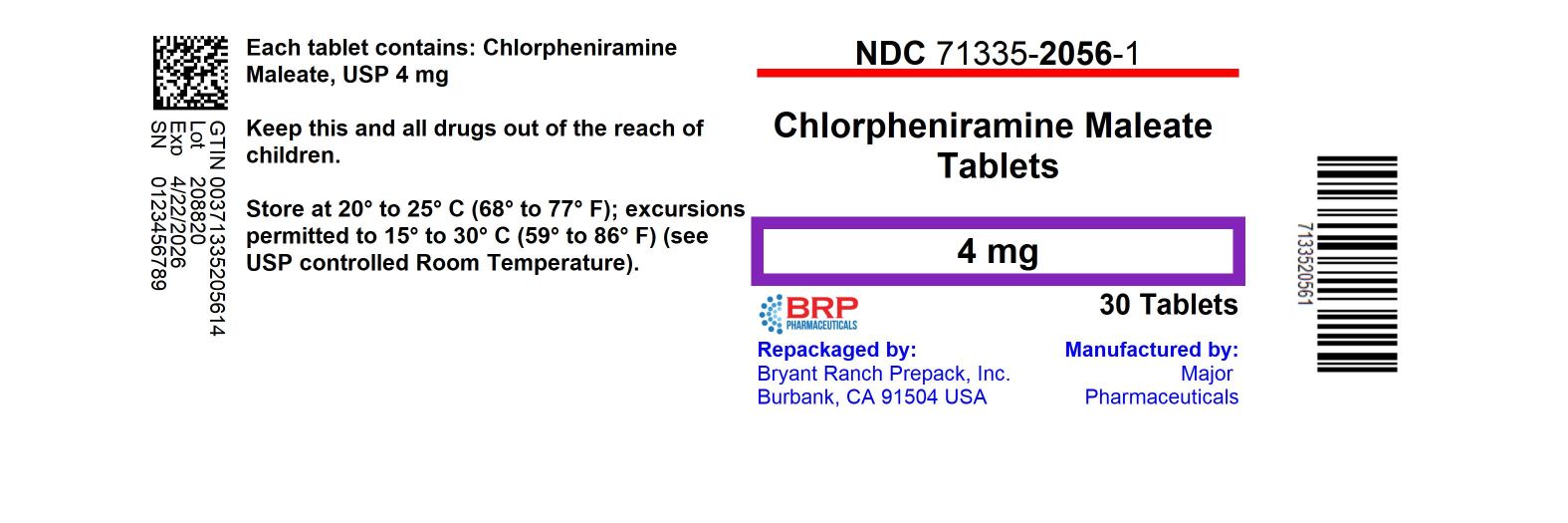
Label: ALLERGY- chlorpheniramine maleate tablet
-
NDC Code(s):
71335-2056-1,
71335-2056-2,
71335-2056-3,
71335-2056-4, view more71335-2056-5, 71335-2056-6
- Packager: Bryant Ranch Prepack
- This is a repackaged label.
- Source NDC Code(s): 0904-0012
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 3, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
HOW SUPPLIED
Chlorpheniramine Maleate 4 mg
NDC: 71335-2056-1: 30 Tablets in a BOTTLE, PLASTIC
NDC: 71335-2056-2: 60 Tablets in a BOTTLE, PLASTIC
NDC: 71335-2056-3: 100 Tablets in a BOTTLE, PLASTIC
NDC: 71335-2056-4: 120 Tablets in a BOTTLE, PLASTIC
NDC: 71335-2056-5: 40 Tablets in a BOTTLE, PLASTIC
NDC: 71335-2056-6: 24 Tablets in a BOTTLE, PLASTIC
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALLERGY
chlorpheniramine maleate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71335-2056(NDC:0904-0012) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 4 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color yellow Score 2 pieces Shape ROUND Size 8mm Flavor Imprint Code 44;194 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71335-2056-1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/02/2022 2 NDC:71335-2056-2 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/03/2024 3 NDC:71335-2056-3 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/03/2024 4 NDC:71335-2056-4 120 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/03/2024 5 NDC:71335-2056-5 40 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/03/2024 6 NDC:71335-2056-6 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/03/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 12/19/1992 Labeler - Bryant Ranch Prepack (171714327) Registrant - Bryant Ranch Prepack (171714327) Establishment Name Address ID/FEI Business Operations Bryant Ranch Prepack 171714327 REPACK(71335-2056) , RELABEL(71335-2056)