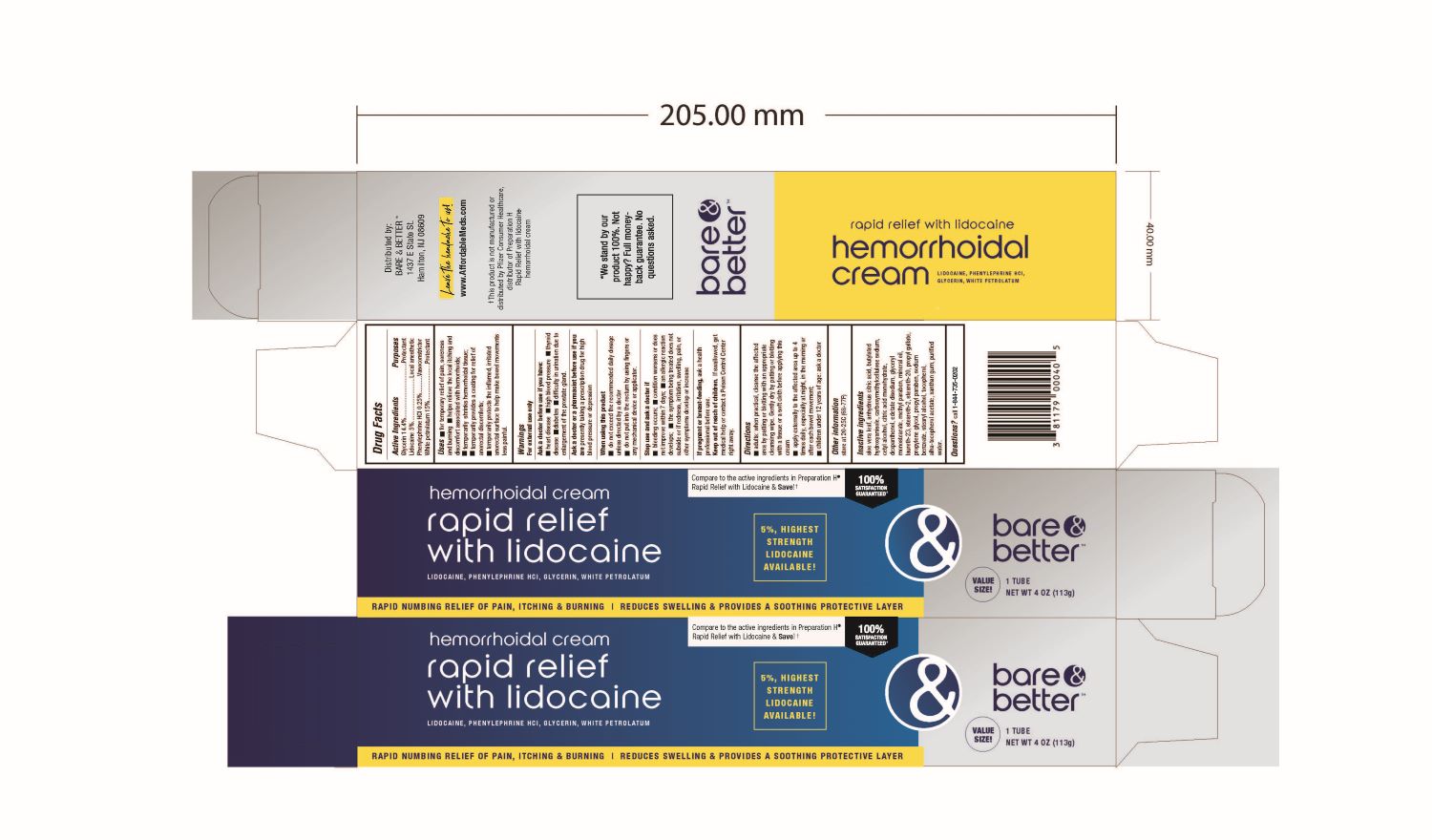
Label: GLYCERIN, LIDOCAINE, PETROLATUM, AND PHENYLEPHRINE HYDROCHLORIDE cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 55629-051-04 - Packager: ONE2ZEE LIMITED LIABILITY COMPANY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 8, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS:
- Active Ingredient:
- Purpose
-
Uses:
- For temporary relief of pain, soreness and burning
- Helps relieve the local itching and discomfort associated with hemorrhoids
- Temporarily shrinks hemorrhoidal tissue
- Temporarily provides a coating for relief of anorectal discomforts
- Temporarily protects the inflamed irritated anorectal surface to help make bowel movements less painful
- Warnings:
-
Directions:
- Adults: when practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with a tissue or a soft cloth before applying this cream.
- Apply externally to the affected area up to 4 times daily, especially at night, in the morning or after each bowel movement
- Children under 12 years of age: ask a doctor
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
-
Inactive Ingredients:
aloe vera leaf, anhydrous citric acid, butylated hydroxyanisole, carboxymethylcellulose sodium, cetyl alcohol, citric acid monohydrate, dexpanthenol, edetate disodium, glyceryl monosterate, methylparaben, mineral oil, laureth-23, steareth-2, steareth-20, propyl gallate, propylene glycol, propylparaben, sodium benzoate, tocopherol, alpha.- tocopherol acetate, xanthan gum, water
- Other Information:
- Package Label
-
INGREDIENTS AND APPEARANCE
GLYCERIN, LIDOCAINE, PETROLATUM, AND PHENYLEPHRINE HYDROCHLORIDE
glycerin, lidocaine, petrolatum, and phenylephrine hydrochloride creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55629-051 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 144 mg in 1 g LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 50 mg in 1 g PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 150 mg in 1 g PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 2.5 mg in 1 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) CETYL ALCOHOL (UNII: 936JST6JCN) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) DEXPANTHENOL (UNII: 1O6C93RI7Z) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) MINERAL OIL (UNII: T5L8T28FGP) LAURETH-23 (UNII: N72LMW566G) STEARETH-2 (UNII: V56DFE46J5) STEARETH-20 (UNII: L0Q8IK9E08) PROPYL GALLATE (UNII: 8D4SNN7V92) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) SODIUM BENZOATE (UNII: OJ245FE5EU) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) TOCOPHEROL (UNII: R0ZB2556P8) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) XANTHAN GUM (UNII: TTV12P4NEE) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55629-051-04 1 in 1 CARTON 02/08/2021 1 113 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part346 02/08/2021 Labeler - ONE2ZEE LIMITED LIABILITY COMPANY (078656111) Registrant - ONE2ZEE LIMITED LIABILITY COMPANY (078656111) Establishment Name Address ID/FEI Business Operations YASH PHARMACEUTICALS 871409551 manufacture(55629-051) , analysis(55629-051)