Label: TEMOZOLOMIDE capsule
-
NDC Code(s):
70771-1092-6,
70771-1092-7,
70771-1093-6,
70771-1093-7, view more70771-1094-6, 70771-1094-7, 70771-1095-6, 70771-1095-7, 70771-1096-6, 70771-1096-7, 70771-1097-6, 70771-1097-7
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
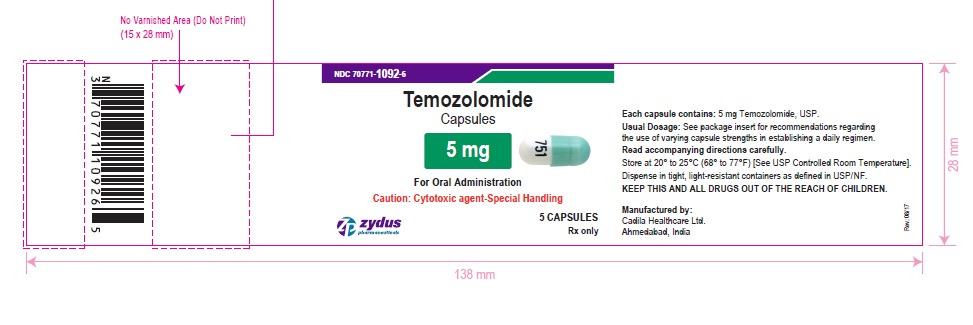
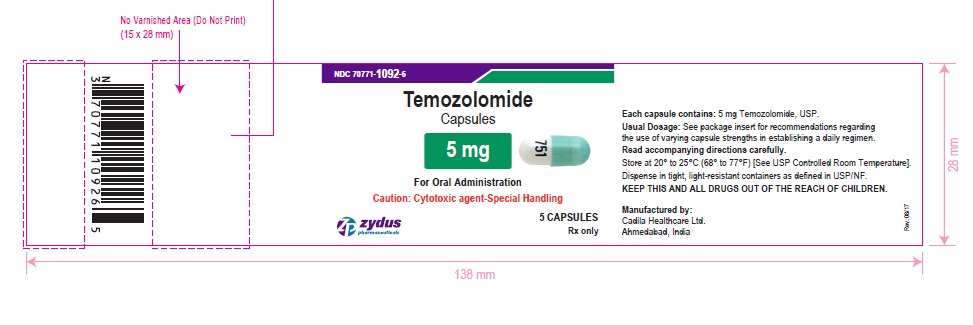
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 70771-1092-6 in bottle of 5 capsules
Temozolomide Capsules, 5 mg
Rx only
5 capsules
NDC 70771-1093-6 in bottle of 5 capsules
Temozolomide Capsules, 20 mg
Rx only
5 capsules

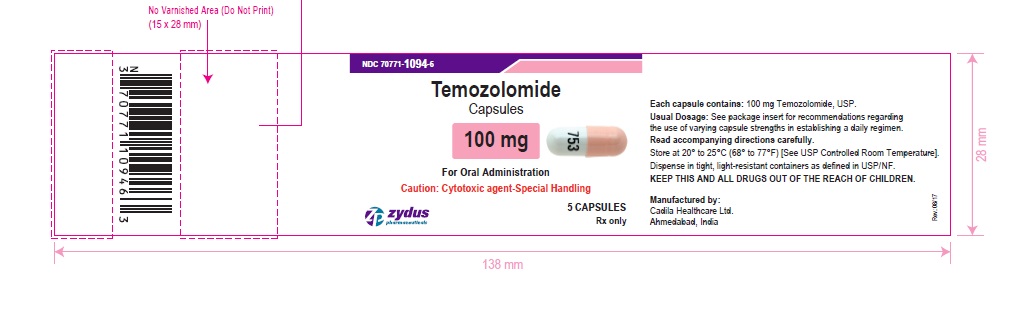
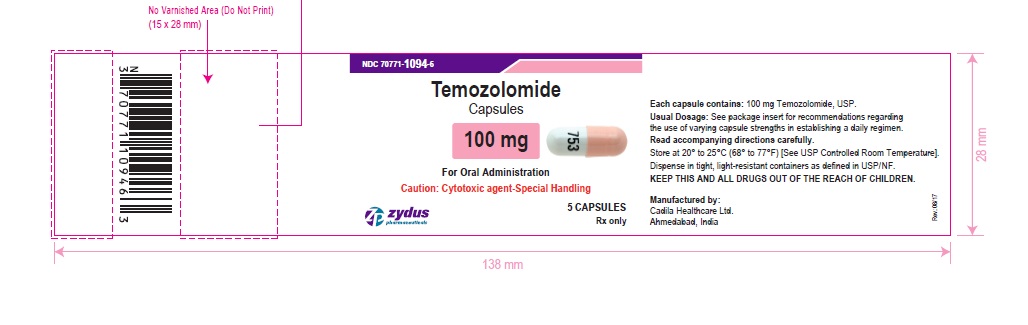
NDC 70771-1094-6 in bottle of 5 capsules
Temozolomide Capsules, 100 mg
Rx only
5 capsules
NDC 70771-1095-6 in bottle of 5 capsules
Temozolomide Capsules, 140 mg
Rx only
5 capsules

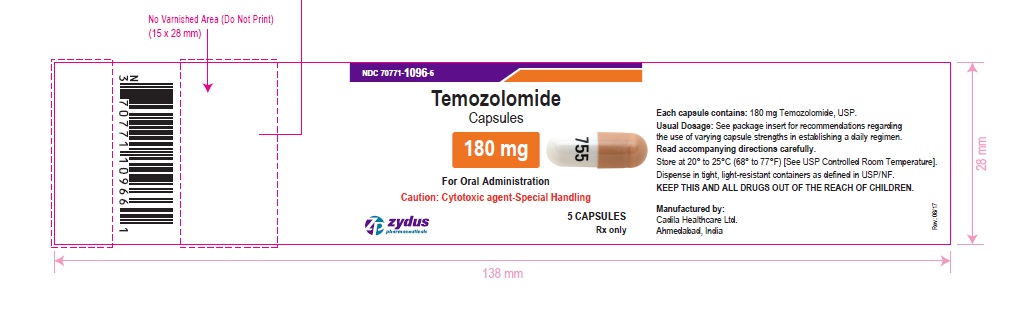
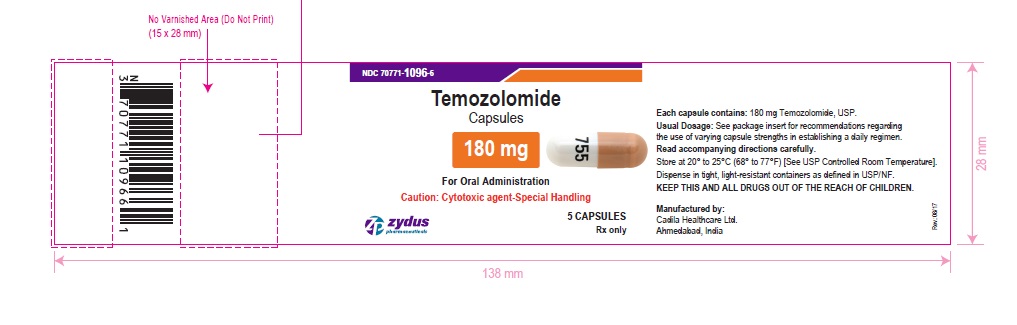
NDC 70771-1096-6 in bottle of 5 capsules
Temozolomide Capsules, 180 mg
Rx only
5 capsules
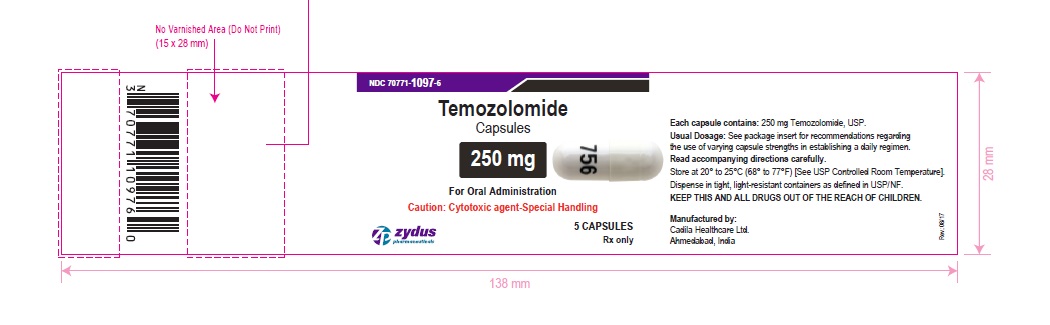
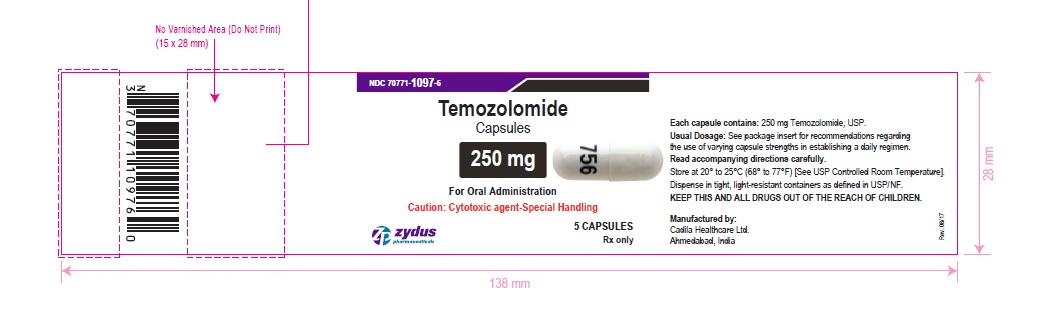
NDC 70771-1097-6 in bottle of 5 capsules
Temozolomide Capsules, 250 mg
Rx only
5 capsules
-
INGREDIENTS AND APPEARANCE
TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1092 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 5 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) STEARIC ACID (UNII: 4ELV7Z65AP) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Product Characteristics Color GREEN (GREEN) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 11mm Flavor Imprint Code 751 