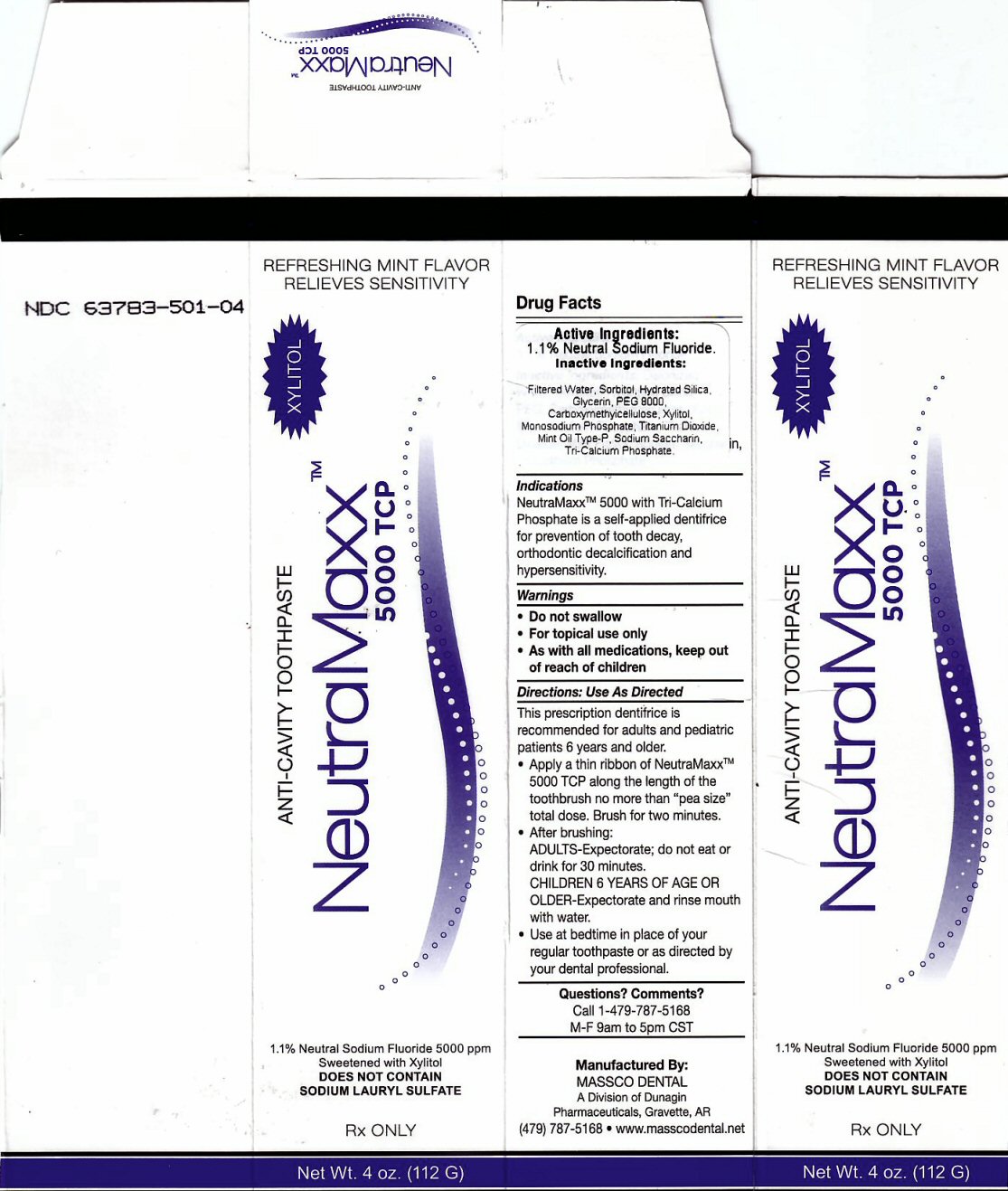
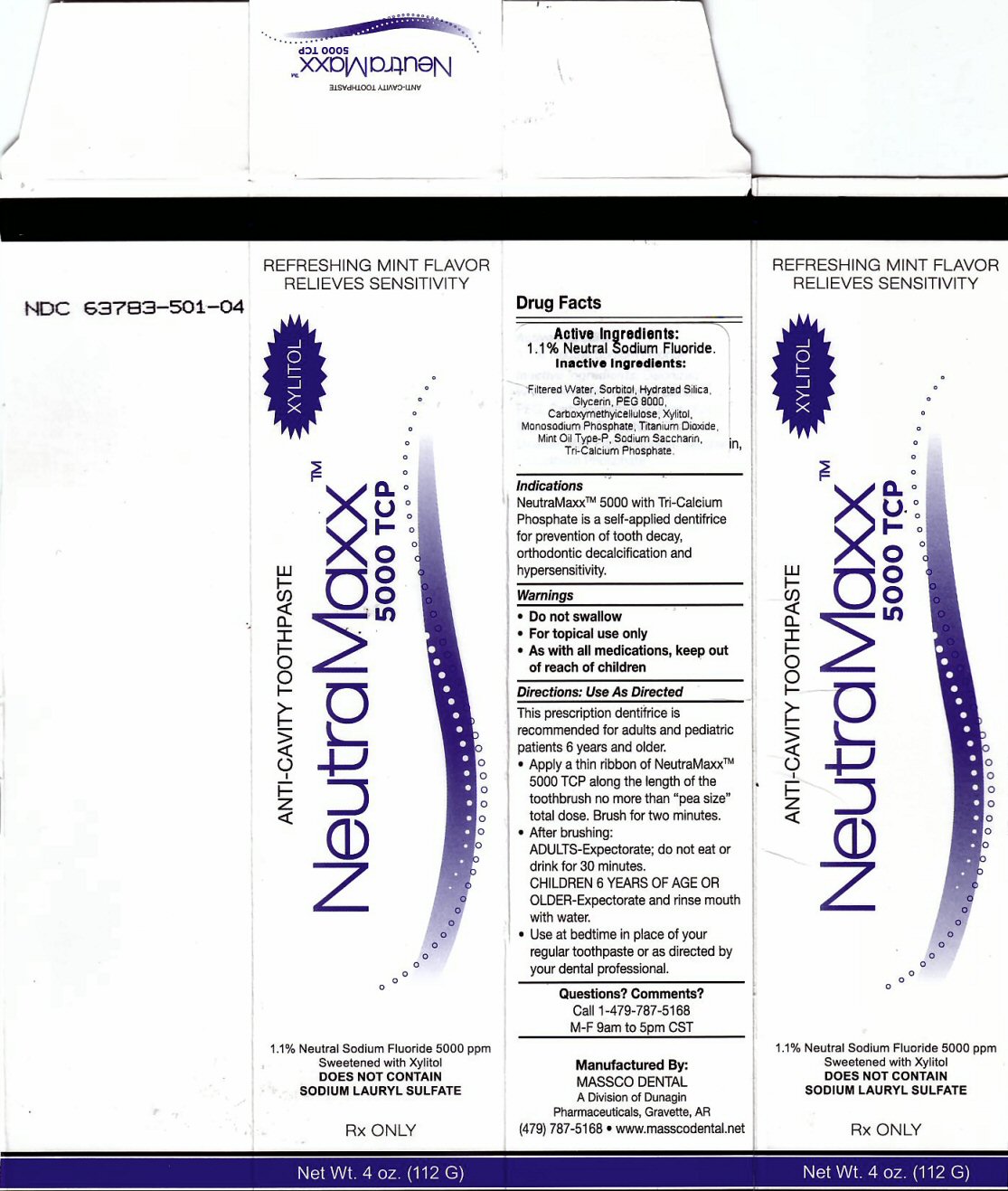
Label: NEUTRAMAXX 5000 TCP- sodium fluoride paste, dentifrice
- NDC Code(s): 63783-501-04
- Packager: Massco Dental A Division of Dunagin Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient Section
- Purpose Section
- Keep Out of Reach of Children Section
- Indications and Usage Section
- Warning Section
-
Directions: Use As Directed Section
This prescription dentifrice is recommended for adults and pediatric patients 6 years and older
Apply a thin ribbon of NeutraMaxxtm 5000 TCP along the length of the toothbrush no more than "pea size" total dose. Brush for two minutes.
After brushing ADULTS - Expectorate, do not eat for 30 minutes. CHILDREN 6 YEARS OF AGE OR OLDER - Expectorate and rinse mouth with water. Use at bedtime in place of your regular toothpaste or as directed by your dental professional.
- Inactive Ingredient Section
- Questions? Comments? Section
-
Package Label
REFRESHING MINT FLAVOR RELIEVES SENSITIVITY
ANTI-CAVITY TOOTHPASTE NeutraMaxx tm 5000 TCP XYLITO1.1% Neutral Sodium Fluoride 5000 ppm Sweetened with Xylitol
DOES NOT CONTAIN SODIUM LAURYL SULFATE RX ONLY Net wt. 4 oz (112 G)
Manufactured By: MASSCO DENTAL A Division of Dunagin Pharmaceuticals, Gravette, AR (479) 787-5168 www.masscodental.net
res
-
INGREDIENTS AND APPEARANCE
NEUTRAMAXX 5000 TCP
sodium fluoride paste, dentifriceProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63783-501 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) SODIUM FLUORIDE 1.1 g in 112 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) HYDRATED SILICA (UNII: Y6O7T4G8P9) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) CARBOXYMETHYLCELLULOSE (UNII: 05JZI7B19X) XYLITOL (UNII: VCQ006KQ1E) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SACCHARIN SODIUM (UNII: SB8ZUX40TY) TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) Product Characteristics Color Score Shape Size Flavor MINT (REFRESHING MINT FLAVOR) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63783-501-04 112 g in 1 TUBE; Type 0: Not a Combination Product 08/01/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/01/2011 Labeler - Massco Dental A Division of Dunagin Pharmaceuticals (008081858) Registrant - Massco Dental A Division of Dunagin Pharmaceuticals (008081858) Establishment Name Address ID/FEI Business Operations Massco Dental A Division of Dunagin Pharmaceuticals 008081858 manufacture(63783-501)