Label: METHYLPHENIDATE HYDROCHLORIDE tablet, chewable
- NDC Code(s): 31722-926-01, 31722-927-01, 31722-928-01
- Packager: Camber Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CII
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
WARNING: ABUSE, MISUSE, AND ADDICTION
Methylphenidate hydrochloride chewable tablets has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride chewable tablets, can result in overdose and death ([see Overdosage]), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride chewable tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride chewable tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction (see Warnings and Precautions and Drug Abuse and Dependence).
-
DESCRIPTION
Methylphenidate hydrochloride chewable tablets are a mild central nervous system (CNS) stimulant, available as 2.5 mg, 5 mg and 10 mg for oral administration. Methylphenidate hydrochloride is methyl α-phenyl-2-piperidineacetate hydrochloride, and its structural formula is
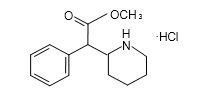
Methylphenidate Hydrochloride
C14H19NO2• HCl MW = 269.77
Methylphenidate hydrochloride USP is a white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone.
Each methylphenidate hydrochloride chewable tablet, for oral administration, contains 2.5 mg, 5 mg or 10 mg of methylphenidate hydrochloride USP. In addition, methylphenidate hydrochloride chewable tablets also contain the following inactive ingredients: aspartame, lactose anhydrous, microcrystalline cellulose, guar gum, grape flavor, pregelatinized starch, and stearic acid.
-
CLINICAL PHARMACOLOGY
Methylphenidate is a racemic mixture comprised of the d- and l-threo enantiomers. The d-threo enantiomer is more pharmacologically active than the l-threo enantiomer.
Methylphenidate HCl is a central nervous system (CNS) stimulant.
The mode of therapeutic action in humans is not completely understood, but methylphenidate presumably activates the brain stem arousal system and cortex to produce its stimulant effect. Methylphenidate is thought to block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
There is neither specific evidence which clearly establishes the mechanism whereby methylphenidate hydrochloride chewable tablets produces its mental and behavioral effects in children, nor conclusive evidence regarding how these effects relate to the condition of the central nervous system.
Pharmacokinetics
Absorption
Methylphenidate hydrochloride chewable tablets are readily absorbed. Following oral administration of methylphenidate hydrochloride chewable tablets, peak plasma methylphenidate concentrations are achieved at about 1 to 2 hours. Methylphenidate hydrochloride chewable tablets have been shown to be bioequivalent to Ritalin® tablet. The mean Cmax following a 20 mg dose is approximately 10 ng/mL.Food Effect
In a study in adult volunteers investigating the effects of a high-fat meal on the bioavailability of methylphenidate hydrochloride chewable tablets at a dose of 20 mg, the presence of food delayed the peak concentrations by approximately 1 hour (1.5 hours, fasted and 2.4 hours, fed). Overall, a high-fat meal increased the AUC of methylphenidate hydrochloride chewable tablets by about 20%, on average. Through a cross-study comparison, the magnitude of food effect is found to be comparable between the methylphenidate hydrochloride chewable tablets and Ritalin, the immediate release tablet.Metabolism and Excretion
In humans, methylphenidate is metabolized primarily via deesterification to alpha-phenylpiperidine acetic acid (PPA, ritalinic acid). The metabolite has little or no pharmacologic activity.After oral dosing of radiolabeled methylphenidate in humans, about 90% of the radioactivity was recovered in urine. The main urinary metabolite was PPA, accounting for approximately 80% of the dose.
The pharmacokinetics of the methylphenidate hydrochloride chewable tablets have been studied in healthy adult volunteers. The mean terminal half-life (t½) of methylphenidate following administration of 20 mg methylphenidate hydrochloride chewable tablets (t½ = 3 hours) is comparable to the mean terminal t½ following administration of Ritalin (methylphenidate hydrochloride immediate-release tablets) (t½ = 2.8 hours) in healthy adult volunteers.
Special Populations
Gender – The effect of gender on the pharmacokinetics of methylphenidate after methylphenidate hydrochloride chewable tablets administration has not been studied.Race – The influence of race on the pharmacokinetics of methylphenidate after methylphenidate hydrochloride chewable tablets administration has not been studied.
Age – The pharmacokinetics of methylphenidate after methylphenidate hydrochloride chewable tablets administration have not been studied in pediatrics.
