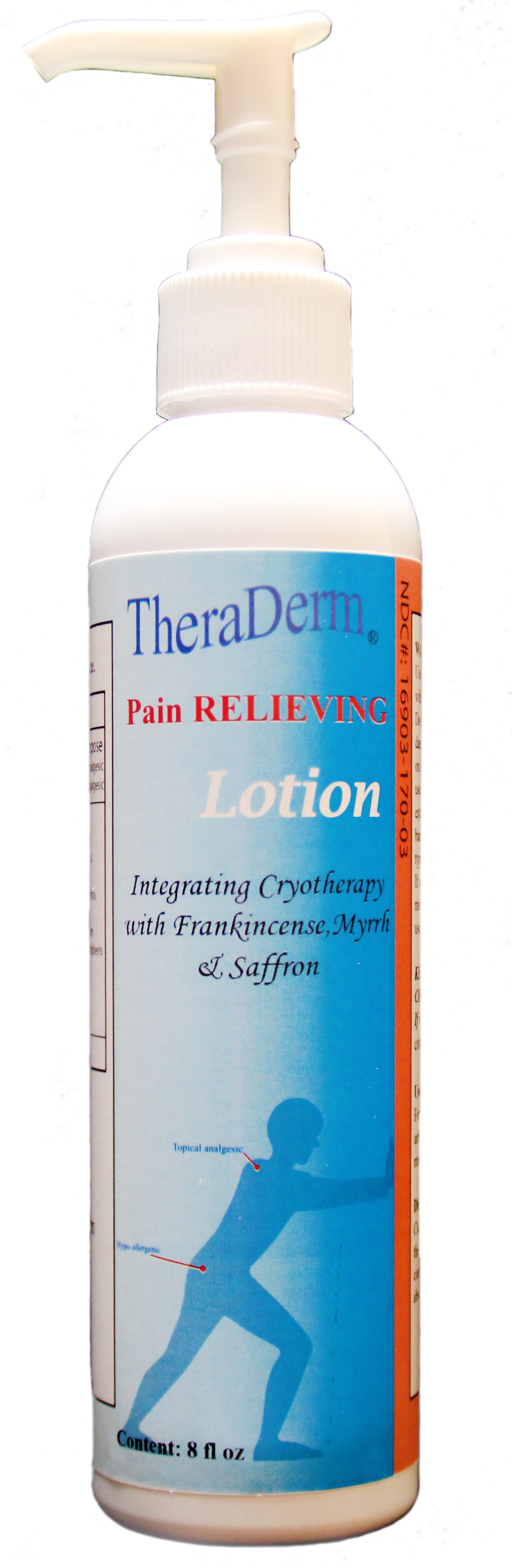
Label: THERA DERM- methyl salicylate lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 16903-170-03 - Packager: Manna Omni International Incorporated
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated February 19, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Directions:
- INSTRUCTIONS FOR USE
-
Warning:
for external use only
Use only as directed. Avoid contact with the eyes and mucous membranes. DO not apply to open wounds, damaged or very sensitive skin. Test on a small area before use. Do not use in combination with other external analgesic products. Do not bandage tightly or cover with any type of wrap, except clothing. If condition worsens, or persists for more than seven days, discontinue use and consult a physician.
- KEEP OUT OF THE REACH OF CHILDREN
- Active Ingredients:
-
Inactive Ingredients
Salvia Miltiorrhiza root, Carthamus Tinctorius Flower Oil, Lavender Oil, Angelica Actiloba Flowering Top, Aloe,Zingiber Cassumunar Root Oil,Angelicae Pubescens Root,Stearic Acid, Prunus Persica Flower Bud,Notopterygium Franchetii Root,Frankincense Oil, Ligusticum Sinese Subsc, Chuanxiong Root, Myrrh,nGlycerin, Vitamin E Oil,Coconut Oil.
- Purpose
- Indications and Usage
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
THERA DERM
methyl salicylate lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:16903-170 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR LEAF OIL (UNII: 51D0RGY52V) (CAMPHOR LEAF OIL - UNII:51D0RGY52V) CAMPHOR LEAF OIL 6 mg in 240 mg 7-AMINODESACETOXYCEPHALOSPORANIC ACID (UNII: ANM3MSM8TN) (7-AMINODESACETOXYCEPHALOSPORANIC ACID - UNII:ANM3MSM8TN) 7-AMINODESACETOXYCEPHALOSPORANIC ACID 450 mg in 240 mg MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 13 mg in 240 mg METHYL SALICYLATE 2-ETHYLBUTYRATE (UNII: J8D9175Q0G) (METHYL SALICYLATE 2-ETHYLBUTYRATE - UNII:J8D9175Q0G) METHYL SALICYLATE 2-ETHYLBUTYRATE 28 mg in 240 mg PEPPERMINT OIL (UNII: AV092KU4JH) (PEPPERMINT - UNII:V95R5KMY2B) PEPPERMINT OIL 13 mg in 240 mg Inactive Ingredients Ingredient Name Strength PAEONIA LACTIFLORA FLOWER (UNII: R73687A534) 4 mg in 240 mg SALVIA MILTIORRHIZA ROOT (UNII: 1693AM5SBN) 4 mg in 240 mg ANGELICA SINENSIS ROOT OIL (UNII: T8CL3168L1) 4 mg in 240 mg LIGUSTICUM SINENSE SUBSP. CHUANXIONG ROOT (UNII: RR83T99U97) 4 mg in 240 mg 12-HYDROXYSTEARIC ACID (UNII: 933ANU3H2S) 15 mg in 240 mg MYRRH OIL (UNII: H74221J5J4) 4 mg in 240 mg LAVENDER OIL (UNII: ZBP1YXW0H8) 3 mg in 240 mg PERSICARIA FILIFORMIS FLOWERING TOP (UNII: M4VKX9Q8AJ) 4 mg in 240 mg NOTOPTERYGIUM INCISUM ROOT (UNII: 5Z2WW4J6RI) 4 mg in 240 mg ANGELICA PUBESCENS ROOT (UNII: 0MF1EI01KJ) 4 mg in 240 mg ZINGIBER CASSUMUNAR ROOT OIL (UNII: O47HX41O6C) 4 mg in 240 mg COCONUT OIL (UNII: Q9L0O73W7L) 4 mg in 240 mg ALOE (UNII: V5VD430YW9) 7.5 mg in 240 mg CARTHAMUS TINCTORIUS FLOWER OIL (UNII: SDQ136WIM5) 4 mg in 240 mg FRANKINCENSE OIL (UNII: 67ZYA5T02K) 4 mg in 240 mg Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16903-170-03 240 mg in 1 BOTTLE; Type 0: Not a Combination Product 10/31/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/31/2017 Labeler - Manna Omni International Incorporated (019109468) Registrant - Manna Omni International Incorporated (019109468) Establishment Name Address ID/FEI Business Operations Manna Omni International Incorporated 019109468 manufacture(16903-170)