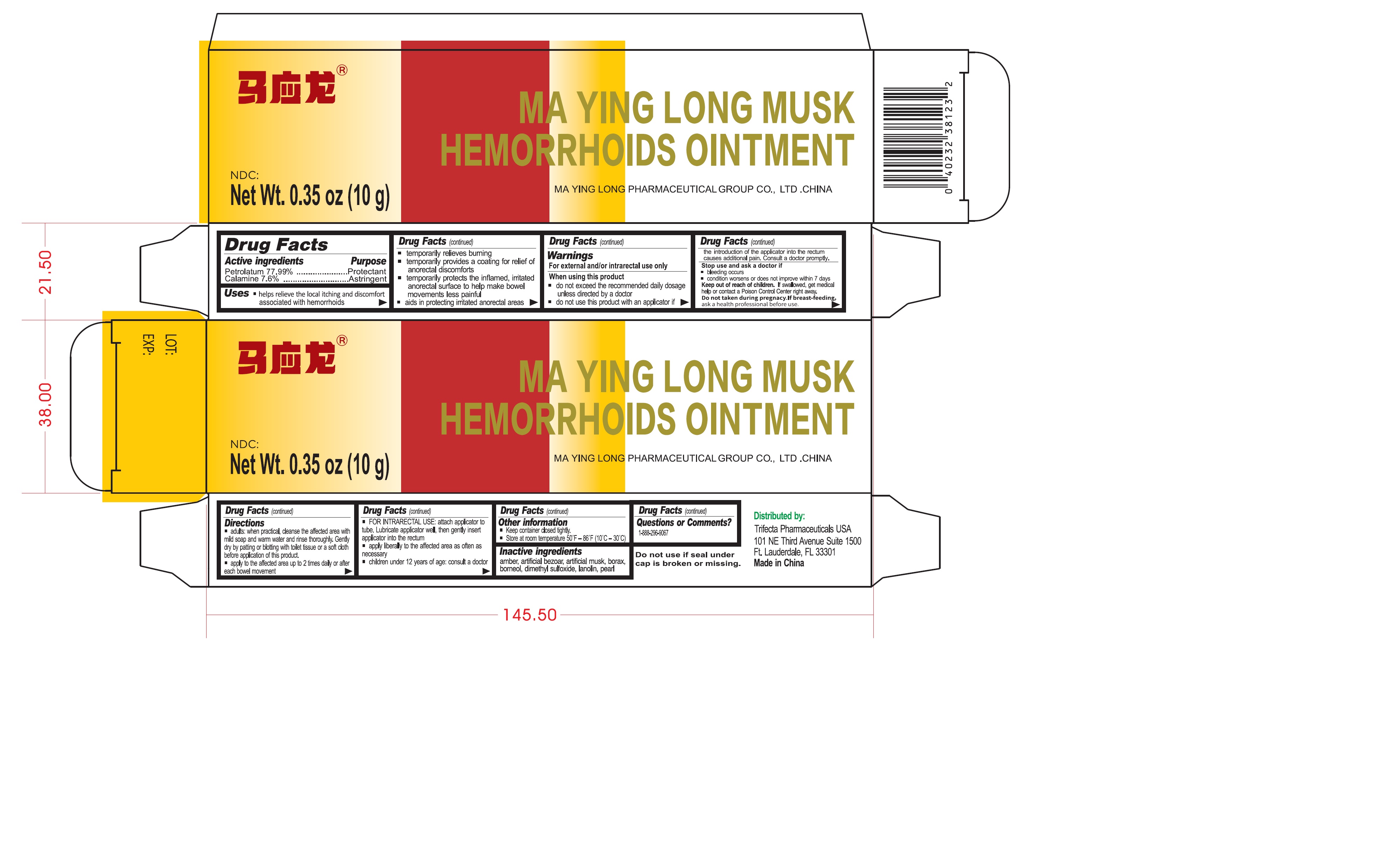
Label: MA YING LONG MUSK HEMORRHOIDS- petrolatum, zinc oxide ointment
- NDC Code(s): 68511-997-01, 68511-997-02, 68511-997-20
- Packager: Mayinglong Pharmaceutical Group Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
-
Uses
- helps relieve the local itching and discomfort associated with hemorrhoids
- temporaily relieves burning
- temporarily provides a coating for relief of anorectal discomforts
- temporarily protects the inflamed, irritated anorectal surface to help make bowel movements less painful
- aids in protecting irritated anorectal areas
-
Warnings
For external and/or intrarectal use only
When using this product
- do not exceed the recommended daily dosage unless directed by a doctor
- do not use this product with an applicator if the introduction of the applicator into the rectum causes additional pain. Consult a doctor promptly.
-
Directions
- adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product.
- apply to the affected area up to 2 times daily or after each bowel movement
- FOR INTRARECTAL USE: attach applicator to tube. Lubricant applicator well, then gently insert applicator into the rectum
- apply liberally to the affected area as often as necessary
- children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Questions or Comments?
- Package Labeling
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
MA YING LONG MUSK HEMORRHOIDS
petrolatum, zinc oxide ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68511-997 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 779.9 mg in 1 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 76 mg in 1 g Inactive Ingredients Ingredient Name Strength BOS TAURUS GALLSTONE (UNII: 4162LZ0N4O) AMBER (UNII: 70J9Z0J26P) 6-TERT-BUTYL-3-METHYL-2,4-DINITROANISOLE (UNII: 55V150W8R6) SODIUM BORATE (UNII: 91MBZ8H3QO) DIMETHYL SULFOXIDE (UNII: YOW8V9698H) LANOLIN (UNII: 7EV65EAW6H) PEARL (HYRIOPSIS CUMINGII) (UNII: A75L5FZ40U) BORNEOL (UNII: M89NIB437X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68511-997-01 1 in 1 BOX 12/15/2003 1 10 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:68511-997-02 1 in 1 BOX 06/22/2022 2 10 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:68511-997-20 1 in 1 BOX 05/15/2024 3 20 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M015 12/15/2003 Labeler - Mayinglong Pharmaceutical Group Co., Ltd. (526823828)