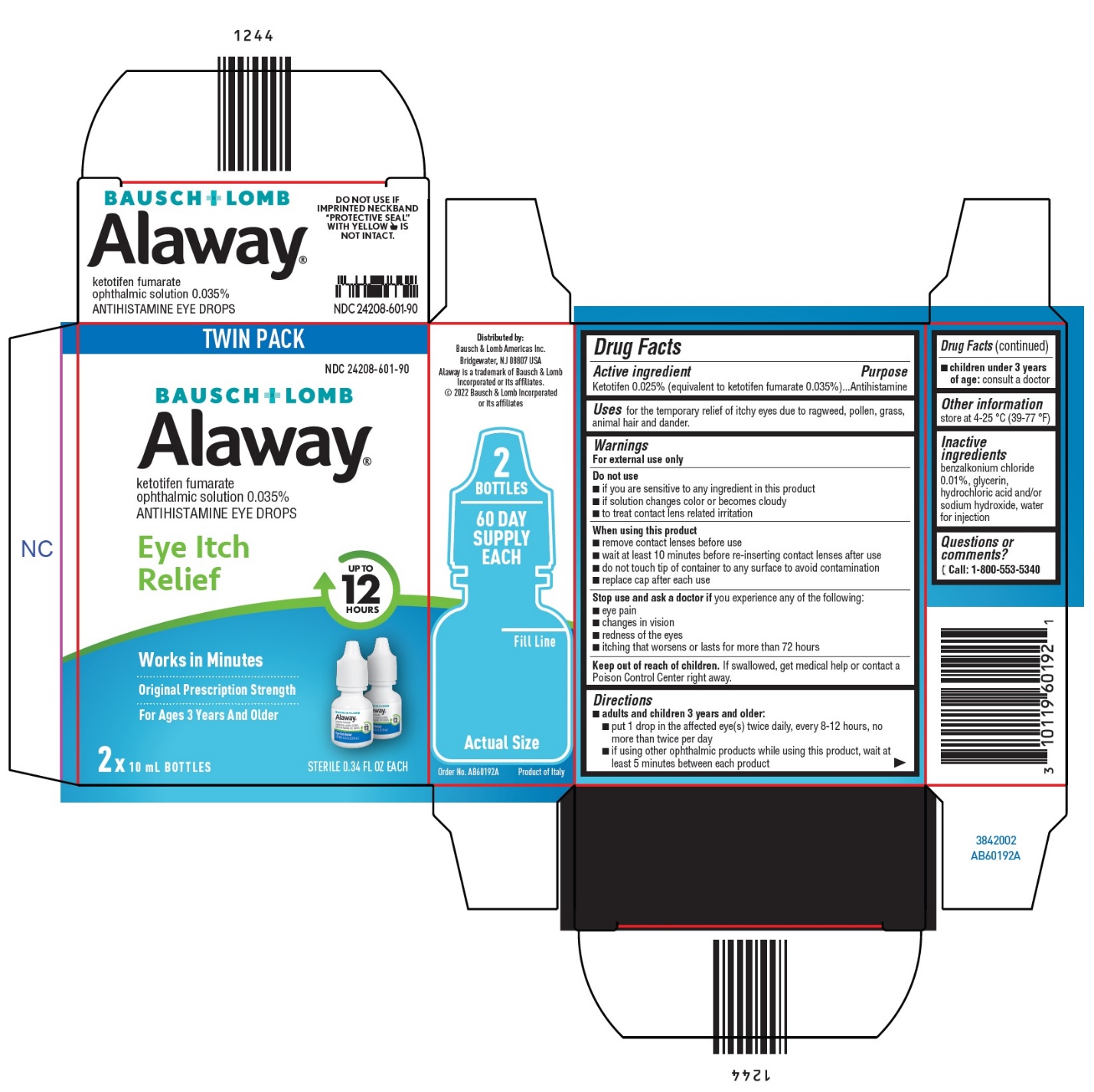
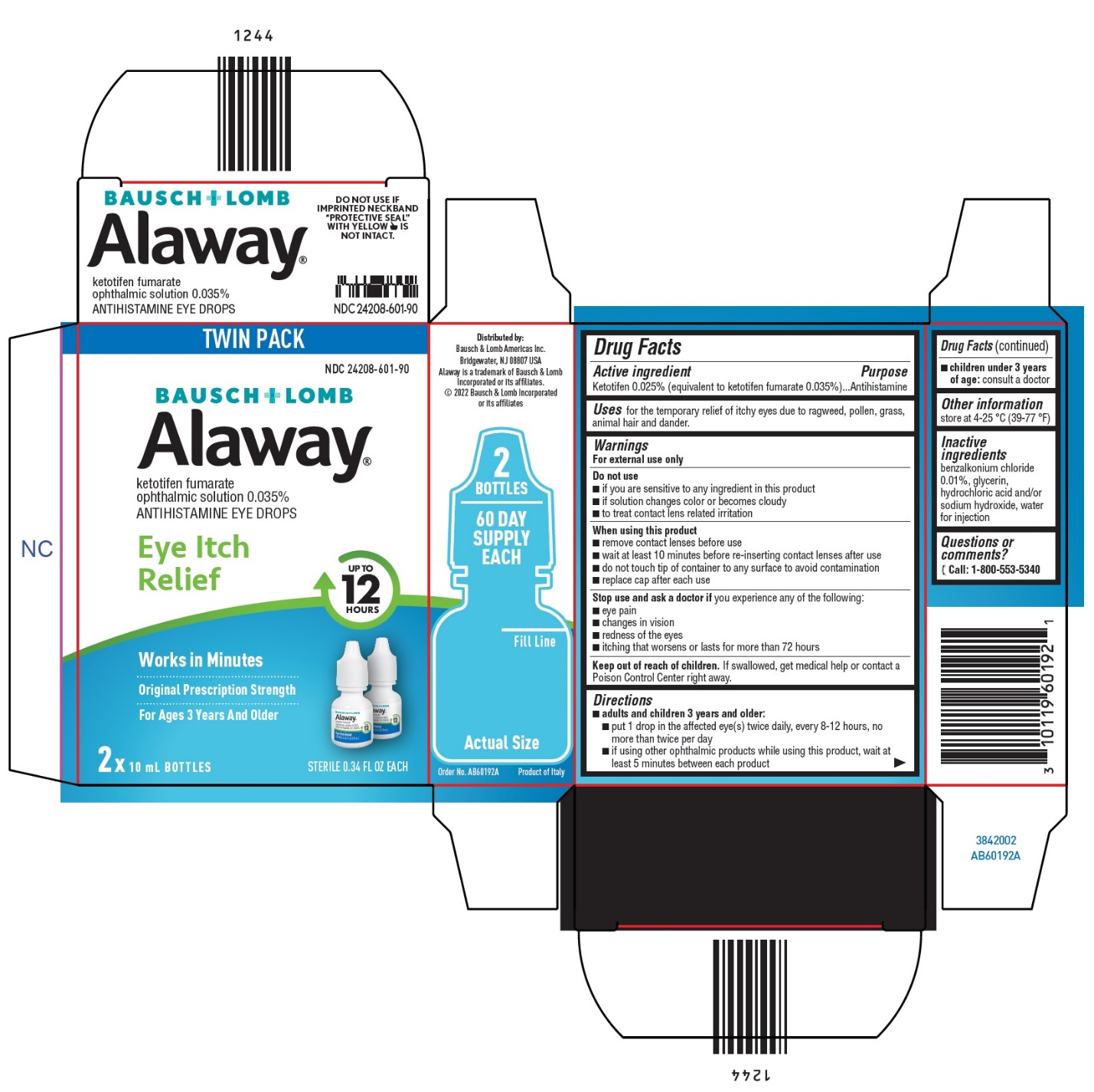
Label: ALAWAY- ketotifen fumarate solution/ drops
- NDC Code(s): 24208-601-05, 24208-601-10, 24208-601-90, 24208-601-95
- Packager: Bausch & Lomb Incorporated
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 1, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- •
- if you are sensitive to any ingredient in this product
- •
- if solution changes color or becomes cloudy
- •
- to treat contact lens related irritation
When using this product
- •
- remove contact lenses before use
- •
- wait at least 10 minutes before re-inserting contact lenses after use
- •
- do not touch tip of container to any surface to avoid contamination
- •
- replace cap after each use
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ALAWAY
ketotifen fumarate solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:24208-601 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength KETOTIFEN FUMARATE (UNII: HBD503WORO) (KETOTIFEN - UNII:X49220T18G) KETOTIFEN 0.25 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) GLYCERIN (UNII: PDC6A3C0OX) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24208-601-10 1 in 1 CARTON 12/01/2006 1 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:24208-601-95 1 in 1 CARTON 12/01/2006 09/30/2015 2 1 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC:24208-601-05 1 in 1 CARTON 12/01/2006 3 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 4 NDC:24208-601-90 2 in 1 CARTON 12/01/2006 4 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021996 12/01/2006 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated 079587625 MANUFACTURE(24208-601)