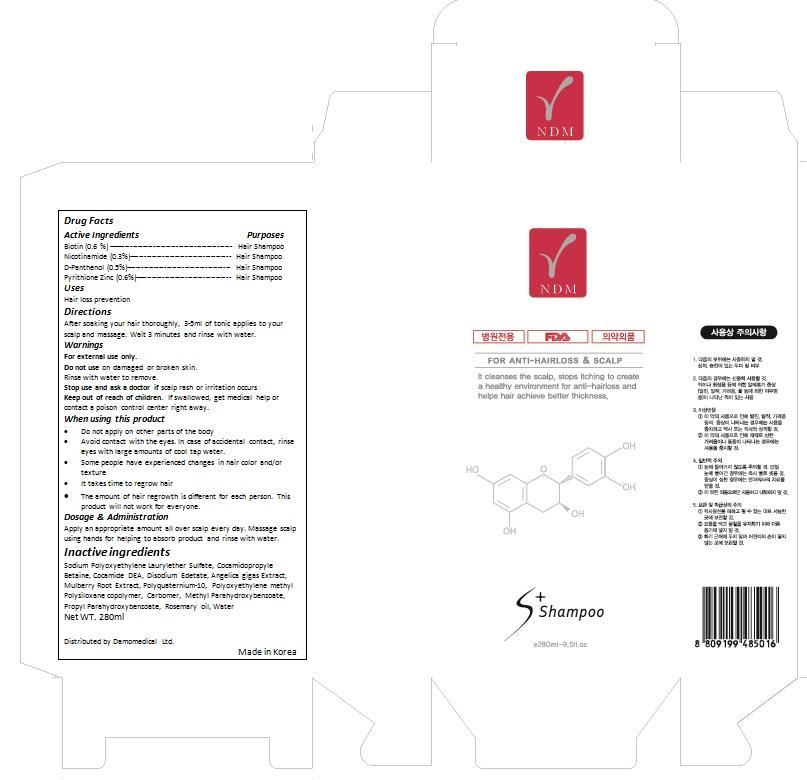
Label: NDM SCALP- biotin, nicotinamide, dpanthenol, pyrithione zinc shampoo
-
Contains inactivated NDC Code(s)
NDC Code(s): 69288-001-01, 69288-001-02 - Packager: Damomedical Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 27, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- PURPOSE
- INSTRUCTIONS FOR USE
- KEEP OUT OF REACH OF CHILDREN
- ASK DOCTOR
- WHEN USING
- DO NOT USE
-
INACTIVE INGREDIENT
Water, Butylene Glycol, Dipropylene Glycol, Glycerin, Alcohol, Trehalose, Lavandula Angustifolia (Lavender) Extract(1%), Diethoxyethyl Succinate, Bis-PEG-18 Methyl Ether Dimethyl Silane, Caprylic/Capric Triglyceride, PEG-60 Hydrogenated Castor Oil, Glyceryl Stearate, Sucrose Polystearate, Cetearyl Glucoside, Hydroxyethyl Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Carbomer, Phenoxyethanol, Sophora Angustifolia Root Extract, Lonicera Japonica (Honeysuckle) Flower Extract, Camellia Sinensis Leaf Extract, Behenyl Alcohol, Cetearyl Alcohol, Prunus Persica (Peach) Leaf Extract, Ginkgo Biloba Leaf Extract, Potassium Hydroxide, Fragrance, Ethylhexylglycerin, Scutellaria Baicalensis Root Extract, Hydrogenated Lecithin, Glyceryl Polyacrylate, Disodium EDTA, Hydrolyzed Collagen, Caprylyl Glycol, Glyceryl Caprylate, Sodium Hyaluronate
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NDM SCALP
biotin, nicotinamide, dpanthenol, pyrithione zinc shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69288-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 0.168 g in 280 mL PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 0.168 g in 280 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 0.84 g in 280 mL DEXPANTHENOL (UNII: 1O6C93RI7Z) (DEXPANTHENOL - UNII:1O6C93RI7Z) DEXPANTHENOL 1.4 g in 280 mL Inactive Ingredients Ingredient Name Strength SODIUM LAURETH-4 PHOSPHATE (UNII: 4N589ZVP40) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) EDETATE DISODIUM (UNII: 7FLD91C86K) ANGELICA GIGAS ROOT (UNII: 32766B2FHX) POLYQUATERNIUM-10 (1000 MPA.S AT 2%) (UNII: GMR4PEN8PK) CARBOMER 940 (UNII: 4Q93RCW27E) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) ROSEMARY OIL (UNII: 8LGU7VM393) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69288-001-02 1 in 1 CARTON 10/27/2014 1 NDC:69288-001-01 280 mL in 1 CONTAINER Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/27/2014 Labeler - Damomedical Ltd. (688277608) Registrant - Damomedical Ltd. (688277608) Establishment Name Address ID/FEI Business Operations Damomedical Ltd. 688277608 manufacture(69288-001)