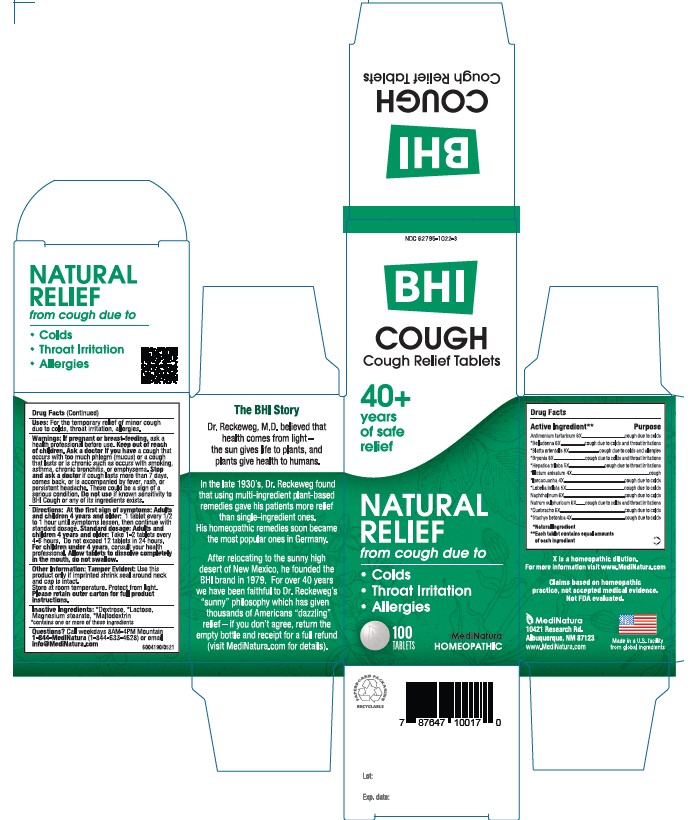
ACTIVE INGREDIENTS
Each tablet contains: Antimonium tartaricum 6X, *Belladonna 6X, *Blatta orientalis 8X, *Bryonia alba 8X, *Hepatica triloba 5X, *Illicium anisatum 4X, *Ipecacuanha 4X, *Lobelia inflata 5X, Naphthalinum 6X, Natrum sulphuricum 6X, *Quebracho 6X, *Stachys betonica 4X 25 mg each.
*Natural Ingredients
INACTIVE INGREDIENTS
Inactive Ingredients: **Dextrose, ** Lactose, Magnesium Stearate, **Maltodextrin
**contains one or more of these ingredients.
DIRECTIONS
At first sign of symptoms: Adults and children 4years and older:
1 tablet every 1/2 to 1 hour until symptoms lessen, then continue with standard dosage.
Standard dosage: Adults and children 4 years and older:
Take 1-2 tablets every 4 to 6 hours. Do not exceed 12 tablets in 24 hours. For children under 4, consult your health professional.
Allow tablets to dissolve completely in the mouth, do not swallow
.
WARNINGS
If pregnant or breast-feeding, ask a health professional before use. Keep out of reach of children. Ask a doctor if you have a cough that occurs with too much phlegm (mucus) or a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema. Stop and ask a doctor if cough lasts for more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition. Do not use if known sensitivity to Cough or any of its ingredients exists.