Label: CVS IODINE- iodine, sodium iodide and alcohol liquid
- NDC Code(s): 69842-213-91
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 20, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
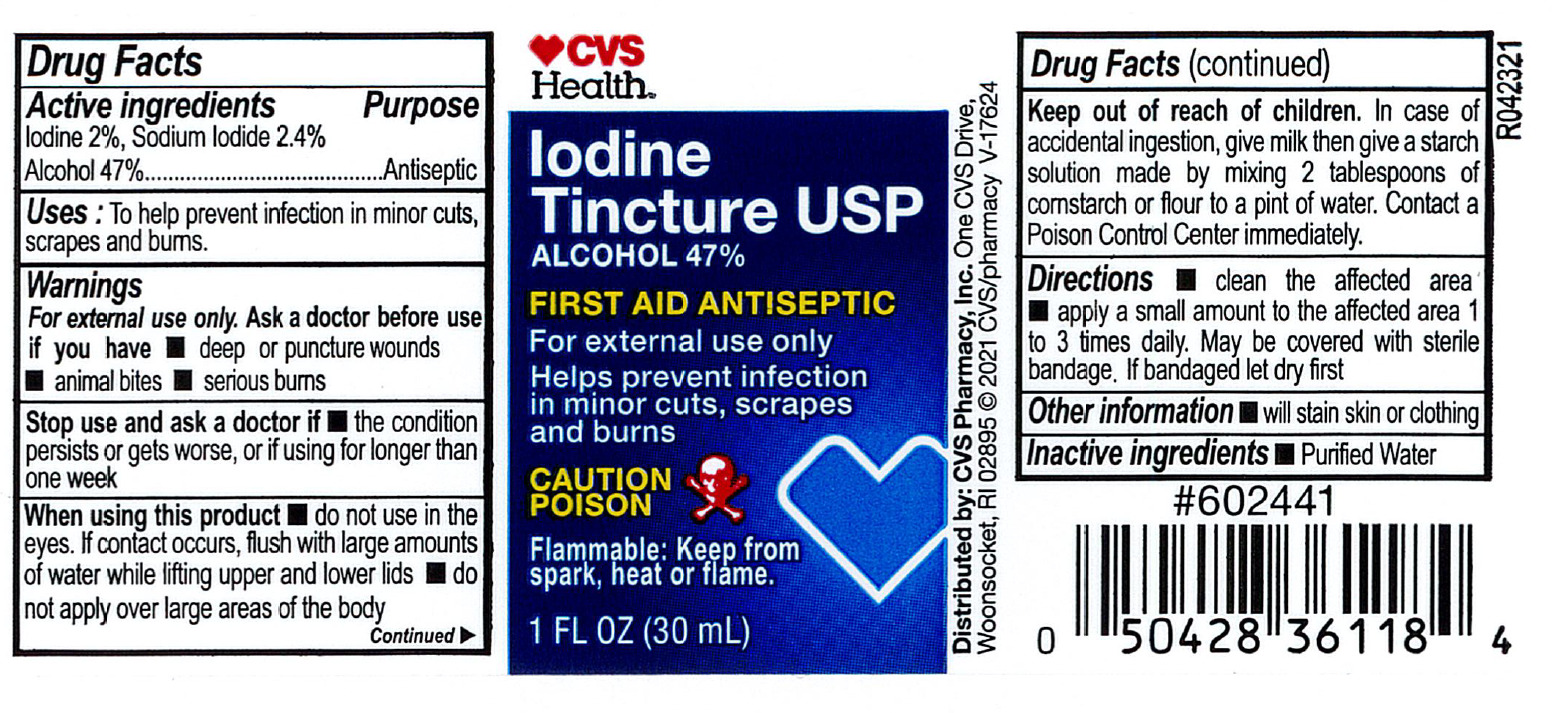
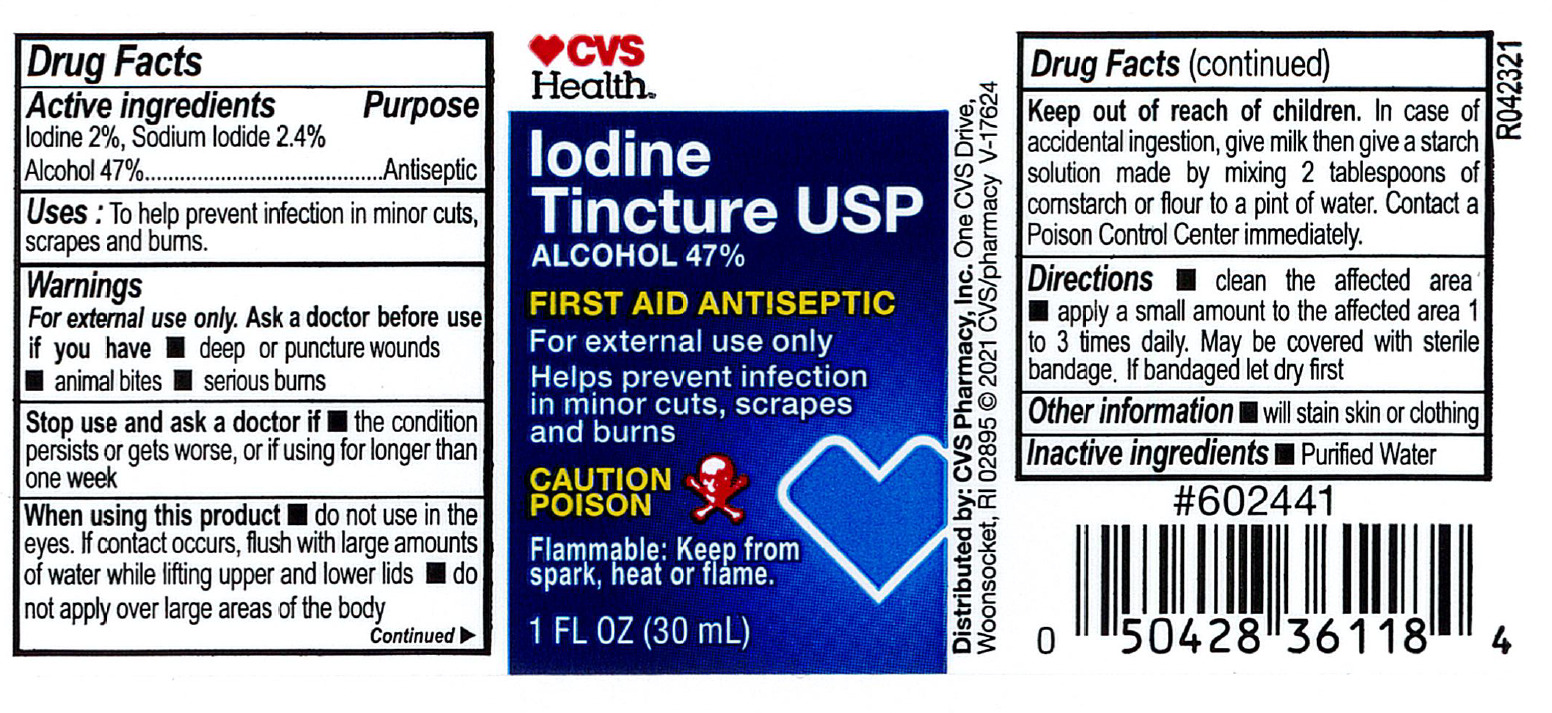
- Active Ingredient
- Purpose
- Indications
- Warnings:
- Directions
- Inactive Ingredient
- Other Information
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CVS IODINE
iodine, sodium iodide and alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-213 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IODINE (UNII: 9679TC07X4) (IODINE - UNII:9679TC07X4) IODINE 20 mg in 1 mL SODIUM IODIDE (UNII: F5WR8N145C) (IODIDE ION - UNII:09G4I6V86Q) IODIDE ION 20.4 mg in 1 mL ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 470 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-213-91 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 01/01/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/01/2008 Labeler - CVS Pharmacy (062312574) Registrant - Pharma Nobis, LLC (118564114) Establishment Name Address ID/FEI Business Operations Pharma Nobis, LLC 118564114 manufacture(69842-213) , analysis(69842-213) , pack(69842-213) , label(69842-213)




 CVS Health
CVS Health
