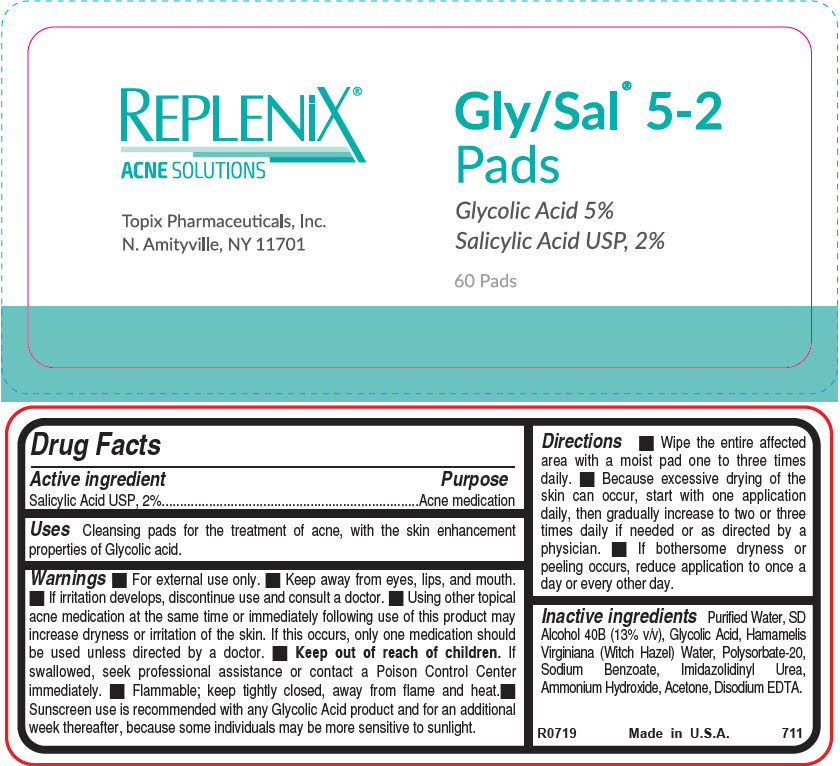
Label: GLY-SAL 5-2 PADS- salicylic acid cloth
- NDC Code(s): 51326-038-60
- Packager: Topiderm, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 22, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
- SPL UNCLASSIFIED SECTION
-
Directions
- Wipe the entire affected area with a moist pad one to three times daily.
- Because excessive drying of the skin can occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a physician.
- If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL - 60 Pad Jar Label
-
INGREDIENTS AND APPEARANCE
GLY-SAL 5-2 PADS
salicylic acid clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51326-038 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.02 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) GLYCOLIC ACID (UNII: 0WT12SX38S) HAMAMELIS VIRGINIANA TOP WATER (UNII: NT00Y05A2V) POLYSORBATE 20 (UNII: 7T1F30V5YH) SODIUM BENZOATE (UNII: OJ245FE5EU) IMIDUREA (UNII: M629807ATL) AMMONIA (UNII: 5138Q19F1X) ACETONE (UNII: 1364PS73AF) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51326-038-60 60 in 1 JAR; Type 0: Not a Combination Product 02/27/1990 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M006 02/27/1990 Labeler - Topiderm, Inc. (049121643) Registrant - Topiderm, Inc. (049121643) Establishment Name Address ID/FEI Business Operations Topiderm, Inc. 049121643 MANUFACTURE(51326-038) Establishment Name Address ID/FEI Business Operations Topix Pharmaceuticals, Inc. 117745066 PACK(51326-038)