Label: FOLITIN-Z- ferrous fumarate, folic acid tablet
- NDC Code(s): 59088-181-58
- Packager: PureTek Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION:
Full Prescribing Information:
Each Serving Size of 2 caplets contain:
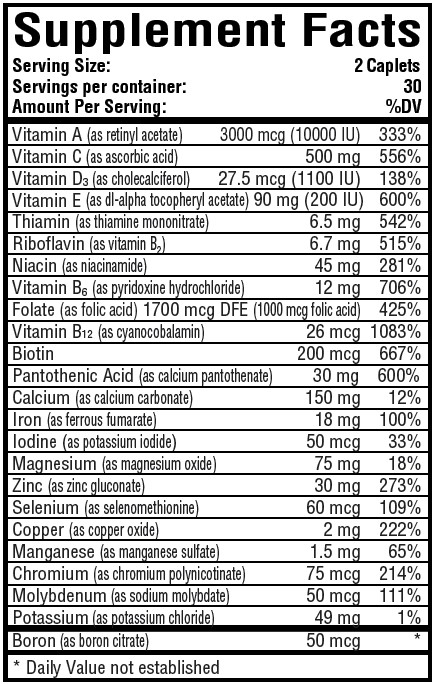
Other Ingredients: Microcrystalline Cellulose, Silicon Dioxide, Crospovidone, Magnesium Stearate, Coating:(Sodium Carboxymethylcellulose, Dextrose Monohydrate, Titanium Dioxide, Dextrin, Purified Stearic Acid, FD&C Yellow #6/Sunset Yellow FCF Aluminum Lake).
-
INDICATIONS:
Folitin-Z TM is indicated for the treatment of iron deficiency anemia and folate deficiency as in extended convalescence, menorrhagia, pregnancy, puberty, excessive blood loss, and advanced age. Also for the treatment of the condition in which iron deficiency and vitamin C deficiency occurs together, along with a deficient intake or increased need for B-Complex vitamins in chronic and acute illness, as well as cases of metabolic stress, and in convalescence.
-
CONTRAINDICATIONS:
This product is contraindicated in patients with known hypersensitivity to any of its ingredients; also, all iron compounds are contraindicated in patients with hemosiderosis, hemochromatosis, or hemolytic anemias. Pernicious anemia is a contraindication, as folic acid may obscure its signs and symptoms.
-
WARNING:
Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately.
Administration of folic acid alone is improper therapy for pernicious anemia and other megaloblastic anemias in which vitamin B12 is deficient.Precaution Section
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia, in that hematologic remission can occur while neurological manifestations remain progressive.
There is a potential danger in administering folic acid to patients with undiagnosed anemia, since folic acid may obscure the diagnosis of pernicious anemia by alleviating the hematologic manifestations of the disease while allowing the neurologic complications to progress. This may result in severe nervous system damage before the correct diagnosis is made. Adequate doses of vitamin B12 may prevent, halt, or improve the neurologic changes caused by pernicious anemia.
The patient’s medical conditions and consumption of other drugs, herbs, and/or supplements should be considered.For use on the order of a healthcare practitioner.
Call your doctor about side effects. To report side effects, call PureTek Corporation at
1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.Drug Interactions:
Folitin-Z TM is not recommended for and should not be given to patients receiving levodopa because the action of levodopa is antagonized by pyridoxine. There is a possibility of increased bleeding due to pyridoxine interaction with anticoagulants (e.g., Aspirin, Heparin, or Clopidogrel).
Adverse Reactions:
Folic Acid: Allergic sensitizations have been reported following both oral and parenteral administration of folic acid.
