Label: POVIDONE-IODINE SWABSTICKS SINGLES- povidone-iodine swab
POVIDONE-IODINE SWABSTICKS TRIPLES- povidone-iodine swab
POVIDONE-IODINE PREP PADS MEDIUM- povidone-iodine swab
POVIDONE-IODINE PREP PADS MEDIUM BULK- povidone-iodine swab
POVIDONE-IODINE TOPICAL ANTISEPTIC- povidone-iodine solution
PREP SOLUTION TOPICAL ANTISEPTIC- povidone-iodine solution
PREP SOLUTION WITH SPRAY BOTTLE- povidone-iodine solution
-
NDC Code(s):
53329-900-13,
53329-900-55,
53329-904-04,
53329-939-06, view more53329-939-08, 53329-939-13, 53329-939-23, 53329-939-25, 53329-939-44, 53329-939-69, 53329-940-09, 53329-940-97, 53329-941-30, 53329-945-09, 53329-945-29, 53329-946-02, 53329-946-75
- Packager: Medline Industries, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 23, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only.
Do not use
- in the eyes
- longer than 1 week unless directed by a doctor
- on individuals who are allergic or sensitive to iodine
- or apply over large areas of the body
- Directions
- Other information
- Inactive ingredients
- Questions
- Principal Display Panel - Swabstick Singles pouch
- Principal Display Panel - Swabstick Singles box
- Drug Facts Panel - Swabstick Singles box
- Principal Display Panel - Swabstick Triples box
- Drug Facts Panel - Swabstick Triples box
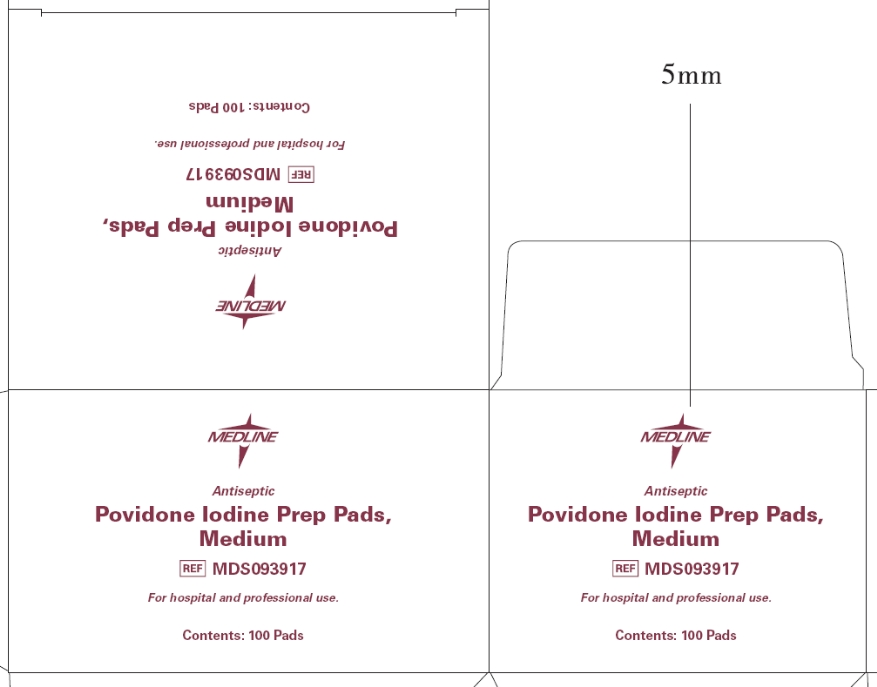
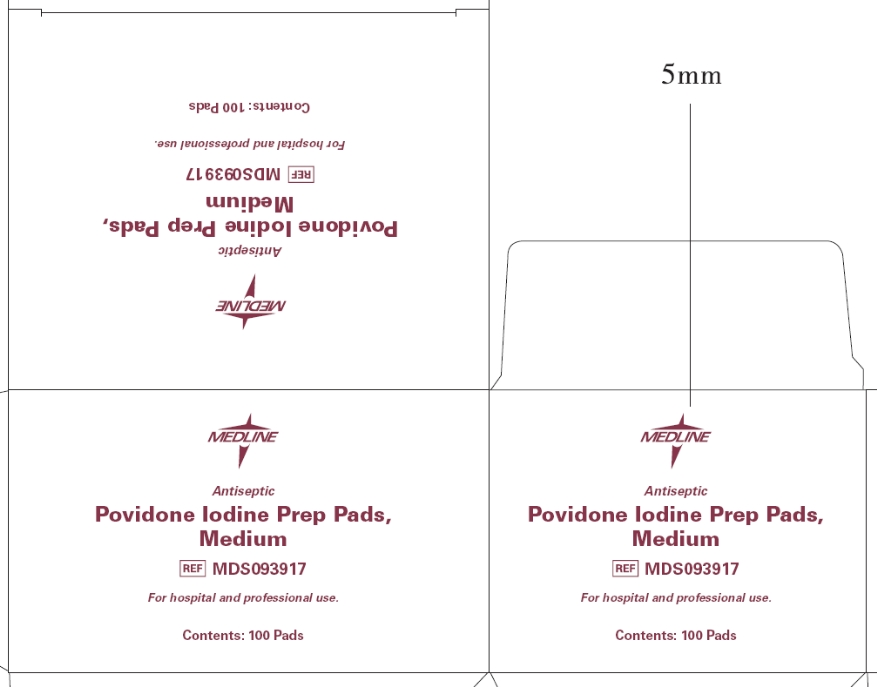
- Principal Display Panel - Prep Pads, Medium box
- Drug Facts Panel - Prep Pads, Medium box
- Principal Display Panel - Prep Pad Bulk case
- Drug Facts Panel - Prep Pad Bulk case
- Principal Display Panel - Prep Solution bottle
- Principal Display Panel - Prep Solution label
- Principal Display Panel - Prep Solution label
-
INGREDIENTS AND APPEARANCE
POVIDONE-IODINE SWABSTICKS SINGLES
povidone-iodine swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-945 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 0.10 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-945-29 50 in 1 BOX 1 NDC:53329-945-09 1 in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 POVIDONE-IODINE SWABSTICKS TRIPLES
povidone-iodine swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-946 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 0.10 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-946-75 25 in 1 BOX 1 NDC:53329-946-02 3 in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 POVIDONE-IODINE PREP PADS MEDIUM