Renal Insufficiency
There is no experience with the use of methylphenidate hydrochloride chewable tablets in patients with renal insufficiency. After oral administration of radiolabeled methylphenidate in humans, methylphenidate was extensively metabolized and approximately 80% of the radioactivity was excreted in the urine in the form of ritalinic acid. Since renal clearance is not an important route of methylphenidate clearance, renal insufficiency is expected to have little effect on the pharmacokinetics of methylphenidate hydrochloride chewable tablets.Hepatic Insufficiency
There is no experience with the use of methylphenidate hydrochloride chewable tablets in patients with hepatic insufficiency. -
INDICATIONS AND USAGE
Attention Deficit Disorders, Narcolepsy
Attention Deficit Disorders (previously known as Minimal Brain Dysfunction in Children). Other terms being used to describe the behavioral syndrome below include: Hyperkinetic Child Syndrome, Minimal Brain Damage, Minimal Cerebral Dysfunction, Minor Cerebral Dysfunction.Methylphenidate hydrochloride chewable tablets are indicated as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with a behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate-to-severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. The diagnosis of this syndrome should not be made with finality when these symptoms are only of comparatively recent origin. Nonlocalizing (soft) neurological signs, learning disability, and abnormal EEG may or may not be present, and a diagnosis of central nervous system dysfunction may or may not be warranted.
Special Diagnostic Considerations
Specific etiology of this syndrome is unknown, and there is no single diagnostic test. Adequate diagnosis requires the use not only of medical but of special psychological, educational, and social resources.Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, and moderate-to-severe hyperactivity; minor neurological signs and abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics.
Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in the child who exhibits symptoms secondary to environmental factors and/or primary psychiatric disorders, including psychosis. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medication will depend upon the physician’s assessment of the chronicity and severity of the child’s symptoms.
-
CONTRAINDICATIONS
Methylphenidate hydrochloride chewable tablets are contraindicated in patients known to be hypersensitive to methylphenidate or other components of the drug.
Methylphenidate hydrochloride chewable tablets are contraindicated during treatment with monoamine oxidase inhibitors, and also within a minimum of 14 days following discontinuation of a monoamine oxidase inhibitor (hypertensive crises may result).
-
WARNINGS
Abuse, Misuse, and Addiction
Methylphenidate hydrochloride chewable tablets has a high potential for abuse and misuse. The use of methylphenidate hydrochloride chewable tablets exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Methylphenidate hydrochloride chewable tablets can be diverted for non-medical use into illicit channels or distribution (see DRUG ABUSE and DEPENDENCE). Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride chewable tablets can result in overdose and death (see OVERDOSAGE), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.Before prescribing methylphenidate hydrochloride chewable tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store methylphenidate in a safe place, preferably locked, and instruct patients to not give methylphenidate hydrochloride chewable tablets to anyone else. Throughout methylphenidate hydrochloride chewable tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Risks to Patients with Serious Cardiac Disease
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who are treated with CNS stimulants at the recommended ADHD dosages.Avoid methylphenidate hydrochloride chewable tablets use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase about 2 to 4 mm Hg) and heart rate (mean increase about 3 to 6 bpm). Monitor all patients for potential tachycardia and hypertension.Psychiatric Adverse Reactions
Exacerbations of Pre-Existing Psychosis – CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a pre-existing psychotic disorder.Induction of a Manic Episode in Patients with Bipolar Disease – CNS stimulants may induce a manic or mixed episode in patients. Prior to initiating treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms – CNS stimulants, at recommended doses, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients, compared with 0% of placebo-treated patients. If such symptoms occur, consideration discontinuing methylphenidate hydrochloride chewable tablets.
Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in absence of seizures, and, very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, the drug should be discontinued.Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate products in both pediatric and adult patients. Priapism was not reported with drug initiation but developed after some time on the drug, often subsequent to an increase in dose. Priapism has also appeared during a period of drug withdrawal (drug holidays or discontinuation): Patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.Peripheral Vasculopathy, Including Raynaud’s Phenomenon
Stimulants, including methylphenidate hydrochloride chewable tablets, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in postmarketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.Long-Term Suppression of Growth in Pediatric Patients
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in methylphenidate hydrochloride chewable tablets-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted (see PRECAUTIONS, PEDIATRIC USE).
Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.Although the mechanism is not clear, methylphenidate hydrochloride chewable tablets-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment (see Adverse Reactions).Prescribe methylphenidate hydrochloride chewable tablets to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor methylphenidate hydrochloride chewable tablets-treated patients with a history of abnormally increased IOP or open angle glaucoma.
Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette’s syndrome has also been reported. Assess the family history and clinically evaluate patients for tics or Tourette’s syndrome before initiating methylphenidate hydrochloride chewable tablets. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome with methylphenidate hydrochloride chewable tablets, and discontinue treatment if clinically appropriate.USE IN CHILDREN LESS THAN SIX YEARS OF AGE
Methylphenidate hydrochloride chewable tablets should not be used in children under six years, since safety and efficacy in this age group have not been established. -
DRUG ABUSE AND DEPENDENCE
Controlled Substance
Methylphenidate hydrochloride chewable tablets contains methylphenidate, a Schedule II controlled substance.Abuse
Methylphenidate hydrochloride chewable tablets has a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction (see Warnings and Precautions). Methylphenidate hydrochloride chewable tablets can be diverted for non-medical use into illicit channels or distribution.Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of methylphenidate may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride chewable tablets, can result in overdose and death (see Overdosage), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Dependence
Physical Dependence
Methylphenidate hydrochloride chewable tablets may produce physical dependence. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.Withdrawal signs and symptoms after abrupt discontinuation or dose reduction following prolonged use of CNS stimulants including methylphenidate hydrochloride chewable tablets include dysphoric mood; depression; fatigue; vivid, unpleasant dreams; insomnia or hypersomnia; increased appetite; and psychomotor retardation or agitation.
Tolerance
Methylphenidate hydrochloride chewable tablets may produce tolerance. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose). -
PRECAUTIONS
General
Patients with an element of agitation may react adversely; discontinue therapy if necessary.Periodic CBC, differential, and platelet counts are advised during prolonged therapy.
Drug treatment is not indicated in all cases of this behavioral syndrome and should be considered only in light of the complete history and evaluation of the child. The decision to prescribe methylphenidate hydrochloride chewable tablets should depend on the physician’s assessment of the chronicity and severity of the child’s symptoms and their appropriateness for his/her age. Prescription should not depend solely on the presence of one or more of the behavioral characteristics.
When these symptoms are associated with acute stress reactions, treatment with methylphenidate hydrochloride chewable tablets are usually not indicated.
Long-term effects of methylphenidate hydrochloride chewable tablets in children have not been well established.
Information for Patients
Advise the patient to read the FDA-approved patient labeling (Medication Guide).Abuse, Misuse, and Addiction
Educate patients and their families about the risks of abuse, misuse, and addiction of methylphenidate hydrochloride chewable tablets, which can lead to overdose and death, and proper disposal of any unused drug (see WARNINGS and PRECAUTIONS, DRUG ABUSE AND DEPENDENCE, OVERDOSAGE). Advise patients to store methylphenidate hydrochloride chewable tablets in a safe place, preferably locked, and instruct patients to not give methylphenidate hydrochloride chewable tablets to anyone else.Risks to Patients with Serious Cardiac Disease
Advise patients that there are potential risks to patients with serious cardiac disease, including sudden death, with methylphenidate hydrochloride chewable tablets use. Instruct patients to contact a healthcare provider immediately if they develop symptoms such as exertional chest pain, unexplained syncope, or other symptoms suggestive of cardiac disease (see WARNINGS).Increased Blood Pressure and Heart Rate
Advise patients that methylphenidate hydrochloride chewable tablets can elevate blood pressure and heart rate (see WARNINGS).Psychiatric Adverse Reactions
Advise patients that methylphenidate hydrochloride chewable tablets, at recommended doses, can cause psychotic or manic symptoms, even in patients without a prior history of psychotic symptoms or mania (see WARNINGS).Priapism
Advise patients, caregivers, and family members of the possibility of painful or prolonged penile erections (priapism). Instruct the patient to seek immediate medical attention in the event of priapism.Circulation Problems in Fingers and Toes [Peripheral Vasculopathy, Including Raynaud’s Phenomenon]
- Instruct patients beginning treatment with methylphenidate hydrochloride chewable tablets about the risk of peripheral vasculopathy, including Raynaud’s phenomenon, and associated signs and symptoms: fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red.
