Label: TC MAX- calcium carbonate powder, for solution
TC MAX- calcium carbonate kit
- NDC Code(s): 80733-005-01, 80733-005-05, 80733-007-06
- Packager: Romavision Investing LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 29, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
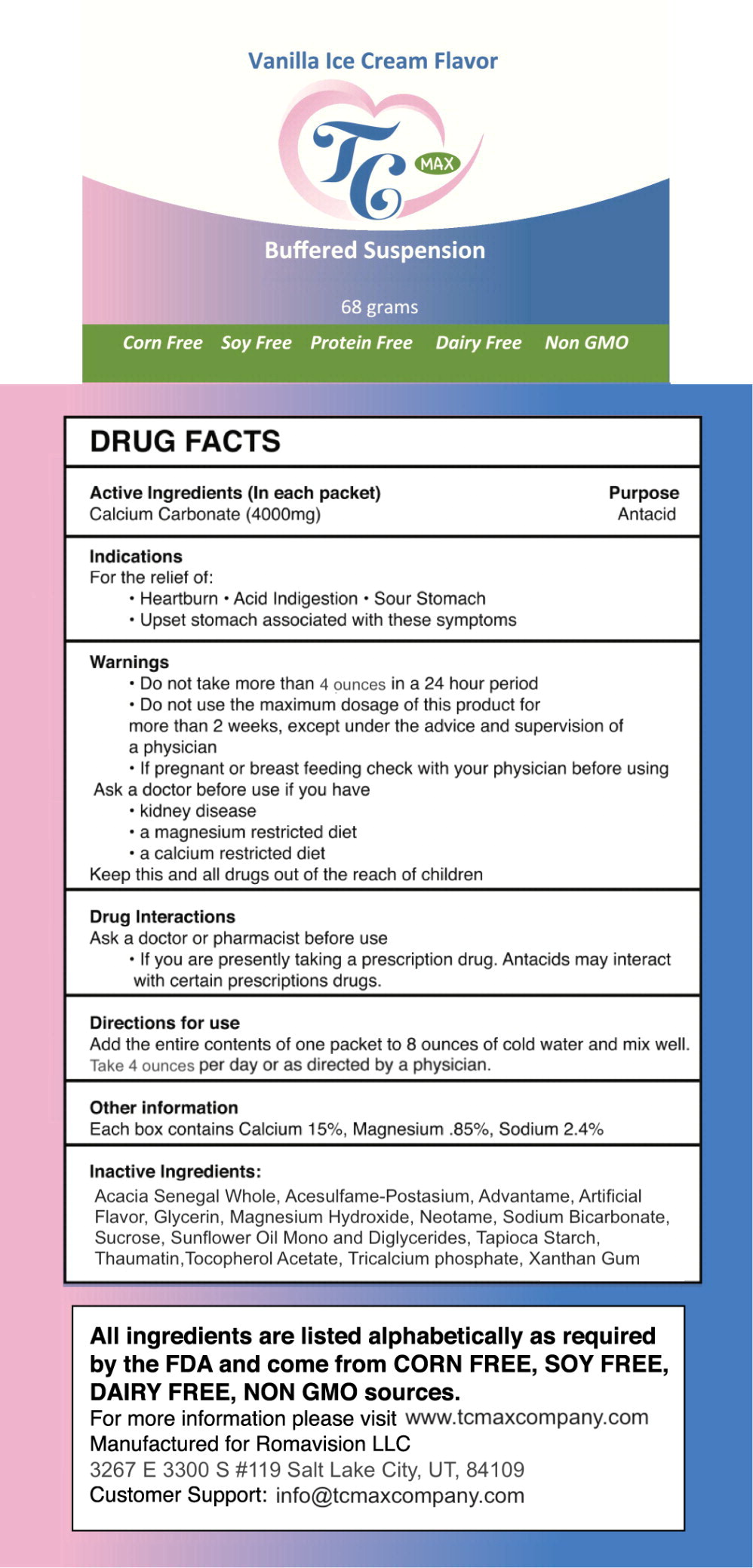
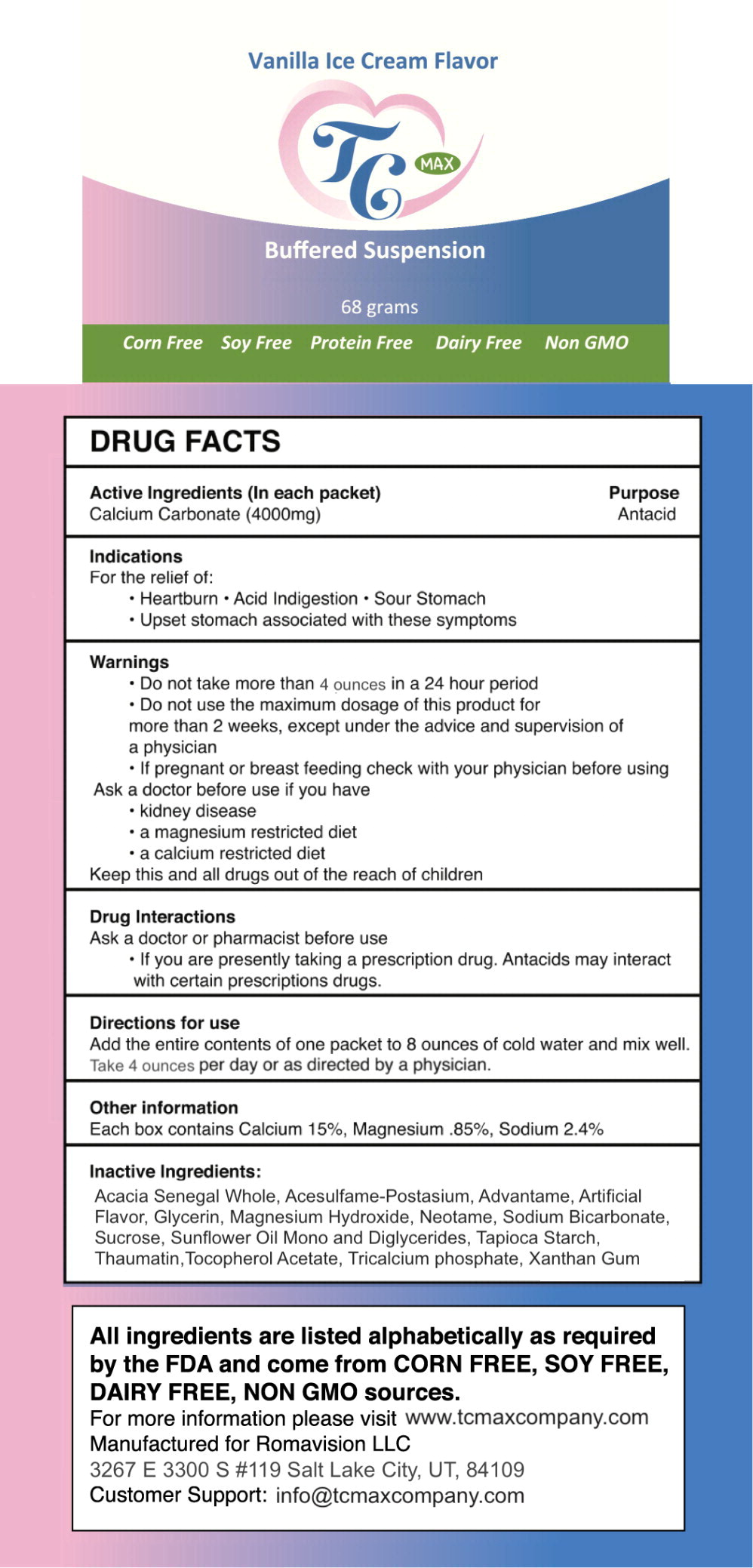
- Active Ingredients (In each packet)
- Purpose
- Indications
- Warnings
- Drug Interactions
- Directions for use
- Other information
- Inactive Ingredients:
- Principal Display Panel - 68 gram Packet Label
- Principal Display Panel - 68 gram Packet Label
-
INGREDIENTS AND APPEARANCE
TC MAX
calcium carbonate powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80733-005 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength calcium carbonate (UNII: H0G9379FGK) (calcium cation - UNII:2M83C4R6ZB) calcium carbonate 4 g in 237 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) STARCH, TAPIOCA (UNII: 24SC3U704I) MALTODEXTRIN (UNII: 7CVR7L4A2D) TRICALCIUM PHOSPHATE (UNII: K4C08XP666) SUNFLOWER OIL MONO/DIGLYCERIDES (UNII: HS7EB0C1ZH) SUNFLOWER OIL (UNII: 3W1JG795YI) XANTHAN GUM (UNII: TTV12P4NEE) NEOTAME (UNII: VJ597D52EX) SODIUM BICARBONATE (UNII: 8MDF5V39QO) MAGNESIUM HYDROXIDE (UNII: NBZ3QY004S) ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) THAUMATIN (UNII: KNC9Q0EE6G) SUCROSE (UNII: C151H8M554) ADVANTAME (UNII: 3ZA6810AWX) ACACIA SENEGAL WHOLE (UNII: QP4QYZ033C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80733-005-01 237 mL in 1 PACKET; Type 0: Not a Combination Product 07/18/2019 2 NDC:80733-005-05 5 in 1 BOX 07/18/2019 2 NDC:80733-005-01 237 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part331 06/03/2019 TC MAX
