Label: ARNICA VALUE PACK- arnica montana kit
- NDC Code(s): 0220-9047-78
- Packager: Laboratoires Boiron
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 18, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
INACTIVE INGREDIENT
Arnicare Cream Value Pack:
Cream: alcohol, caprylyl, glycol, carbomer, cetyl palmitate, EDTA disodium, glycerin, lauroyl macrogolglycerides, pegoxol-7 stearate, purified water, sodium hydroxide, sorbic acid, 1,2-hexanediol.
Pellets: Lactose, Sucrose.
Arnicare Cream Value Pack (without disodium EDTA):
Cream: alcohol, caprylyl, glycol, carbomer, cetyl palmitate, glycerin, lauroyl macrogolglycerides, pegoxol-7 stearate, purified water, sodium hydroxide, sorbic acid, 1,2-hexanediol.
Pellets: Lactose, Sucrose.
- HOW SUPPLIED
- PURPOSE
- STORAGE AND HANDLING
- QUESTIONS
- ASK DOCTOR
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- WHEN USING
- DRUG INTERACTIONS
- WARNINGS
-
DOSAGE & ADMINISTRATION
Cream:
Apply a thin layer of Arnicare Cream of affected area and massage gently as soon as possible after minor injury. Repeat 3 times a day or as needed. If heat or ice is applied, wait 5 minutes before applying Arnicare Cream.
Pellets:
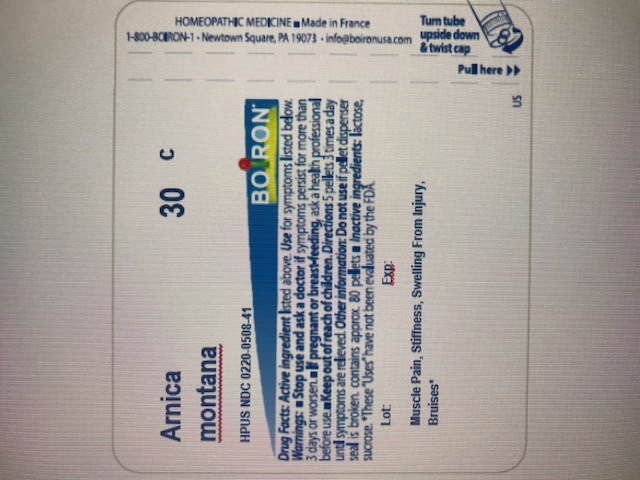
Adults and children 2 years of age and older: Dissolve 5 pellets in the mouth 3 times a day until relieved or as directed by a doctor.How to use Arnica 30C Pellets: Turn tube upside down. Twist until 5 pellets are dispensed
- PREGNANCY OR BREAST FEEDING
-
SPL UNCLASSIFIED SECTION
ɨ Homeopathic mother tincture made from Arnica montana fresh whole plant.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
**C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
For general pain relief, Arnica 30C pellets can be take alone or as a complement to Arnicare Cream.
Non-drowsy
No known drug interactions
Local & General Pain
Combine for total relief
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ARNICA VALUE PACK
arnica montana kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0220-9047 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0220-9047-78 1 in 1 BOX 11/01/2008 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 80 Part 2 1 TUBE 70 g Part 1 of 2 ARNICA
arnica montana pelletProduct Information Item Code (Source) NDC:0220-0508 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 30 [hp_C] Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) LACTOSE (UNII: J2B2A4N98G) Product Characteristics Color white Score no score Shape ROUND (Pellet) Size 4mm Flavor Imprint Code ; Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 80 in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/01/2008 Part 2 of 2 ARNICARE
arnica montana creamProduct Information Item Code (Source) NDC:0220-9021 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 1 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARBOMER 934 (UNII: Z135WT9208) CETYL PALMITATE (UNII: 5ZA2S6B08X) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBIC ACID (UNII: X045WJ989B) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) GLYCERIN (UNII: PDC6A3C0OX) PEGOXOL 7 STEARATE (UNII: 3EW5AXE5X5) ALCOHOL (UNII: 3K9958V90M) HYDROGENATED PALM/PALM KERNEL OIL PEG-6 ESTERS (UNII: 8EPU9MJ01K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 70 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/01/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/01/2008 Labeler - Laboratoires Boiron (282560473) Registrant - Boiron Inc. (014892269) Establishment Name Address ID/FEI Business Operations Boiron 282560473 manufacture(0220-9047)