- Instruct patients to report to their physician any new numbness, pain, skin color change, or sensitivity to temperature in fingers or toes.
- Instruct patients to call their physician immediately with any signs of unexplained wounds appearing on fingers or toes while taking methylphenidate hydrochloride chewable tablets.
- Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
Suppression of Growth in Pediatric Patients
Advise patients, caregivers, and family members that methylphenidate hydrochloride chewable tablets can cause slowing of growth and weight loss (see Warnings).Increased Intraocular Pressure and Glaucoma
Advise patients that IOP and glaucoma may occur during treatment with methylphenidate hydrochloride chewable tablets (see Warnings).Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
Advise patients that motor and verbal tics and worsening of Tourette’s Syndrome may occur during treatment with methylphenidate hydrochloride chewable tablets. Instruct the patients to notify their healthcare provider if emergence or worsening of tics or Tourette’s syndrome occurs (see WARNINGS).Choking – Taking this product without adequate fluid may cause it to swell and block your throat or esophagus and may cause choking. Do not take this product if you have difficulty in swallowing. If you experience chest pain, vomiting, or difficulty in swallowing or breathing after taking this product, seek immediate medical attention.
Directions – Take this product (child or adult dose) with at least 8 ounces (a full glass) of water or other fluid. Taking this product without enough liquid may cause choking. See choking warning.
Phenylketonurics – Phenylalanine is a component of aspartame. Each 2.5 mg methylphenidate hydrochloride chewable tablet contains 0.35 mg of phenylalanine; each 5 mg methylphenidate hydrochloride chewable tablet contains 0.70 mg of phenylalanine and each 10 mg methylphenidate hydrochloride chewable tablet contains 1.40 mg of phenylalanine.
Drug Interactions
Monoamine Oxidase Inhibitors (MAOI)
Concomitant use of MAOIs and CNS stimulants, including methylphenidate hydrochloride chewable tablets can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure (see CONTRAINDICATIONS). Concomitant use of methylphenidate hydrochloride chewable tablets with MAOIs or within 14 days after discontinuing MAOI treatment is contraindicated.Antihypertensive Drugs
Methylphenidate hydrochloride chewable tablets may decrease the effectiveness of drugs used to treat hypertension. Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed.Risperidone
Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS). Monitor for signs of EPS.Methylphenidate hydrochloride chewable tablets may decrease the hypotensive effect of guanethidine. Use cautiously with pressor agents.
Human pharmacologic studies have shown that methylphenidate hydrochloride chewable tablets may inhibit the metabolism of coumarin anticoagulants, anticonvulsants (phenobarbital, diphenylhydantoin, primidone), phenylbutazone, and tricyclic drugs (imipramine, clomipramine, desipramine). Downward dosage adjustments of these drugs may be required when given concomitantly with methylphenidate hydrochloride chewable tablets.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a lifetime carcinogenicity study carried out in B6C3F1 mice, methylphenidate caused an increase in hepatocellular adenomas and, in males only, an increase in hepatoblastomas, at a daily dose of approximately 60 mg/kg/day. This dose is approximately 30 times and 2.5 times the maximum recommended human dose on a mg/kg and mg/m2 basis, respectively. Hepatoblastoma is a relatively rare rodent malignant tumor type. There was no increase in total malignant hepatic tumors. The mouse strain used is sensitive to the development of hepatic tumors, and the significance of these results to humans is unknown.Methylphenidate did not cause any increase in tumors in a lifetime carcinogenicity study carried out in F344 rats; the highest dose used was approximately 45 mg/kg/day, which is approximately 22 times and 4 times the maximum recommended human dose on a mg/kg and mg/m2 basis, respectively.
Methylphenidate was not mutagenic in the in vitro Ames reverse mutation assay or in the in vitro mouse lymphoma cell forward mutation assay. Sister chromatid exchanges and chromosome aberrations were increased, indicative of a weak clastogenic response, in an in vitro assay in cultured Chinese Hamster Ovary (CHO) cells. The genotoxic potential of methylphenidate has not been evaluated in an in vivo assay.
