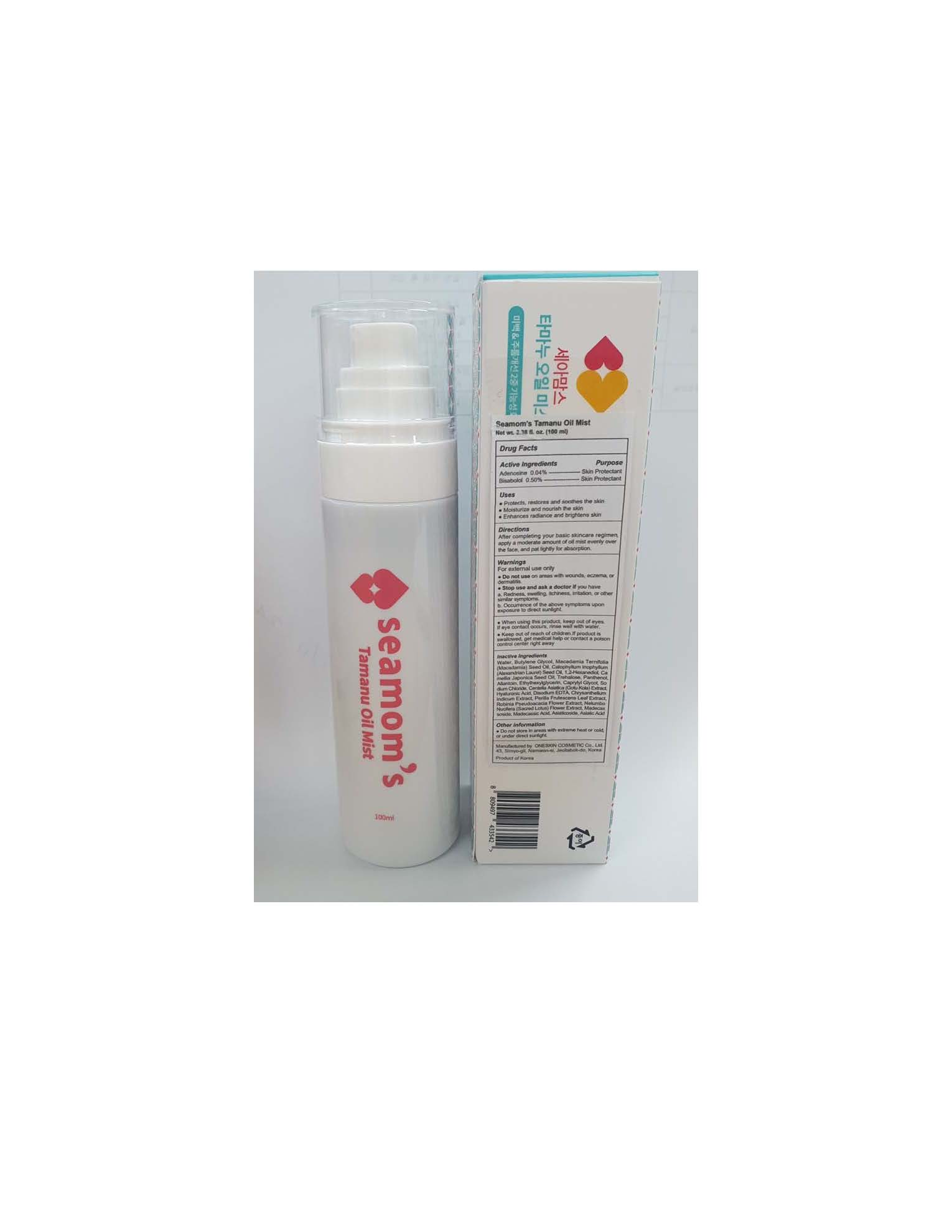
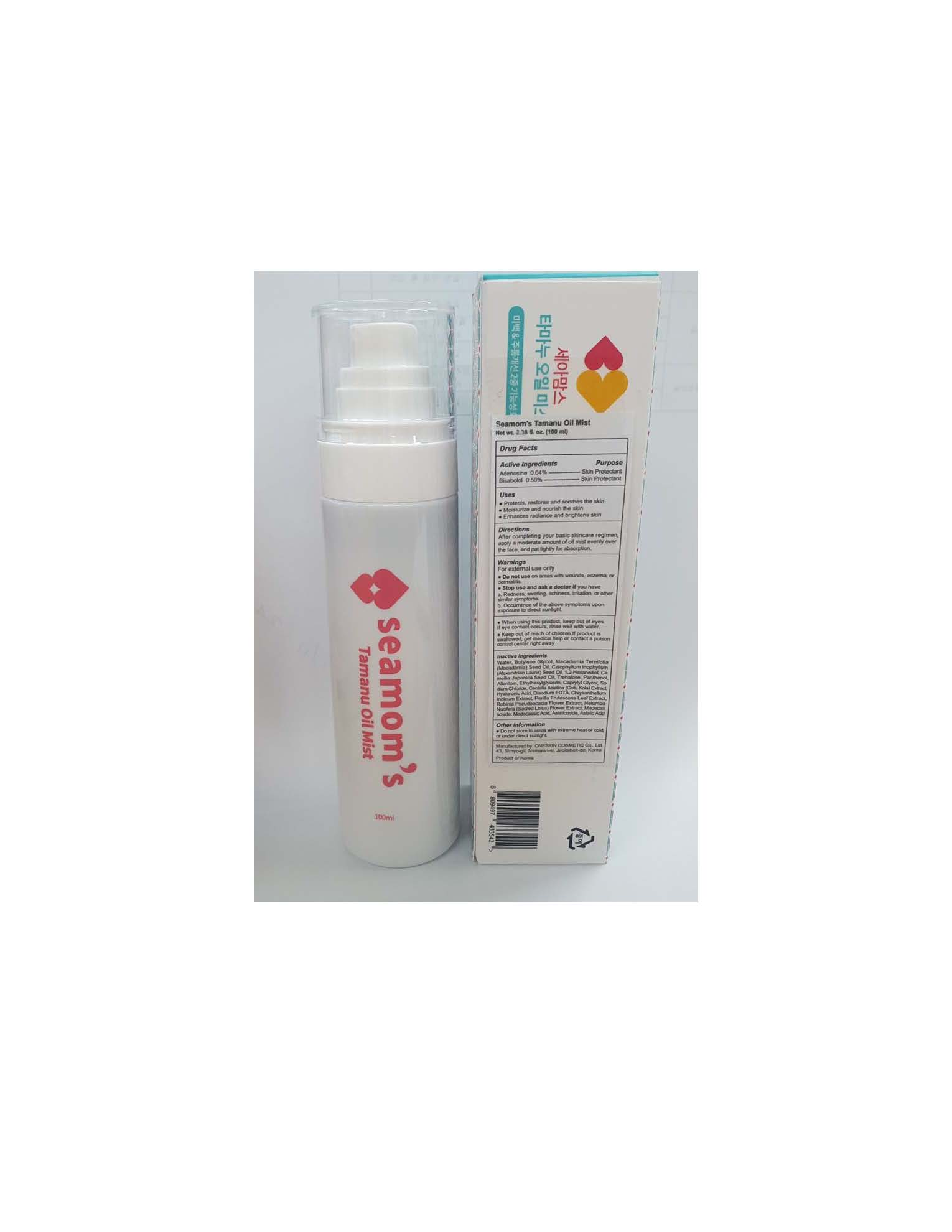
Label: SEAMOMS TAMANU OIL MIST- bisabolol and adenosine oil mist oil
-
Contains inactivated NDC Code(s)
NDC Code(s): 70889-920-10 - Packager: Oneskin Cosmetics Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 27, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
-
WARNINGS
For external use only
- Do not use on areas with wounds, eczema, or dermatitis.
- Stop use and ask a doctor if you have
a. Redness, swelling, itchiness, irritation, or other similar symptoms.
b. Occurrence of the above symptoms upon exposure to direct sunlight.- When using this product, keep out of eyes. If eye contact occurs, rinse well with water.
- Keep out of reach of children. If product is swallowed, get medical help or contact a poison control center right away
- KEEP OUT OF REACH OF CHILDREN
-
INACTIVE INGREDIENT
Water, Butylene Glycol, Macadamia Ternifolia (Macadamia) Seed Oil, Calophyllum Inophyllum (Alexandrian Laurel) Seed Oil, 1,2-Hexanediol, Camellia Japonica Seed Oil, Trehalose, Panthenol, Allantoin, Ethylhexylglycerin, Caprylyl Glycol, Sodium Chloride, Centella Asiatica (Gotu Kola) Extract, Hyaluronic Acid, Disodium EDTA, Chrysanthellum Indicum Extract, Perilla Frutescens Leaf Extract, Robinia Pseudoacacia Flower Extract, Nelumbo Nucifera (Sacred Lotus) Flower Extract, Madecassoside, Madecassic Acid, Asiaticoside, Asiatic Acid.
