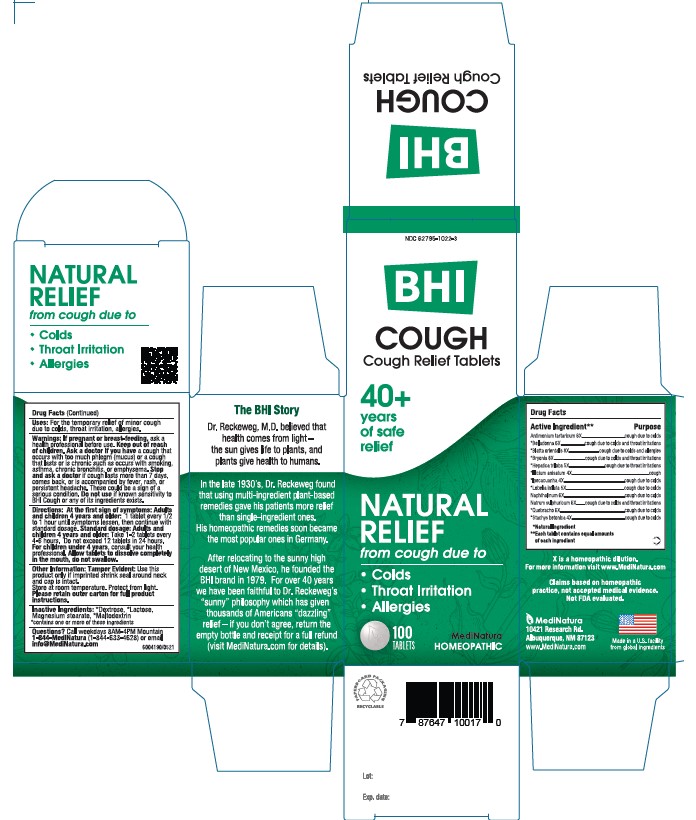
Label: BHI COUGH- antimony potassium tartrate, atropa belladonna, blatta orientalis, bryonia alba whole, anemone americana, star anise, ipecac, lobelia inflata, naphthalene, sodium sulfide nonahydrate, aspidosperma quebracho-blanco bark, and stachys officinalis tablet
- NDC Code(s): 62795-1022-3
- Packager: MediNatura Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated July 11, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- INACTIVE INGREDIENTS
- PURPOSE
- USES
-
DIRECTIONS
At first sign of symptoms: Adults and children 4years and older:
1 tablet every 1/2 to 1 hour until symptoms lessen, then continue with standard dosage.
Standard dosage: Adults and children 4 years and older:
Take 1-2 tablets every 4 to 6 hours. Do not exceed 12 tablets in 24 hours. For children under 4, consult your health professional.
Allow tablets to dissolve completely in the mouth, do not swallow
.
-
WARNINGS
If pregnant or breast-feeding, ask a health professional before use. Keep out of reach of children. Ask a doctor if you have a cough that occurs with too much phlegm (mucus) or a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema. Stop and ask a doctor if cough lasts for more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition. Do not use if known sensitivity to Cough or any of its ingredients exists.
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BHI COUGH
antimony potassium tartrate, atropa belladonna, blatta orientalis, bryonia alba whole, anemone americana, star anise, ipecac, lobelia inflata, naphthalene, sodium sulfide nonahydrate, aspidosperma quebracho-blanco bark, and stachys officinalis tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62795-1022 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ANTIMONY POTASSIUM TARTRATE (UNII: DL6OZ476V3) (ANTIMONY CATION (3+) - UNII:069647RPT5) ANTIMONY POTASSIUM TARTRATE 6 [hp_X] ATROPA BELLADONNA (UNII: WQZ3G9PF0H) (ATROPA BELLADONNA - UNII:WQZ3G9PF0H) ATROPA BELLADONNA 6 [hp_X] BLATTA ORIENTALIS (UNII: 535787266D) (BLATTA ORIENTALIS - UNII:535787266D) BLATTA ORIENTALIS 8 [hp_X] BRYONIA ALBA WHOLE (UNII: 56K0VVT47P) (BRYONIA ALBA WHOLE - UNII:56K0VVT47P) BRYONIA ALBA WHOLE 8 [hp_X] ANEMONE HEPATICA VAR. OBTUSA (UNII: 6ICN6T7FEM) (ANEMONE HEPATICA VAR. OBTUSA - UNII:6ICN6T7FEM) ANEMONE HEPATICA VAR. OBTUSA 5 [hp_X] STAR ANISE (UNII: XKC1657P78) (STAR ANISE - UNII:XKC1657P78) STAR ANISE 4 [hp_X] IPECAC (UNII: 62I3C8233L) (IPECAC - UNII:62I3C8233L) IPECAC 4 [hp_X] LOBELIA INFLATA (UNII: 9PP1T3TC5U) (LOBELIA INFLATA - UNII:9PP1T3TC5U) LOBELIA INFLATA 5 [hp_X] NAPHTHALENE (UNII: 2166IN72UN) (NAPHTHALENE - UNII:2166IN72UN) NAPHTHALENE 6 [hp_X] SODIUM SULFIDE NONAHYDRATE (UNII: C02T02993U) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM SULFIDE NONAHYDRATE 6 [hp_X] STACHYS OFFICINALIS (UNII: UO9989Y17N) (STACHYS OFFICINALIS - UNII:UO9989Y17N) STACHYS OFFICINALIS 4 [hp_X] ASPIDOSPERMA QUEBRACHO-BLANCO BARK (UNII: 52B1340190) (ASPIDOSPERMA QUEBRACHO-BLANCO BARK - UNII:52B1340190) ASPIDOSPERMA QUEBRACHO-BLANCO BARK 6 [hp_X] Inactive Ingredients Ingredient Name Strength LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) DEXTROSE (UNII: IY9XDZ35W2) Product Characteristics Color white Score no score Shape ROUND Size 9mm Flavor Imprint Code Leafman Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62795-1022-3 1 in 1 CARTON 08/30/2014 03/31/2025 1 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/30/2014 03/31/2025 Labeler - MediNatura Inc (079324099) Establishment Name Address ID/FEI Business Operations MediNatura Inc 102783016 manufacture(62795-1022)