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1092-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1092-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1093 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 20 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) STEARIC ACID (UNII: 4ELV7Z65AP) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Product Characteristics Color YELLOW (YELLOW) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 11mm Flavor Imprint Code 752 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1093-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1093-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1094 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 100 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) TARTARIC ACID (UNII: W4888I119H) STEARIC ACID (UNII: 4ELV7Z65AP) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) ALCOHOL (UNII: 3K9958V90M) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) AMMONIA (UNII: 5138Q19F1X) WATER (UNII: 059QF0KO0R) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM LAURYL SULFATE (UNII: 368GB5141J) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color PINK (PINK) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 16mm Flavor Imprint Code 753 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1094-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1094-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1095 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 140 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) STEARIC ACID (UNII: 4ELV7Z65AP) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Product Characteristics Color BLUE (BLUE) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 18mm Flavor Imprint Code 754 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1095-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1095-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1096 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 180 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) STEARIC ACID (UNII: 4ELV7Z65AP) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Product Characteristics Color ORANGE (ORANGE) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 21mm Flavor Imprint Code 755 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1096-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1096-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1097 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 250 mg Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) STEARIC ACID (UNII: 4ELV7Z65AP) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Product Characteristics Color WHITE (WHITE) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 24mm Flavor Imprint Code 756 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1097-6 5 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 2 NDC:70771-1097-7 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/26/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206750 10/26/2017 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Lifesciences Limited (918596198) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 863362789 ANALYSIS(70771-1092, 70771-1093, 70771-1094, 70771-1095, 70771-1096, 70771-1097) , MANUFACTURE(70771-1092, 70771-1093, 70771-1094, 70771-1095, 70771-1096, 70771-1097)