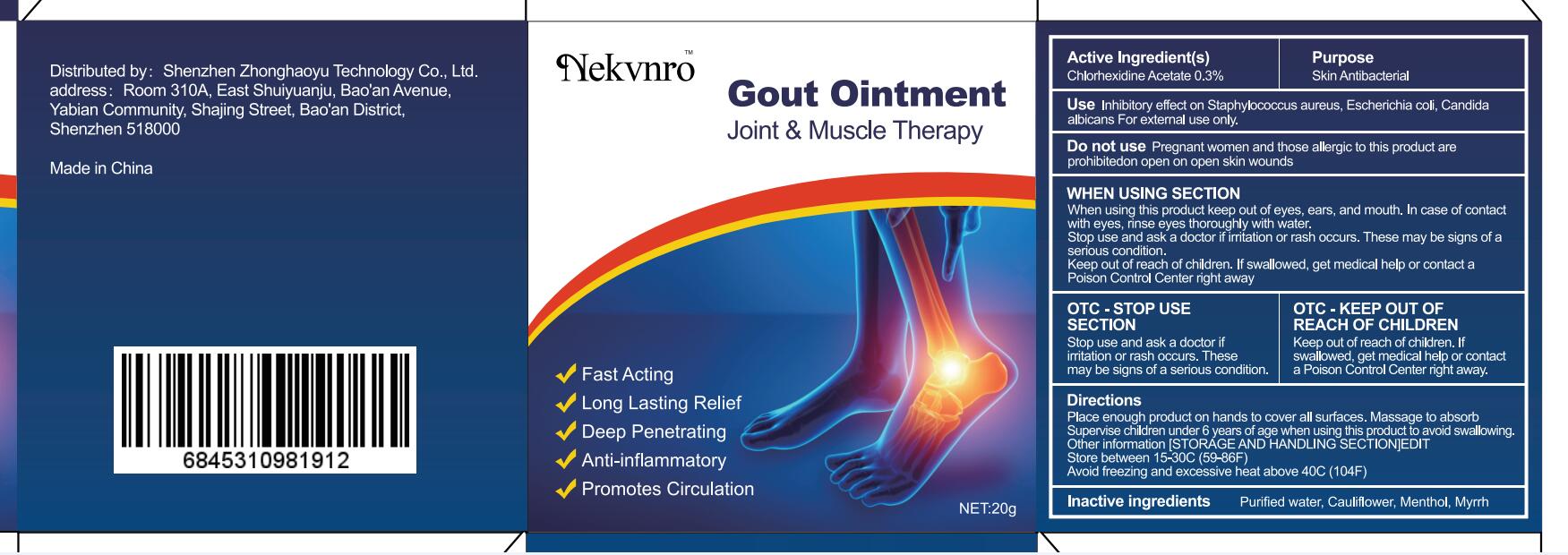
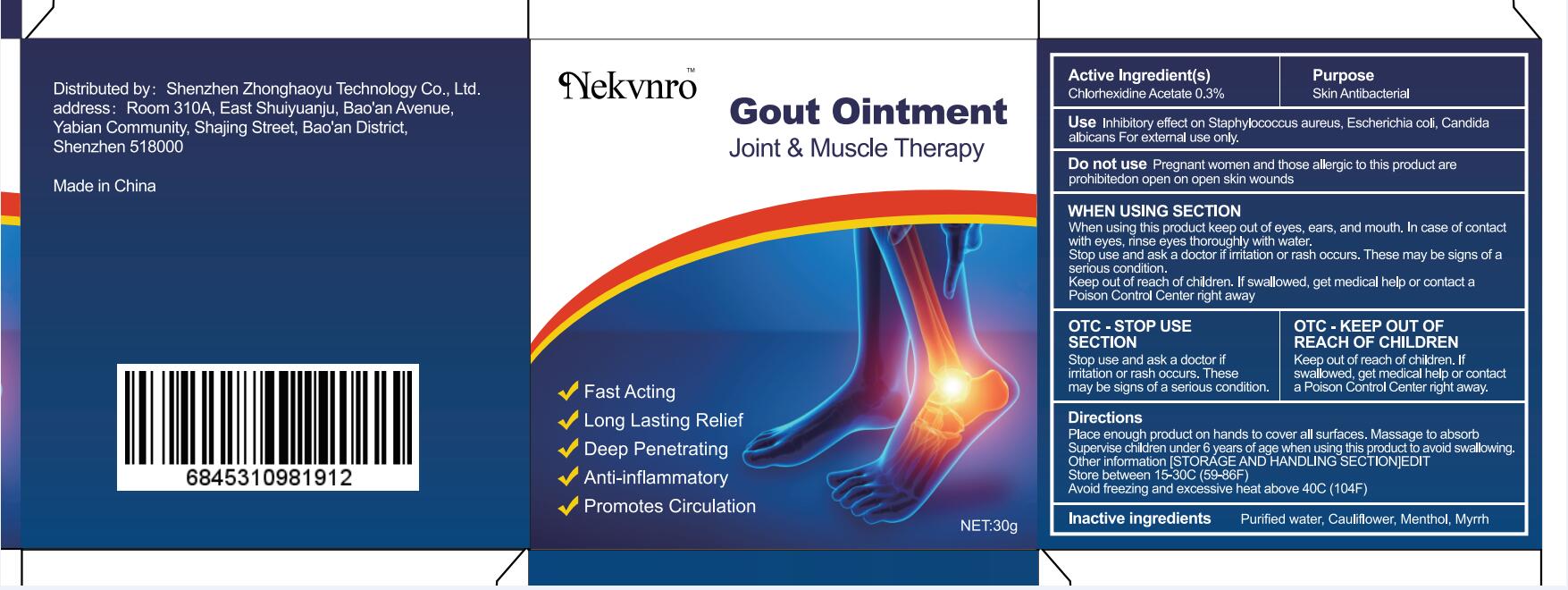
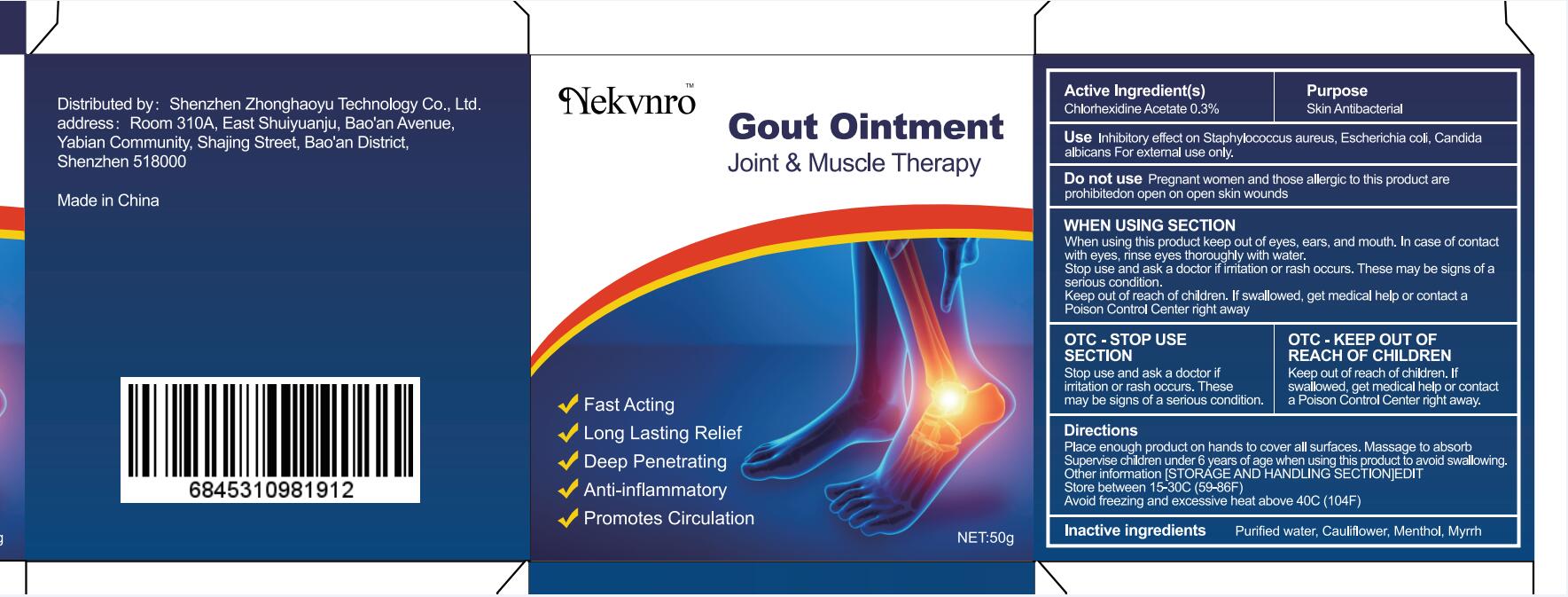
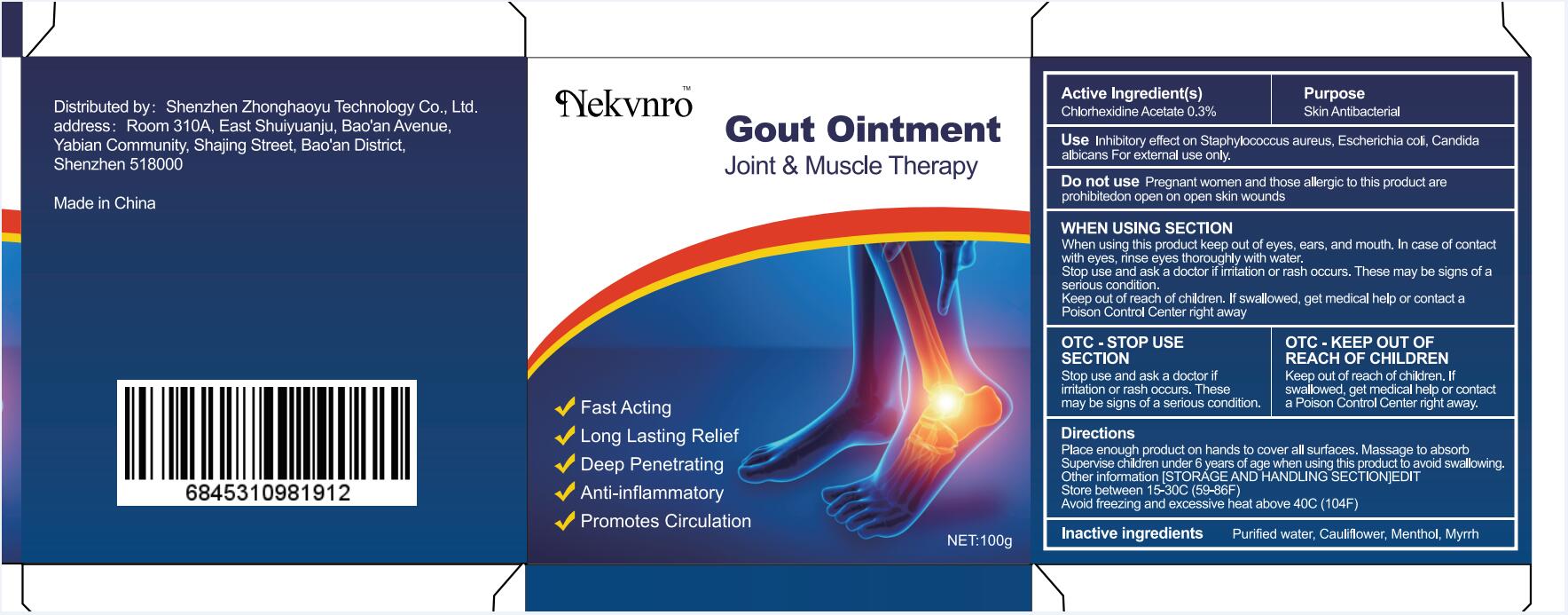
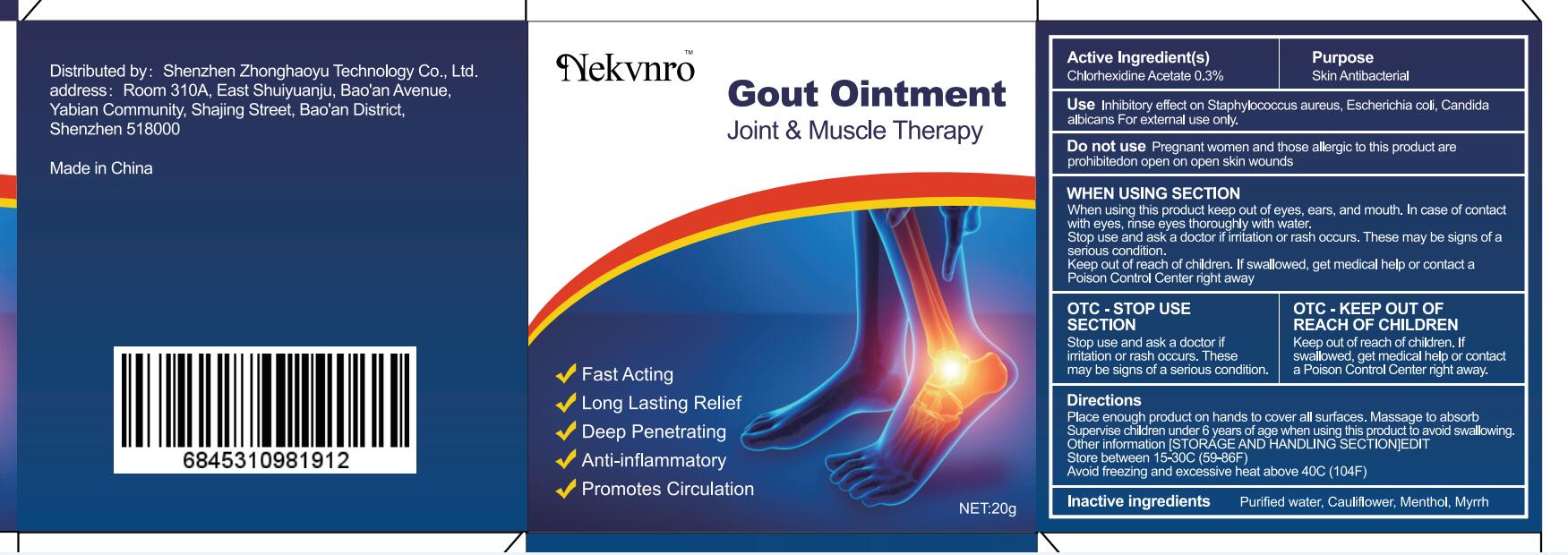
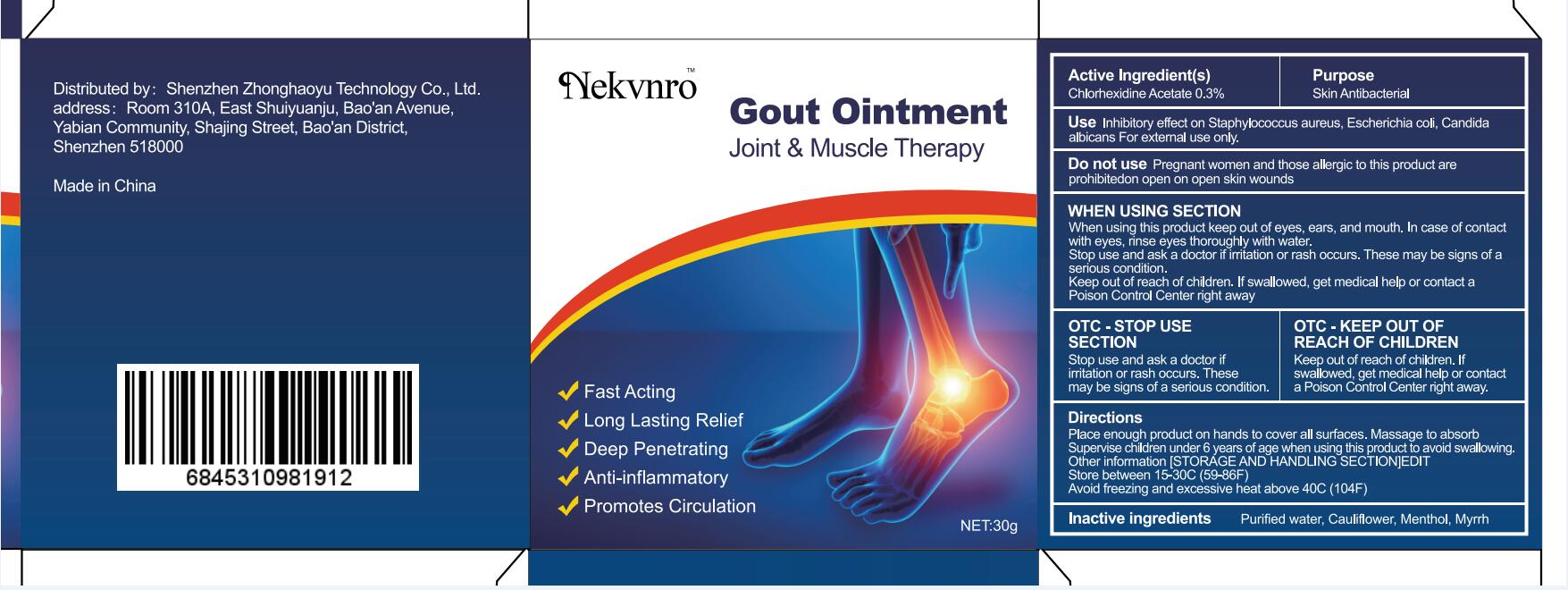
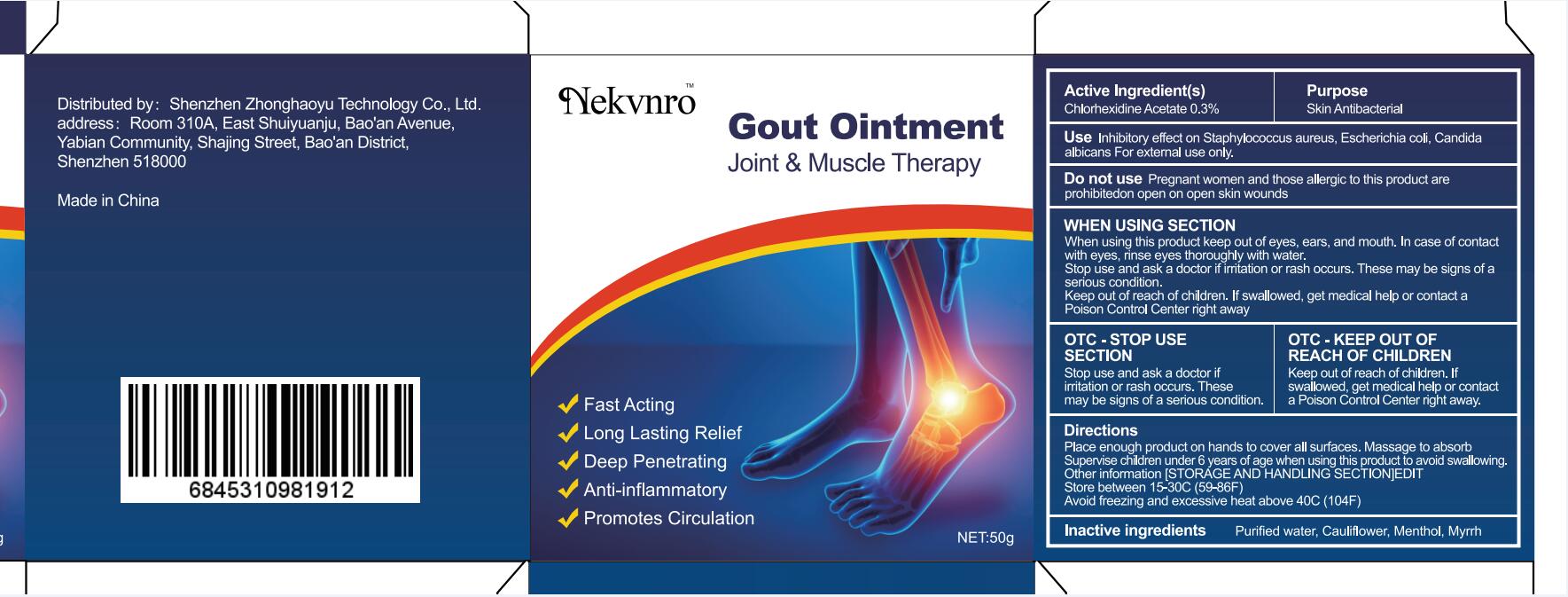
