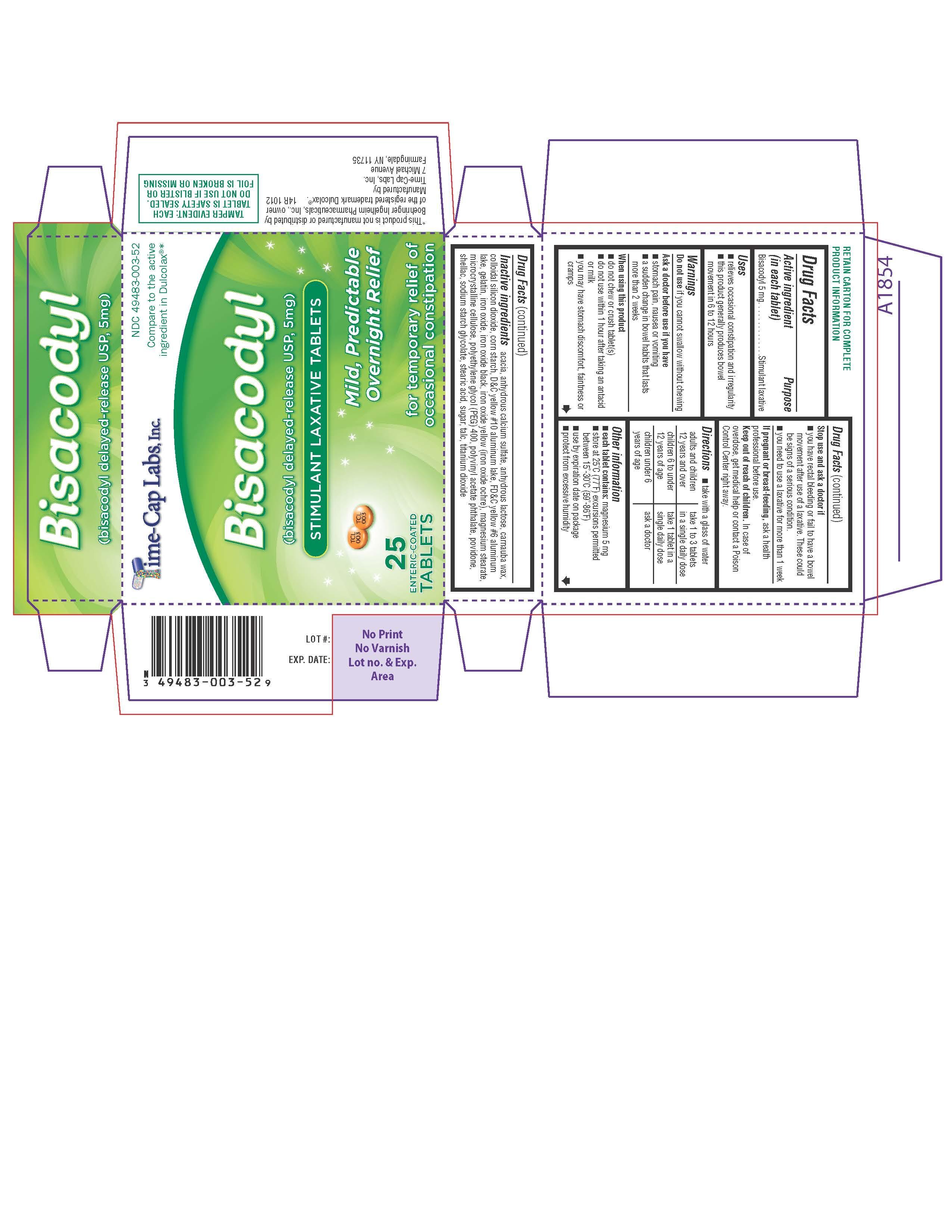
Label: STIMULANT LAXATIVE- bisacodyl tablet, delayed release
- NDC Code(s): 49483-003-00, 49483-003-01, 49483-003-10, 49483-003-52
- Packager: TIME CAP LABORATORIES, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 27, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Acadia, Anhydrous Calcium Sulfate, Anhydrous Lactose, Carnauba Wax, Colloidal Silicon Dioxide, Corn Starch, D&C Yellow #10 Aluminum Lake, FD&C yellow #6 aluminum lake, Gelatin, Iron Oxide, Iron Oxide Black, Iron Oxide Yellow(Iron Oxide Ochre), Magnesium Stearate, Microcrystalline Cellulose, Polyethylene Glycol (PEG) 400, Polyvinyl Acetate Phthalates, Povidone, Shellac, Sodium Starch Glycolate, Stearic Acid, Sugar, Talc, Titanium Dioxide
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
Warning: Do not use if you cannot swallow without chewing.
Ask a doctor before use if you have: stomach pain, nausea or vomiting; A sudden change in bowel habits that lasts more than two weeks.
When using this product: Do not chew or crush tablet(s); Do not use within 1 hour after taking an antacid or milk; Do not use this product if you have stomach discomfort, faintness or cramps.
- DOSAGE & ADMINISTRATION
- 25 COUNT BLISTER
- 100 count label 14R
- 1000 CT 14R
-
INGREDIENTS AND APPEARANCE
STIMULANT LAXATIVE
bisacodyl tablet, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49483-003 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BISACODYL (UNII: 10X0709Y6I) (DEACETYLBISACODYL - UNII:R09078E41Y) BISACODYL 5 mg Inactive Ingredients Ingredient Name Strength ACACIA (UNII: 5C5403N26O) CALCIUM SULFATE ANHYDROUS (UNII: E934B3V59H) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYVINYL ACETATE PHTHALATE (UNII: 58QVG85GW3) POVIDONE (UNII: FZ989GH94E) SHELLAC (UNII: 46N107B71O) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) STEARIC ACID (UNII: 4ELV7Z65AP) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange Score no score Shape ROUND Size 5mm Flavor Imprint Code TCL003 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49483-003-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 12/07/2018 2 NDC:49483-003-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 12/07/2018 3 NDC:49483-003-52 25 in 1 BLISTER PACK; Type 0: Not a Combination Product 12/07/2018 4 NDC:49483-003-00 100000 in 1 CARTON; Type 0: Not a Combination Product 12/07/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 04/01/2014 Labeler - TIME CAP LABORATORIES, INC. (037052099) Establishment Name Address ID/FEI Business Operations TIME CAP LABORATORIES, INC. 037052099 manufacture(49483-003)