Usage in Pregnancy
Adequate animal reproduction studies to establish safe use of methylphenidate hydrochloride chewable tablet during pregnancy have not been conducted. However, in a recently conducted study, methylphenidate has been shown to have teratogenic effects in rabbits when given in doses of 200 mg/kg/day, which is approximately 167 times and 78 times the maximum recommended human dose on a mg/kg and a mg/m2 basis, respectively. In rats, teratogenic effects were not seen when the drug was given in doses of 75 mg/kg/day, which is approximately 62.5 and 13.5 times the maximum recommended human dose on a mg/kg and a mg/m2 basis, respectively. Therefore, until more information is available, methylphenidate should not be prescribed for women of childbearing age unless, in the opinion of the physician, the potential benefits outweigh the possible risks. -
ADVERSE REACTIONS
Nervousness and insomnia are the most common adverse reactions but are usually controlled by reducing dosage and omitting the drug in the afternoon or evening. Other reactions include hypersensitivity (including skin rash, urticaria, fever, arthralgia, exfoliative dermatitis, erythema multiforme with histopathological findings of necrotizing vasculitis, and thrombocytopenic purpura); anorexia; nausea; dizziness; palpitations; headache; dyskinesia; drowsiness; blood pressure and pulse changes, both up and down; tachycardia; angina; cardiac arrhythmia; abdominal pain; weight loss during prolonged therapy; libido changes; and rhabdomyolysis increased intraocular pressure and glaucoma, motor and verbal tics. There have been rare reports of Tourette’s syndrome. Toxic psychosis has been reported. Although a definite causal relationship has not been established, the following have been reported in patients taking this drug: instances of abnormal liver function, ranging from transaminase elevation to severe hepatic injury; isolated cases of cerebral arteritis and/or occlusion; leukopenia and/or anemia; transient depressed mood; a few instances of scalp hair loss; serotonin syndrome in combination with serotonergic drugs. Very rare reports of neuroleptic malignant syndrome (NMS) have been received, and, in most of these, patients were concurrently receiving therapies associated with NMS. In a single report, a ten year old boy who had been taking methylphenidate for approximately 18 months experienced an NMS-like event within 45 minutes of ingesting his first dose of venlafaxine. It is uncertain whether this case represented a drug-drug interaction, a response to either drug alone, or some other cause.
In children, loss of appetite, abdominal pain, weight loss during prolonged therapy, insomnia, and tachycardia may occur more frequently; however, any of the other adverse reactions listed above may also occur.
-
OVERDOSAGE
Clinical Effects of Overdose
Overdose of CNS stimulants is characterized by the following sympathomimetic effects:- Cardiovascular effects including tachyarrhythmias, and hypertension or hypotension. Vasospasm, myocardial infarction, or aortic dissection may precipitate sudden cardiac death. Takotsubo cardiomyopathy may develop.
- CNS effects including psychomotor agitation, confusion, and hallucinations. Serotonin syndrome, seizures, cerebral vascular accidents, and coma may occur.
- Life-threatening hyperthermia (temperatures greater than 104°F) and rhabdomyolysis may develop.
Overdose Management
Consider the possibility of multiple drug ingestion. Because methylphenidate has a large volume of distribution and is rapidly metabolized, dialysis is not useful. Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations. -
DOSAGE AND ADMINISTRATION
Prior to treating patients with Methylphenidate hydrochloride chewable tablets assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) (see WARNINGS).
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome (see WARNINGS).
Dosage should be individualized according to the needs and responses of the patient.
Directions – Take this product (child or adult dose) with at least 8 ounces (a full glass) of water or other fluid. Taking this product without enough liquid may cause choking. See choking warning.
Adults
Administer in divided doses 2 or 3 times daily, preferably 30 to 45 minutes before meals. Average dosage is 20 to 30 mg daily. Some patients may require 40 to 60 mg daily. In others, 10 to 15 mg daily will be adequate. Patients who are unable to sleep if medication is taken late in the day should take the last dose before 6 p.m.Children (6 years and over)
Methylphenidate hydrochloride chewable tablets should be initiated in small doses, with gradual weekly increments. Daily dosage above 60 mg is not recommended.If paradoxical aggravation of symptoms or other adverse effects occur, reduce dosage, or, if necessary, discontinue methylphenidate hydrochloride chewable tablets. Methylphenidate hydrochloride chewable tablets should be periodically discontinued to assess the patient’s condition. If improvement is not observed after appropriate dosage adjustment over a one-month period, the drug should be discontinued.