calcium carbonate kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80733-007 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80733-007-06 1 in 1 BOX 07/18/2019 1 1 in 1 KIT; Type 1: Convenience Kit of Co-Package Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 6 PACKET 1422 mL Part 1 of 1 TC MAX
calcium carbonate powder, for solutionProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength calcium carbonate (UNII: H0G9379FGK) (calcium cation - UNII:2M83C4R6ZB) calcium carbonate 4 g in 237 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) STARCH, TAPIOCA (UNII: 24SC3U704I) MALTODEXTRIN (UNII: 7CVR7L4A2D) TRICALCIUM PHOSPHATE (UNII: K4C08XP666) SUNFLOWER OIL MONO/DIGLYCERIDES (UNII: HS7EB0C1ZH) SUNFLOWER OIL (UNII: 3W1JG795YI) XANTHAN GUM (UNII: TTV12P4NEE) NEOTAME (UNII: VJ597D52EX) SODIUM BICARBONATE (UNII: 8MDF5V39QO) MAGNESIUM HYDROXIDE (UNII: NBZ3QY004S) ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) THAUMATIN (UNII: KNC9Q0EE6G) SUCROSE (UNII: C151H8M554) ADVANTAME (UNII: 3ZA6810AWX) ACACIA SENEGAL WHOLE (UNII: QP4QYZ033C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 6 in 1 NOT APPLICABLE 1 237 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part331 07/18/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part331 07/18/2019 Labeler - Romavision Investing LLC (080808687) Establishment Name Address ID/FEI Business Operations Identipak, Inc. 942862350 MANUFACTURE(80733-005, 80733-007)