Label: QUICK RELIEF COLLOIDAL OATMEAL TREATMENT FOR FACE, EYES AND BODY- colloidal oatmeal solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 70381-123-01, 70381-123-02 - Packager: Murad, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
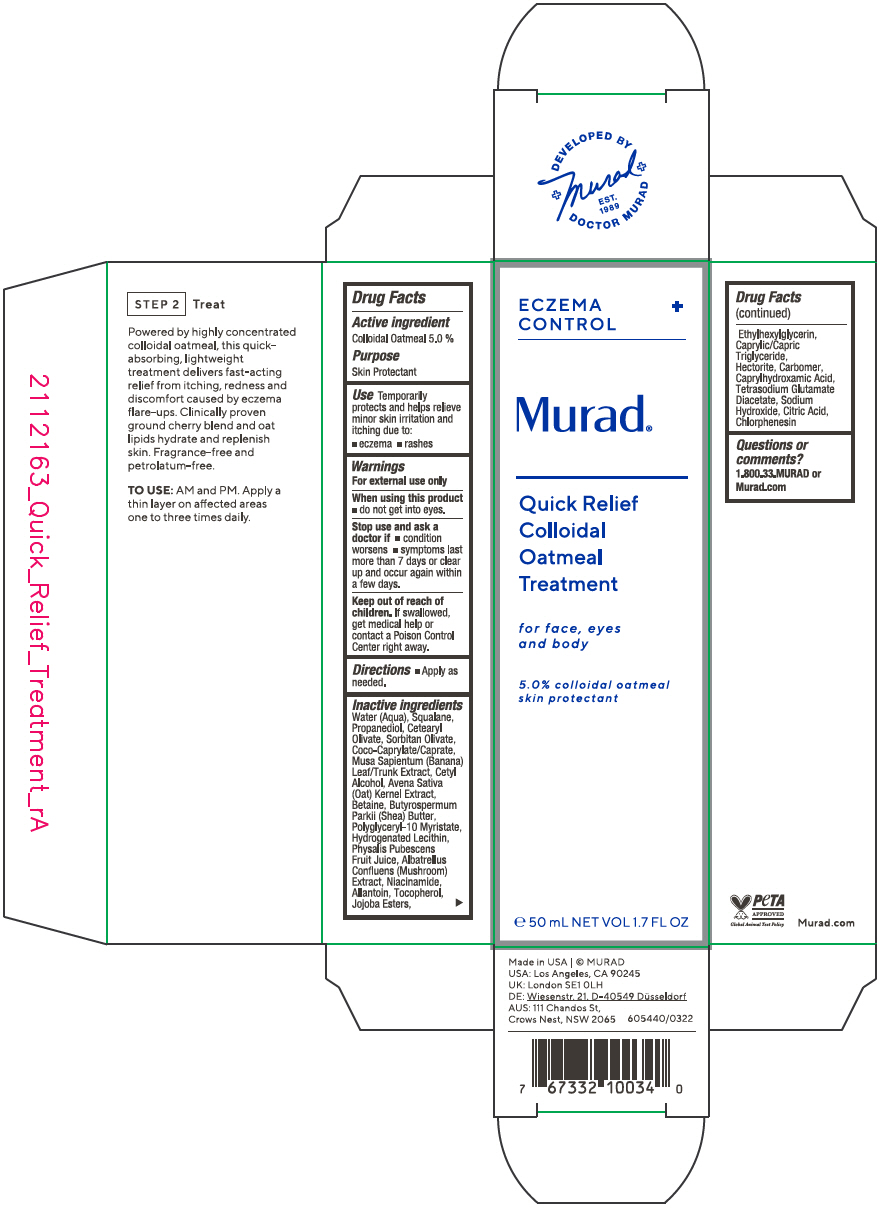
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
-
Inactive ingredients
Water (Aqua), Squalane, Propanediol, Cetearyl Olivate, Sorbitan Olivate, Coco-Caprylate/Caprate, Musa Sapientum (Banana) Leaf/Trunk Extract, Cetyl Alcohol, Avena Sativa (Oat) Kernel Extract, Betaine, Butyrospermum Parkii (Shea) Butter, Polyglyceryl-10 Myristate, Hydrogenated Lecithin, Physalis Pubescens Fruit Juice, Albatrellus Confluens (Mushroom) Extract, Niacinamide, Allantoin, Tocopherol, Jojoba Esters, Ethylhexylglycerin, Caprylic/Capric Triglyceride, Hectorite, Carbomer, Caprylhydroxamic Acid, Tetrasodium Glutamate Diacetate, Sodium Hydroxide, Citric Acid, Chlorphenesin
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 50 mL Tube Carton
-
INGREDIENTS AND APPEARANCE
QUICK RELIEF COLLOIDAL OATMEAL TREATMENT FOR FACE, EYES AND BODY
colloidal oatmeal solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70381-123 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OATMEAL (UNII: 8PI54V663Y) (OATMEAL - UNII:8PI54V663Y) OATMEAL 5 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SQUALANE (UNII: GW89575KF9) PROPANEDIOL (UNII: 5965N8W85T) CETEARYL OLIVATE (UNII: 58B69Q84JO) SORBITAN OLIVATE (UNII: MDL271E3GR) COCOYL CAPRYLOCAPRATE (UNII: 8D9H4QU99H) CETYL ALCOHOL (UNII: 936JST6JCN) OAT (UNII: Z6J799EAJK) BETAINE (UNII: 3SCV180C9W) SHEA BUTTER (UNII: K49155WL9Y) POLYGLYCERYL-10 MYRISTATE (UNII: Y9WL8QN3ZB) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) PHYSALIS PUBESCENS FRUIT JUICE (UNII: GM2TDN9DZI) ALBATRELLUS CONFLUENS FRUITING BODY (UNII: 7UVG8A4DP0) NIACINAMIDE (UNII: 25X51I8RD4) ALLANTOIN (UNII: 344S277G0Z) TOCOPHEROL (UNII: R0ZB2556P8) HYDROGENATED JOJOBA OIL, RANDOMIZED (UNII: Q47ST02F58) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) HECTORITE (UNII: 08X4KI73EZ) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) TETRASODIUM GLUTAMATE DIACETATE (UNII: 5EHL50I4MY) SODIUM HYDROXIDE (UNII: 55X04QC32I) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CHLORPHENESIN (UNII: I670DAL4SZ) MUSA X PARADISIACA LEAF (UNII: 2UIJ27N6ED) Product Characteristics Color WHITE (Light Beige to Beige) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70381-123-02 1 in 1 CARTON 11/02/2022 1 NDC:70381-123-01 50 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part347 11/02/2022 Labeler - Murad, LLC (781254792) Establishment Name Address ID/FEI Business Operations KDC/One Chatsworth, Inc. 118542196 MANUFACTURE(70381-123)