- advantus has not been tested in pregnant or nursing dogs.
- •
- advantus starts to kill fleas within 1 hour. A study demonstrated that advantus achieves greater than 96% effectiveness against adult fleas within 4 hours.
- •
- advantus may be used together with other products, including heartworm preventatives, corticosteroids, antibiotics, vaccines, de-worming medications and shampoos.
- •
- advantus does not protect against ticks or mosquitoes.
-
Flea Infestations on Dogs:
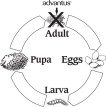
Treatment of flea infestations is important to your dog's health. Fleas in your dog's environment can re-infest your dog. Therefore, you may need to treat with advantus more than one time. The following diagram illustrates the flea's life cycle and where advantus works:
Fleas reproduce rapidly. A single female flea may produce up to 2,000 eggs over her lifetime. Eggs hatch and may grow into adults in only three weeks. Adult female fleas feed by ingesting blood from your dog and lay eggs that drop off from your dog's coat. Within days, larvae hatch from these eggs and live undetected in your dog's surroundings, such as in the carpet and bedding. Fleas can emerge to re-infest your pet, continuing the life cycle. When these new fleas are seen on your dog, treat with advantus.
If you have any questions about flea treatment or medical problems associated with flea infestations, consult your veterinarian.
- Storage Information:
-
How Supplied:
advantus is available in two strengths: 7.5 mg or 37.5 mg imidacloprid. Both strengths are packaged in 7 count bottles containing 7 flavored soft chews or 30 count bottles containing 30 flavored soft chews.
Approved by FDA under NADA # 141-435
Manufactured for:
Elanco US Inc.
Greenfield, IN 46140 U.S.A.90207038_PA600215X
Revised: January 2022
Made in Spain.
advantus, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
© 2022 Elanco or its affiliates
TAKE TIME
OBSERVE LABEL DIRECTIONS
ElancoTM
-
Principal Display Panel - 7.5 mg 7 Tablet Carton Label
ORAL FLEA
TREATMENT FOR
DOGS 10 WEEKS
OF AGE AND
OLDER,
BETWEEN
4–22 lbs.Elanco
advantusTM(imidacloprid) soft chew
7.5 mg
ORAL FLEA TREATMENT
Starts to kill fleas on dogs
within one hour7
SOFT
CHEWSEach soft chew contains
7.5 mg of imidacloprid.Approved by FDA under NADA # 141-435
KEEP OUT OF REACH
OF CHILDREN -
Principal Display Panel - 7.5 mg 30 Tablet Bottle Label
Peel
HereORAL FLEA
TREATMENT FOR
DOGS 10 WEEKS
OF AGE AND
OLDER,
BETWEEN
4–22 lbs.Elanco
advantus™(imidacloprid) soft chew
7.5 mg
ORAL FLEA TREATMENT
Starts to kill fleas on dogs
within one hour30
SOFT
CHEWSEach soft chew contains 7.5 mg of imidacloprid.
Approved by FDA under NADA # 141-435
KEEP OUT OF REACH
OF CHILDREN -
Principal Display Panel - 37.5 mg 7 Tablet Carton Label
ORAL FLEA
TREATMENT FOR
DOGS 10 WEEKS
OF AGE AND
OLDER,
BETWEEN
23–110 lbs.Elanco
advantusTM(imidacloprid) soft chew
37.5 mg
ORAL FLEA TREATMENT
Starts to kill fleas on dogs
within one hour7
SOFT
CHEWSEach soft chew contains
37.5 mg of imidacloprid.Approved by FDA under NADA # 141-435
KEEP OUT OF REACH
OF CHILDREN -
Principal Display Panel - 37.5 mg 30 Tablet Bottle Label
Peel
HereORAL FLEA
TREATMENT FOR
DOGS 10 WEEKS
OF AGE AND
OLDER,
BETWEEN
23–110 lbs.Elanco
advantus™(imidacloprid) soft chew
37.5 mg
ORAL FLEA TREATMENT
Starts to kill fleas on dogs
within one hour30
SOFT
CHEWSEach soft chew contains 37.5 mg of imidacloprid.
Approved by FDA under NADA # 141-435
KEEP OUT OF REACH
OF CHILDREN -
INGREDIENTS AND APPEARANCE
ADVANTUS
imidacloprid tablet, chewableProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:58198-0017 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IMIDACLOPRID (UNII: 3BN7M937V8) (IMIDACLOPRID - UNII:3BN7M937V8) IMIDACLOPRID 7.5 mg Product Characteristics Color red (red) Score no score Shape TRAPEZOID (TRAPEZOID) Size 14mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58198-0017-1 1 in 1 CARTON 1 NDC:58198-0017-2 7 in 1 BOTTLE 2 NDC:58198-0017-3 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141435 01/01/2017 ADVANTUS
imidacloprid tablet, chewableProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:58198-0018 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IMIDACLOPRID (UNII: 3BN7M937V8) (IMIDACLOPRID - UNII:3BN7M937V8) IMIDACLOPRID 37.5 mg Product Characteristics Color red (red) Score no score Shape TRAPEZOID (TRAPEZOID) Size 14mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58198-0018-1 1 in 1 CARTON 1 NDC:58198-0018-2 7 in 1 BOTTLE 2 NDC:58198-0018-3 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141435 01/01/2017 Labeler - Elanco US Inc. (966985624)