Label: NYSTATIN cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-0242-0, 54868-0242-1 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 51672-1289
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 10, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Nystatin Cream is for dermatologic use.
Nystatin is a polyene antimycotic obtained from Streptomyces noursei. It is a yellow to light tan powder with a cereal-like odor, very soluble in water, and slightly to sparingly soluble in alcohol. Structural formula:
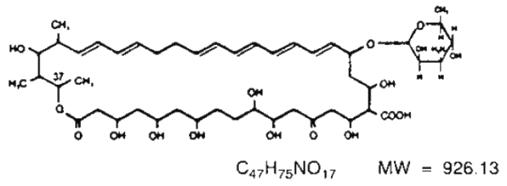
Nystatin Cream contains the antifungal antibiotic nystatin at a concentration of 100,000 USP Nystatin Units per gram in an aqueous, perfumed cream base containing purified water, propylene glycol, methylparaben, propylparaben, white petrolatum, glyceryl monostearate, polyethylene glycol 400 monostearate, ceteareth-15, medical antifoam AF emulsion, aluminum hydroxide gel, titanium dioxide, sorbitol solution, and, if necessary, sodium hydroxide for pH adjustment.
-
CLINICAL PHARMACOLOGY
Nystatin is an antifungal antibiotic which is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi. It probably acts by binding to sterols in the cell membrane of the fungus with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin is the first well tolerated antifungal antibiotic of dependable efficacy for the treatment of cutaneous, oral and intestinal infections caused by Candida (Monilia) albicans and other Candida species. It exhibits no appreciable activity against bacteria.
Nystatin provides specific therapy for all localized forms of candidiasis. Symptomatic relief is rapid, often occurring within 24 to 72 hours after the initiation of treatment. Cure is effected both clinically and mycologically in most cases of localized candidiasis.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
- PRECAUTIONS
- ADVERSE REACTIONS
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Nystatin Cream is a smooth yellow cream with a characteristic perfume odor.
Nystatin Cream is providing 100,000 USP Nystatin Units per gram in
15 gram tubes
NDC 54868-0242-1
30 gram tubes
NDC 54868-0242-0
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
Mfd. by: Taro Pharmaceuticals Inc.
Brampton, Ontario, Canada L6T 1C1Dist. by: Taro Pharmaceuticals U.S.A., Inc.
Hawthorne, NY 10532Revised: June, 2005
PK-1161-3 262
Relabeling of "Additional" label by:
Physicians Total Care, Inc.
Tulsa, OK 74146
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NYSTATIN
nystatin creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-0242(NDC:51672-1289) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NYSTATIN (UNII: BDF1O1C72E) (NYSTATIN - UNII:BDF1O1C72E) NYSTATIN 100000 [USP'U] in 1 g Inactive Ingredients Ingredient Name Strength ALGELDRATE (UNII: 03J11K103C) CETEARETH-15 (UNII: 867H4YOZ8Z) METHYLPARABEN (UNII: A2I8C7HI9T) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITOL (UNII: 506T60A25R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-0242-1 1 in 1 CARTON 1 15 g in 1 TUBE 2 NDC:54868-0242-0 1 in 1 CARTON 2 30 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA064022 01/31/1995 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel


