Label: ACETAMINOPHEN, ASPIRIN AND CAFFEINE tablet
- NDC Code(s): 62207-680-47, 62207-680-51, 62207-680-98, 62207-680-99
- Packager: Granules India Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
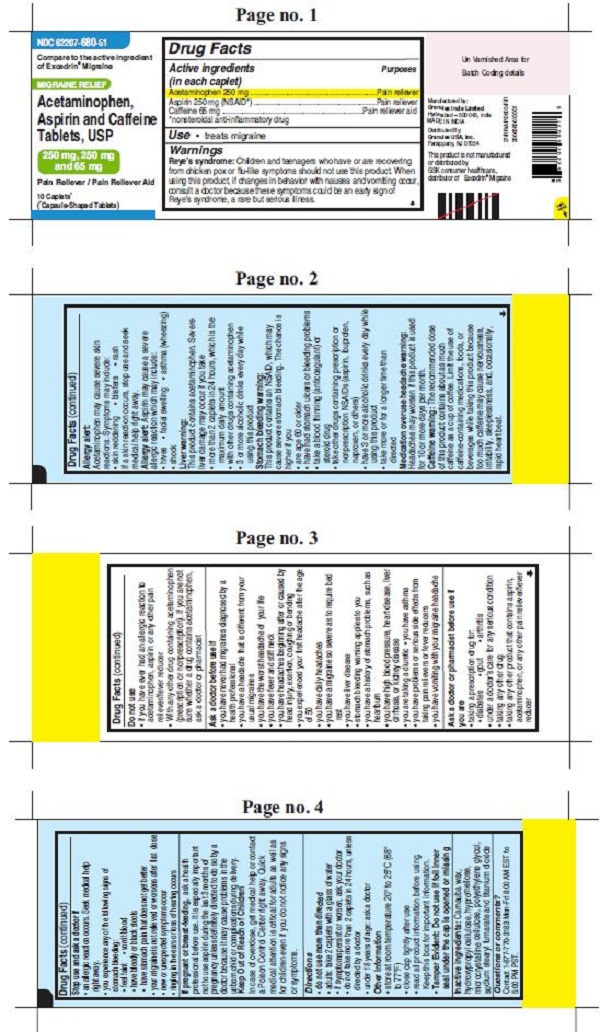
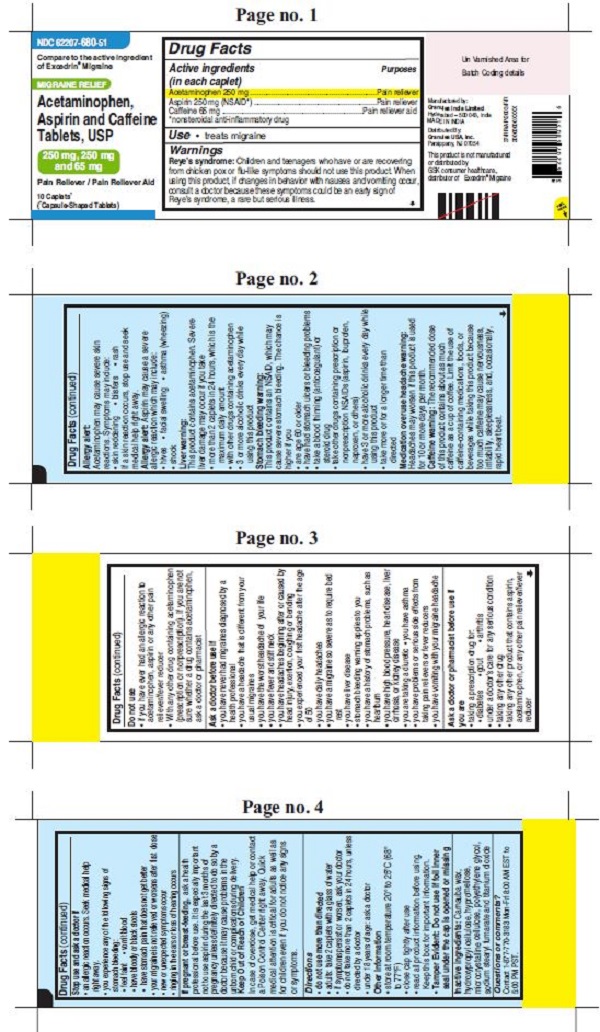
- ACTIVE INGREDIENT(S)
- PURPOSE
- USE(S)
-
WARNINGS
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
• skin reddening
• blisters
• rash
If a skin reaction occurs, stop use and seek medical help right away.Allergy alert: Aspirin may cause a severe allergic reaction which may include:
• hives
• facial swelling
• asthma (wheezing)
• shockLiver warning: This product contains acetaminophen. Severe liver damage may occur if you take
• more than 2 caplets in 24 hours, which is the maximum daily amount
• with other drugs containing acetaminophen
• 3 or more alcoholic drinks every day while using this productStomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
• are age 60 or older
• have had stomach ulcers or bleeding problems
• take a blood thinning (anticoagulant) or steroid drug
• take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
• have 3 or more alcoholic drinks every day while using this product
• take more or for a longer time than directedMedication overuse headache warning: Headaches may worsen if this product is used for 10 or more days per month
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heart beat.
- DO NOT USE
-
ASK A DOCTOR BEFORE USE IF
• you have never had migraines diagnosed by a health professional
• you have a headache that is different from your usual migraines
• you have the worst headache of your life
• you have fever and stiff neck
• you have headaches beginning after or caused by head injury, exertion, coughing or bending
• you experienced your first headache after the age of 50
• you have daily headaches
• you have a migraine so severe as to require bed rest
• you have liver disease
• stomach bleeding warning applies to you
• you have a history of stomach problems, such as heartburn
• you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
• you are taking a diuretic
• you have asthma
• you have problems or serious side effects from taking pain relievers or fever reducers
• you have vomiting with your migraine headache - ASK A DOCTOR OR PHARMACIST BEFORE USE IF
-
STOP USE AND ASK DOCTOR IF
• an allergic reaction occurs. Seek medical help right away.
• you experience any of the following signs of stomach bleeding :
o feel faint
o vomit blood
o have bloody or black stools
o have stomach pain that does not get better
• your migraine is not relieved or worsens after first dose
• new or unexpected symptoms occur
• ringing in the ears or loss of hearing occurs - IF PREGNANT OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN, ASPIRIN AND CAFFEINE
acetaminophen, aspirin and caffeine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62207-680 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 250 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 250 mg CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 65 mg Inactive Ingredients Ingredient Name Strength SODIUM STEARYL FUMARATE (UNII: 7CV7WJK4UI) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARNAUBA WAX (UNII: R12CBM0EIZ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color white Score no score Shape CAPSULE (Capsule shaped Tablet) Size 17mm Flavor Imprint Code I1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62207-680-51 1 in 1 CARTON 04/15/2021 1 10 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:62207-680-47 500 in 1 BOTTLE; Type 0: Not a Combination Product 04/15/2021 3 NDC:62207-680-98 22500 in 1 BOX; Type 0: Not a Combination Product 04/15/2021 4 NDC:62207-680-99 4500 in 1 POUCH; Type 0: Not a Combination Product 04/15/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA214039 04/15/2021 Labeler - Granules India Ltd (915000087) Establishment Name Address ID/FEI Business Operations Granules India Ltd 918609236 analysis(62207-680) , label(62207-680) , manufacture(62207-680) , pack(62207-680)