- OTHER SAFETY INFORMATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SEAMOMS TAMANU OIL MIST
bisabolol and adenosine oil mist oilProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70889-920 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 mL LEVOMENOL (UNII: 24WE03BX2T) (LEVOMENOL - UNII:24WE03BX2T) LEVOMENOL 0.5 g in 100 mL Inactive Ingredients Ingredient Name Strength MACADAMIA OIL (UNII: 515610SU8C) WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALLANTOIN (UNII: 344S277G0Z) EDETATE DISODIUM (UNII: 7FLD91C86K) Product Characteristics Color Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70889-920-10 1 in 1 BOX 01/27/2021 1 100 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 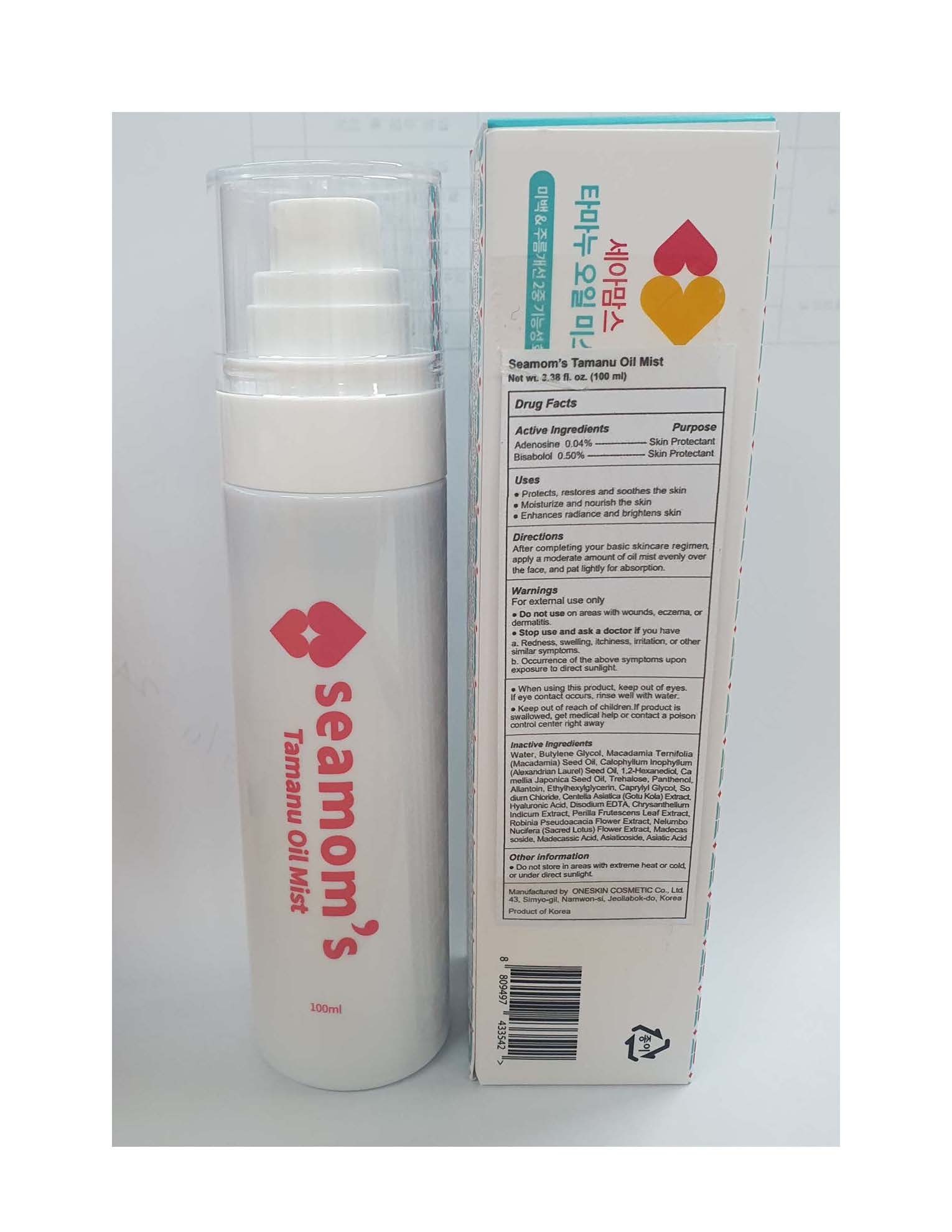
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/27/2021 Labeler - Oneskin Cosmetics Co., Ltd. (689846630) Registrant - Oneskin Cosmetics Co., Ltd. (689846630) Establishment Name Address ID/FEI Business Operations Oneskin Cosmetics Co., Ltd. 689846630 manufacture(70889-920)