Ferrous Fumarate: Gastrointestinal disturbances (anorexia, nausea, diarrhea, constipation) occur occasionally, but are usually mild and may subside with continuation of therapy. Although the absorption of iron is best when taken between meals, giving Folitin-Z TM after meals may control occasional gastrointestinal disturbances. Folitin-Z TM is best absorbed when taken at bedtime. Adverse reactions have been reported with specific vitamins and minerals but generally at levels substantially higher than those contained herein. However, allergic and idiosyncratic reactions are possible at lower levels. Iron, even at the usually recommended levels, has been associated with gastrointestinal intolerance in some patients. -
OVERDOSE:
Iron: Signs and Symptoms: Iron is toxic. Acute overdosage of iron may cause nausea and vomiting and, in severe cases, cardiovascular collapse and death. Other symptoms include pallor and cyanosis, melena, shock, drowsiness, and coma. The estimated overdose of orally ingested iron is 300 mg/kg body weight. When overdoses are ingested by children, severe reactions, including fatalities, have resulted. Folitin-Z TM should be stored beyond the reach of children to prevent against accidental iron poisoning. Keep this and all other drugs out of reach of children.
- DOSAGE AND ADMINISTRATION:
- HOW SUPPLIED:
-
STORAGE:
Do not use if bottle seal is broken.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Store at controlled room temperature 20o to 25oC (68o to 77oF). [See USP].
Protect from light and moisture and avoid excessive heat.
To report a serious adverse event or to obtain product information, contact
1-877-921-7873. - Chronocap
- Folitin-Z TM
-
INGREDIENTS AND APPEARANCE
FOLITIN-Z
ferrous fumarate, folic acid tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59088-181 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VITAMIN A ACETATE (UNII: 3LE3D9D6OY) (VITAMIN A - UNII:81G40H8B0T) VITAMIN A 1500 ug ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 250 mg CHOLECALCIFEROL (UNII: 1C6V77QF41) (CHOLECALCIFEROL - UNII:1C6V77QF41) CHOLECALCIFEROL 13.75 ug .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) (.ALPHA.-TOCOPHEROL, DL- - UNII:7QWA1RIO01) .ALPHA.-TOCOPHEROL, DL- 45 mg THIAMINE MONONITRATE (UNII: 8K0I04919X) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 3.25 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 3.35 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 22.5 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 6 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 500 ug CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 13 ug BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 100 ug PANTOTHENIC ACID (UNII: 19F5HK2737) (PANTOTHENIC ACID - UNII:19F5HK2737) PANTOTHENIC ACID 15 mg CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CATION 75 mg FERROUS FUMARATE (UNII: R5L488RY0Q) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 9 mg POTASSIUM IODIDE (UNII: 1C4QK22F9J) (IODIDE ION - UNII:09G4I6V86Q) IODIDE ION 25 ug MAGNESIUM OXIDE (UNII: 3A3U0GI71G) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM CATION 37.5 mg ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 15 mg SELENIUM (UNII: H6241UJ22B) (SELENIUM - UNII:H6241UJ22B) SELENIUM 30 ug CUPROUS OXIDE (UNII: T8BEA5064F) (CUPROUS OXIDE - UNII:T8BEA5064F) CUPROUS OXIDE 1 mg MANGANESE SULFATE (UNII: W00LYS4T26) (MANGANESE CATION (2+) - UNII:H6EP7W5457) MANGANESE CATION (2+) 0.75 mg CHROMIUM NICOTINATE (UNII: A150AY412V) (NIACIN - UNII:2679MF687A) CHROMIUM NICOTINATE 37.5 ug MOLYBDENUM (UNII: 81AH48963U) (MOLYBDENUM - UNII:81AH48963U) MOLYBDENUM 25 ug POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CATION 24.5 mg BORON (UNII: N9E3X5056Q) (BORON - UNII:N9E3X5056Q) BORON 25 ug Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CROSPOVIDONE (UNII: 2S7830E561) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) DEXTROSE MONOHYDRATE (UNII: LX22YL083G) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ICODEXTRIN (UNII: 2NX48Z0A9G) STEARIC ACID (UNII: 4ELV7Z65AP) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color yellow (With Slightly Brown Specks) Score no score Shape CAPSULE (Oblong Caplet) Size 20mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59088-181-58 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/11/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 02/11/2021 Labeler - PureTek Corporation (785961046)