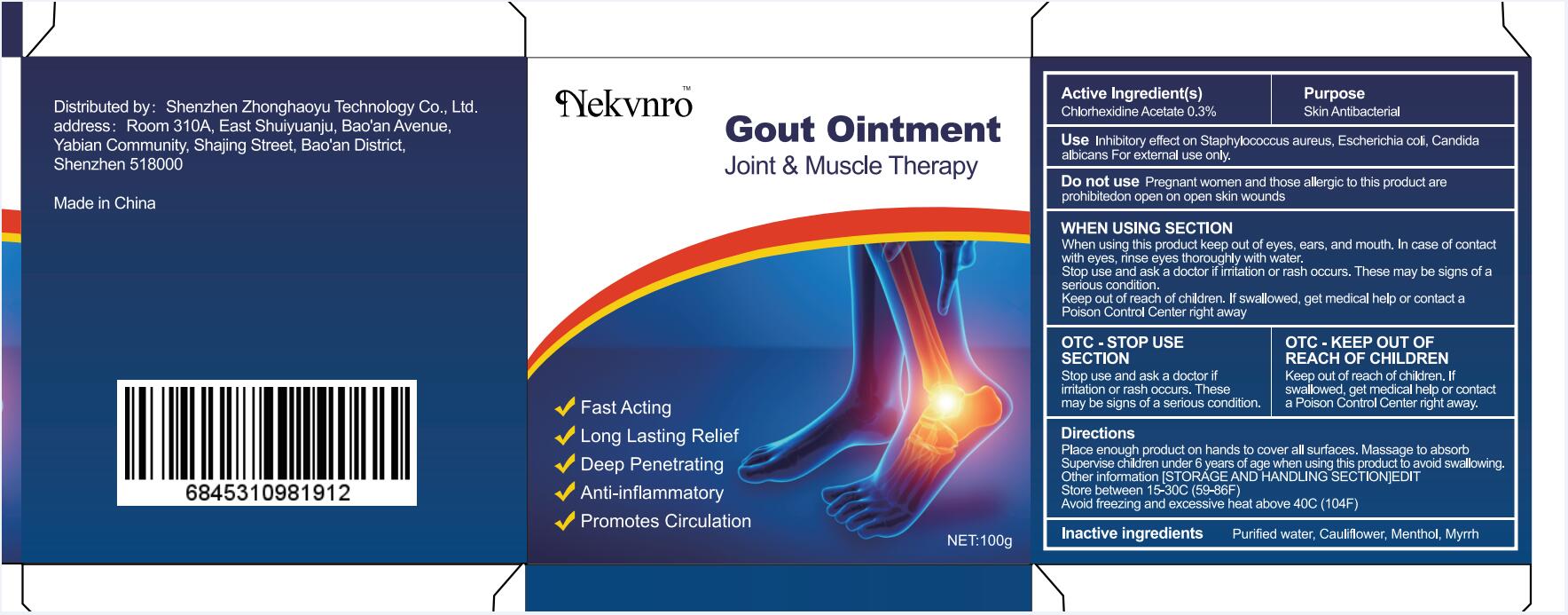
Label: GOUT- gout ointment ointment
-
Contains inactivated NDC Code(s)
NDC Code(s): 82877-001-01, 82877-001-02, 82877-001-03, 82877-001-04 - Packager: Shenzhen Boceda New Technology Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated July 6, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
- Do not use
-
WHEN USING SECTION
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
- OTC - STOP USE SECTION
- OTC - KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Warnings
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
GOUT
gout ointment ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82877-001 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE ACETATE (UNII: 5908ZUF22Y) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE ACETATE 0.03 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CAULIFLOWER (UNII: 138LUT2DWV) MENTHOL (UNII: L7T10EIP3A) MYRRH (UNII: JC71GJ1F3L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82877-001-01 20 g in 1 BOX; Type 0: Not a Combination Product 07/07/2022 2 NDC:82877-001-02 30 g in 1 BOX; Type 0: Not a Combination Product 07/07/2022 3 NDC:82877-001-03 50 g in 1 BOX; Type 0: Not a Combination Product 07/07/2022 4 NDC:82877-001-04 100 g in 1 BOX; Type 0: Not a Combination Product 07/07/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 07/07/2022 Labeler - Shenzhen Boceda New Technology Co., Ltd. (616634883) Establishment Name Address ID/FEI Business Operations Shenzhen Boceda New Technology Co., Ltd. 616634883 manufacture(82877-001)