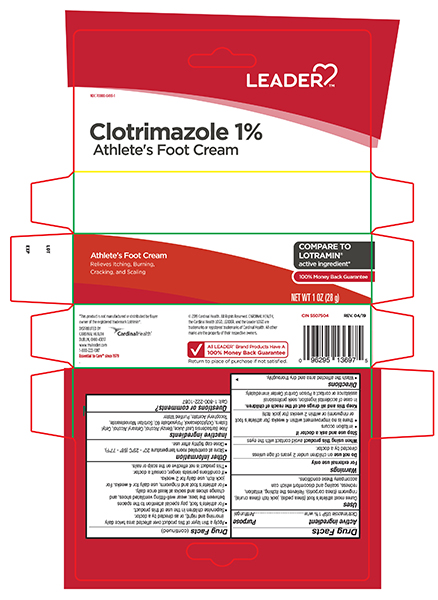
Label: ATHLETES FOOT- clotrimazole cream
- NDC Code(s): 70000-0493-1
- Packager: Cardinal Health 110,Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 18, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
<This product is not effective on the scalp or nails.
- Clean the affected area and dry thoroughly.
- Apply a thin layer of this product over affected area twice daily (morning and night), or as directed by a doctor.
- Supervise children in the use of this product.
- use daily for 2 weeks.
- If conditions persists longer, consult a doctor.>
- Other information
- Inactive ingredients
- Questions?
- Principal Display Panel – 1 oz. Tube
- Principal Display Panel – 0.5 oz. Carton
-
INGREDIENTS AND APPEARANCE
ATHLETES FOOT
clotrimazole creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0493 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOTRIMAZOLE (UNII: G07GZ97H65) (CLOTRIMAZOLE - UNII:G07GZ97H65) CLOTRIMAZOLE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) POLYSORBATE 60 (UNII: CAL22UVI4M) BENZYL ALCOHOL (UNII: LKG8494WBH) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ALOE VERA LEAF (UNII: ZY81Z83H0X) OCTYLDODECANOL (UNII: 461N1O614Y) CETYL PALMITATE (UNII: 5ZA2S6B08X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0493-1 1 in 1 CARTON 05/15/2019 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 05/15/2019 Labeler - Cardinal Health 110,Inc. (063997360)