Start with 5 mg twice daily (before breakfast and lunch) with gradual increments of 5 to 10 mg weekly.
Drug treatment should not and need not be indefinite and usually may be discontinued after puberty.
-
HOW SUPPLIED
Each methylphenidate hydrochloride chewable tablet 2.5 mg is available as a white to off-white round beveled edge chewable tablets debossed with '260' on one side and 'AT' on the other side.
Bottles of 100 NDC 31722-926-01
Each methylphenidate hydrochloride chewable tablet 5 mg is available as a white to off-white round beveled edge chewable tablets debossed with '261' on one side and 'AT' on the other side.
Bottles of 100 NDC 31722-927-01
Each methylphenidate hydrochloride chewable tablet 10 mg is available as a white to off-white flat round chewable tablets debossed with 'AT' on left side and '262' on the right side of the bisect line and other side plain.
Bottles of 100 NDC 31722-928-01
Protect from moisture. Dispense in tight container with child-resistant closure.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].Ritalin® is a registered trademark of Novartis Corporation.
Manufactured by:
Ascent Pharmaceuticals, Inc.
Central Islip, NY 11722Manufactured for:
Camber Pharmaceuticals, Inc.
Piscataway, NJ 08854Rev: 09/23
-
MEDICATION GUIDE Methylphenidate HCl Chewable Tablet, CII(meth'' il fen' i date hye'' droe klor' ide)
Read the Medication Guide that comes with methylphenidate hydrochloride chewable tablets before you or your child starts taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your or your child’s treatment with methylphenidate hydrochloride chewable tablets.
What is the most important information I should know about methylphenidate hydrochloride chewable tablets?
Methylphenidate hydrochloride chewable tablets may cause serious side effects, including:
- Abuse, misuse, and addiction. Methylphenidate hydrochloride chewable tablets have a high chance for abuse and misuse and may lead to substance use problems, including addiction. Misuse and abuse of methylphenidate hydrochloride chewable tablets, other methylphenidate containing medicines, and amphetamine containing medicines, can lead to overdose and death. The risk of overdose and death is increased with higher doses of methylphenidate hydrochloride chewable tablets or when it is used in ways that are not approved, such as snorting or injection.

- Heart-related problems:

- Mental (Psychiatric) problems:

- Circulation problems in fingers and toes [Peripheral vasculopathy, including Raynaud’s phenomenon]: fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red.

What Are methylphenidate hydrochloride chewable tablets?
Methylphenidate hydrochloride chewable tablets are a central nervous system stimulant prescription medicine. Methylphenidate hydrochloride chewable tablets are tablets that are made to be chewed and swallowed. They are used for the treatment of Attention Deficit and Hyperactivity Disorder (ADHD). Methylphenidate hydrochloride chewable tablets may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD.Methylphenidate hydrochloride chewable tablets should be used as a part of a total treatment program for ADHD that may include counseling or other therapies.
Methylphenidate hydrochloride chewable tablets are also used in the treatment of a sleep disorder called narcolepsy.
Methylphenidate hydrochloride chewable tablets are a federally controlled substance (CII) because it contains methylphenidate that can be a target for people who abuse prescription medicines or street drugs. Keep methylphenidate hydrochloride chewable tablets in a safe place to protect it from theft. Never give your methylphenidate hydrochloride chewable tablets to anyone else, because it may cause death or harm them. Selling or giving away methylphenidate hydrochloride chewable tablets may harm others, and is against the law.
Who should not take methylphenidate hydrochloride chewable tablets?
Methylphenidate hydrochloride chewable tablets should not be taken if you or your child:- are very anxious, tense, or agitated
- have eye problems, including increased pressure in your eye, glaucoma, or problems with your close-up vision (farsightedness)
- have or had repeated movements or sounds (tics) or Tourette’s syndrome, or have a family history of tics or Tourette’s syndrome
- are taking or have taken within the past 14 days an antidepression medicine called a monoamine oxidase inhibitor or MAOI.
- are allergic to anything in methylphenidate hydrochloride chewable tablets. See the end of this Medication Guide for a complete list of ingredients.