povidone-iodine swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-941 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 0.10 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-941-30 100 in 1 BOX Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 POVIDONE-IODINE PREP PADS MEDIUM BULK
povidone-iodine swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-940 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 0.10 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-940-97 3000 in 1 CASE 1 NDC:53329-940-09 1 in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 POVIDONE-IODINE TOPICAL ANTISEPTIC
povidone-iodine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-939 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength Citric Acid (UNII: 2968PHW8QP) SODIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: 94255I6E2T) NONOXYNOL-9 (UNII: 48Q180SH9T) SODIUM HYDROXIDE (UNII: 55X04QC32I) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-939-44 118 mL in 1 BOTTLE, PLASTIC 2 NDC:53329-939-06 473 mL in 1 BOTTLE, PLASTIC 3 NDC:53329-939-08 237 mL in 1 BOTTLE, PLASTIC 4 NDC:53329-939-23 946 mL in 1 BOTTLE, PLASTIC 5 NDC:53329-939-25 3785 mL in 1 BOTTLE, PLASTIC 6 NDC:53329-939-69 88 mL in 1 BOTTLE, PLASTIC 7 NDC:53329-939-13 59 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 PREP SOLUTION TOPICAL ANTISEPTIC
povidone-iodine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-900 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) Polysorbate 80 (UNII: 6OZP39ZG8H) Citric Acid (UNII: 2968PHW8QP) Sodium Citrate (UNII: 1Q73Q2JULR) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-900-13 59 mL in 1 BOTTLE, PLASTIC 2 NDC:53329-900-55 59 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 08/31/2014 PREP SOLUTION WITH SPRAY BOTTLE
povidone-iodine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-904 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Povidone-Iodine (UNII: 85H0HZU99M) (Iodine - UNII:9679TC07X4) Povidone-Iodine 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) Polysorbate 80 (UNII: 6OZP39ZG8H) Citric Acid (UNII: 2968PHW8QP) Sodium Citrate (UNII: 1Q73Q2JULR) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-904-04 118 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/01/2007 08/31/2014 Labeler - Medline Industries, Inc. (025460908)