Label: DIMETHYL FUMARATE- dimethyl fumarate kit
DIMETHYL FUMARATE capsule, delayed release
- NDC Code(s): 70771-1530-7, 70771-1531-6, 70771-1531-8, 70771-1532-3
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 13, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
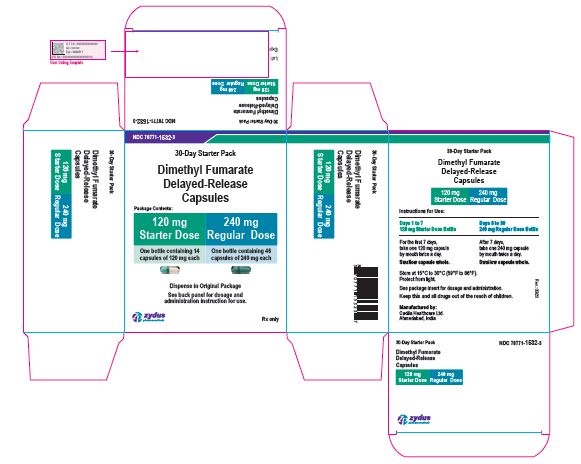
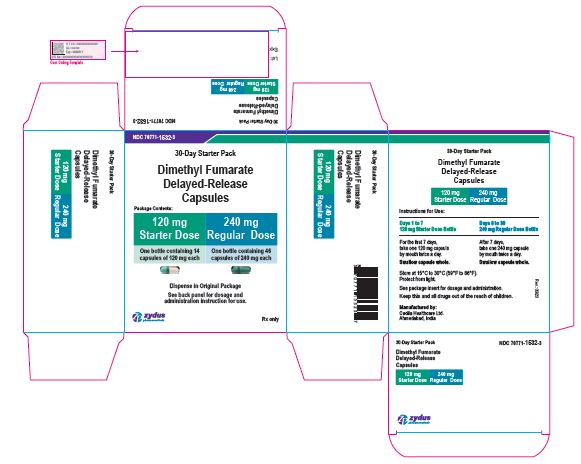
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIMETHYL FUMARATE
dimethyl fumarate kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1532 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1532-3 1 in 1 KIT; Type 0: Not a Combination Product 09/28/2020 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 14 Part 2 1 BOTTLE 46 Part 1 of 2 DIMETHYL FUMARATE
dimethyl fumarate capsule, delayed releaseProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHYL FUMARATE (UNII: FO2303MNI2) (MONOMETHYL FUMARATE - UNII:45IUB1PX8R) DIMETHYL FUMARATE 120 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) GLYCERYL MONO AND DICAPRYLOCAPRATE (UNII: U72Q2I8C85) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID - ETHYL ACRYLATE COPOLYMER (1:1) TYPE A (UNII: NX76LV5T8J) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color GREEN (OPAQUE LIGHT GREEN CAP) , WHITE (OPAQUE WHITE BODY) Score no score Shape CAPSULE (CAPSULE) Size 22mm Flavor Imprint Code 1204 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 14 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210538 09/28/2020 Part 2 of 2 DIMETHYL FUMARATE
dimethyl fumarate capsule, delayed releaseProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHYL FUMARATE (UNII: FO2303MNI2) (MONOMETHYL FUMARATE - UNII:45IUB1PX8R) DIMETHYL FUMARATE 240 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) GLYCERYL MONO AND DICAPRYLOCAPRATE (UNII: U72Q2I8C85) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID - ETHYL ACRYLATE COPOLYMER (1:1) TYPE A (UNII: NX76LV5T8J) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color GREEN (OPAQUE LIGHT GREEN CAP) , GREEN (OPAQUE LIGHT GREEN BODY) Score no score Shape CAPSULE (CAPSULE) Size 22mm Flavor Imprint Code 1205 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 46 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210538 09/28/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210538 09/28/2020 DIMETHYL FUMARATE
dimethyl fumarate capsule, delayed releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1530 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHYL FUMARATE (UNII: FO2303MNI2) (MONOMETHYL FUMARATE - UNII:45IUB1PX8R) DIMETHYL FUMARATE 120 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) CAPRYLIC/CAPRIC MONO/DI-GLYCERIDES (UNII: U72Q2I8C85) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) METHACRYLIC ACID AND ETHYL ACRYLATE COPOLYMER (UNII: NX76LV5T8J) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color GREEN (OPAQUE LIGHT GREEN CAP) , WHITE (OPAQUE WHITE BODY) Score no score Shape CAPSULE (CAPSULE) Size 22mm Flavor Imprint Code 1204 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1530-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 09/28/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210538 09/28/2020 DIMETHYL FUMARATE
dimethyl fumarate capsule, delayed releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1531 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHYL FUMARATE (UNII: FO2303MNI2) (MONOMETHYL FUMARATE - UNII:45IUB1PX8R) DIMETHYL FUMARATE 240 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) CAPRYLIC/CAPRIC MONO/DI-GLYCERIDES (UNII: U72Q2I8C85) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) METHACRYLIC ACID AND ETHYL ACRYLATE COPOLYMER (UNII: NX76LV5T8J) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color GREEN (OPAQUE LIGHT GREEN CAP) , GREEN (OPAQUE LIGHT GREEN BODY) Score no score Shape CAPSULE (CAPSULE) Size 22mm Flavor Imprint Code 1205 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1531-8 46 in 1 BOTTLE; Type 0: Not a Combination Product 09/28/2020 2 NDC:70771-1531-6 60 in 1 BOTTLE; Type 0: Not a Combination Product 09/28/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210538 09/28/2020 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Lifesciences Limited (863362789) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 863362789 ANALYSIS(70771-1530, 70771-1531, 70771-1532) , MANUFACTURE(70771-1530, 70771-1531, 70771-1532)