Methylphenidate hydrochloride chewable tablets should not be used in children less than 6 years old because they have not been studied in this age group.
Methylphenidate hydrochloride chewable tablets may not be right for you or your child. Before starting methylphenidate hydrochloride chewable tablets tell your or your child’s healthcare provider about all health conditions (or a family history of) including:
- have heart problems, heart disease, heart defects, or high blood pressure
- mental problems including psychosis, mania, bipolar illness, or depression
- tics or Tourette’s syndrome
- seizures or have had an abnormal brain wave test (EEG)
- circulation problems in fingers and toes
Tell your healthcare provider if you or your child is pregnant, planning to become pregnant, or breastfeeding.
Can methylphenidate hydrochloride chewable tablets be taken with other medicines?
Tell your healthcare provider about all of the medicines that you or your child take including prescription and nonprescription medicines, vitamins, and herbal supplements.
Methylphenidate hydrochloride chewable tablets and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted while taking methylphenidate hydrochloride chewable tablets.
Your healthcare provider will decide whether methylphenidate hydrochloride chewable tablets can be taken with other medicines.
Especially tell your healthcare provider if you or your child takes:
- antidepression medicines including MAOIs
- seizure medicines
- blood thinner medicines
- blood pressure medicines
- cold or allergy medicines that contain decongestants
Know the medicines that you or your child takes. Keep a list of your medicines with you to show your healthcare provider and pharmacist.
Do not start any new medicine while taking methylphenidate hydrochloride chewable tablets without talking to your healthcare provider first.
How should methylphenidate hydrochloride chewable tablets be taken?
- Take methylphenidate hydrochloride chewable tablets exactly as prescribed. Your healthcare provider may adjust the dose until it is right for you or your child.
- Methylphenidate hydrochloride chewable tablets are usually taken 2 to 3 times a day.
- Take methylphenidate hydrochloride chewable tablets 30 to 45 minutes before a meal.
- Chew methylphenidate hydrochloride chewable tablets well and swallow with at least 8 ounces (a full glass) of water or other liquid. Methylphenidate hydrochloride chewable tablets can swell and cause choking if enough liquid is not taken with them. Get emergency medical care if you have chest pain, vomiting, or trouble swallowing, or breathing after taking a methylphenidate hydrochloride chewable tablet.
- From time to time, your healthcare provider may stop methylphenidate hydrochloride chewable tablets treatment for awhile to check ADHD symptoms.
- Your healthcare provider may do regular checks of the blood, heart, and blood pressure while you are taking methylphenidate hydrochloride chewable tablets. Children should have their height and weight checked often while taking methylphenidate hydrochloride chewable tablets. Methylphenidate hydrochloride chewable tablets treatment may be stopped if a problem is found during these check-ups.
If you or your child take too much methylphenidate hydrochloride chewable tablets, call your healthcare provider or Poison Helpline, at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What are possible side effects of methylphenidate hydrochloride chewable tablets?
See “What is the most important information I should know about methylphenidate hydrochloride chewable tablets?” for information on reported heart and mental problems.
Other serious side effects include:- Slowing of growth (height and weight) in children. Children should have their height and weight checked often during treatment with methylphenidate hydrochloride chewable tablets. Your healthcare provider may stop your child’s methylphenidate hydrochloride chewable tablets treatment if they are not growing or gaining weight as expected.
- Seizures, mainly in patients with a history of seizures
- Painful and prolonged erections (priapism) have occurred with methylphenidate. If you or your child develop priapism, seek medical help right away. Because of the potential for lasting damage, priapism should be evaluated by a healthcare provider immediately.
- Circulation problems in fingers and toes (peripheral vasculopathy, including Raynaud’s phenomenon): fingers or toes may feel numb, cool, painful, and/or may change color from pale, to blue, to red. Tell your healthcare provider if you have or your child has numbness, pain, skin color change, or sensitivity to temperature in your fingers or toes or if you have or your child has any signs of unexplained wounds appearing on fingers or toes while taking methylphenidate hydrochloride chewable tablets.
- Eyes problems (increased pressure in the eye and glaucoma). Call your healthcare provider right away if you or your child develop changes in your vision or eye pain, swelling, or redness.
