Label: BIOGLO- fluorescein sodium strip
- NDC Code(s): 17238-900-11, 17238-900-30, 17238-900-99
- Packager: HUB Pharmaceuticals, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Product Facts
- INDICATIONS:
-
DIRECTIONS FOR USE:
To insure full fluorescence and patient comfort, the BioGlo impregnated tip should be moistened before application. One or two drops of sterile irrigating or saline solution should be used for this purpose. Touch conjunctiva or fornix as required with moistened tip. It is recommended that the patient blink several times after application.
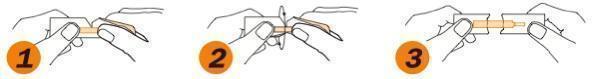
- Grasp tabs between thumbs & index fingers
- Gently pull tabs apart
- Remove strip taking care to touch only the white portion of the strip
- STORAGE:
- NOTE:
- KEEP OUT OF REACH OF CHILDREN
- HOW SUPPLIED:
- SPL UNCLASSIFIED SECTION
- Representative Packaging:
-
INGREDIENTS AND APPEARANCE
BIOGLO
fluorescein sodium stripProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:17238-900 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLUORESCEIN SODIUM (UNII: 93X55PE38X) (FLUORESCEIN - UNII:TPY09G7XIR) FLUORESCEIN SODIUM 1 mg in 1 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17238-900-11 100 in 1 BOX 04/01/2018 1 NDC:17238-900-99 1 mg in 1 PACKET; Type 0: Not a Combination Product 2 NDC:17238-900-30 300 in 1 BOX 04/01/2018 2 NDC:17238-900-99 1 mg in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/01/2012 Labeler - HUB Pharmaceuticals, Inc. (611747945) Establishment Name Address ID/FEI Business Operations OMNI LENS PRIVATE LIMITED 862170057 manufacture(17238-900) Establishment Name Address ID/FEI Business Operations Madhu Instruments Pvt.Ltd. 915827852 manufacture(17238-900)