- New or worsening tics or worsening Tourette’s syndrome. Tell your healthcare provider if you or your child get any new or worsening tics or worsening Tourette’s syndrome during treatment with methylphenidate hydrochloride chewable tablets.
Common side effects include:
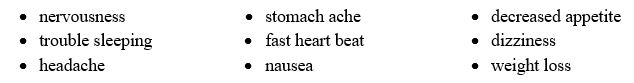
Talk to your healthcare provider if you or your child has side effects that are bothersome or do not go away.
This is not a complete list of possible side effects. Ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store methylphenidate hydrochloride chewable tablets?
- Store methylphenidate hydrochloride chewable tablets in a safe place at room temperature, at 20° to 25°C (68° to 77°F). [see USP Controlled Room Temperature]. Protect from moisture.
- Dispose of remaining, unused, or expired methylphenidate hydrochloride chewable tablets by a medicine take back program at a U.S. Drug Enforcement Administration (DEA) authorized collection site. If no takeback program or DEA authorized collector is available, mix methylphenidate hydrochloride chewable tablets with an undesirable, nontoxic substance such as dirt, cat litter, or used coffee grounds to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and throw away methylphenidate hydrochloride chewable tablets in the household trash. Visit www.fda.gov/drugdisposal for additional information on disposal of unused medicines.
- Keep methylphenidate hydrochloride chewable tablets and all medicines out of the reach of children.
General information about methylphenidate hydrochloride chewable tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use methylphenidate hydrochloride chewable tablets for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride chewable tablets to other people, even if they have the same condition. It may harm them and it is against the law.
This Medication Guide summarizes the most important information about methylphenidate hydrochloride chewable tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about methylphenidate hydrochloride chewable tablets that is written for healthcare professionals. For more information, please contact Camber Pharmaceuticals, Inc. at 1-866-495-8330.What are the ingredients in methylphenidate hydrochloride chewable tablets?
CAUTION PHENYLKETONURICS: Methylphenidate hydrochloride chewable tablets contain phenylalanine.
Active Ingredient: methylphenidate hydrochloride USP
Inactive Ingredients: aspartame, lactose anhydrous, microcrystalline cellulose, guar gum, grape flavor, pregelatinized starch, and stearic acid.
Medication Guide available at http://camberpharma.com/medication-guides
Manufactured by:
Ascent Pharmaceuticals, Inc.
Central Islip, NY 11722Manufactured for:
Camber Pharmaceuticals, Inc.
Piscataway, NJ 08854This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised 09/2023
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
METHYLPHENIDATE HYDROCHLORIDE
methylphenidate hydrochloride tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:31722-926 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 2.5 mg Inactive Ingredients Ingredient Name Strength ASPARTAME (UNII: Z0H242BBR1) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) GUAR GUM (UNII: E89I1637KE) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white (white to off-white) Score no score Shape ROUND Size 7mm Flavor grape Imprint Code 260;AT Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:31722-926-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210354 03/01/2018 METHYLPHENIDATE HYDROCHLORIDE
methylphenidate hydrochloride tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:31722-927 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength ASPARTAME (UNII: Z0H242BBR1) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) GUAR GUM (UNII: E89I1637KE) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white (white to off-white) Score no score Shape ROUND Size 9mm Flavor grape Imprint Code 261;AT Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:31722-927-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210354 03/01/2018 METHYLPHENIDATE HYDROCHLORIDE
methylphenidate hydrochloride tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:31722-928 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLPHENIDATE HYDROCHLORIDE (UNII: 4B3SC438HI) (METHYLPHENIDATE - UNII:207ZZ9QZ49) METHYLPHENIDATE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength ASPARTAME (UNII: Z0H242BBR1) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) GUAR GUM (UNII: E89I1637KE) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white (white to off-white) Score 2 pieces Shape ROUND Size 13mm Flavor grape Imprint Code AT;262 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:31722-928-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210354 03/01/2018 Labeler - Camber Pharmaceuticals Inc. (826774775) Registrant - Ascent Pharmaceuticals Inc (080938961) Establishment Name Address ID/FEI Business Operations Ascent Pharmaceuticals Inc 080938961 manufacture(31722-926, 31722-927, 31722-928) , analysis(31722-926, 31722-927, 31722-928) , pack(31722-926, 31722-927, 31722-928)






