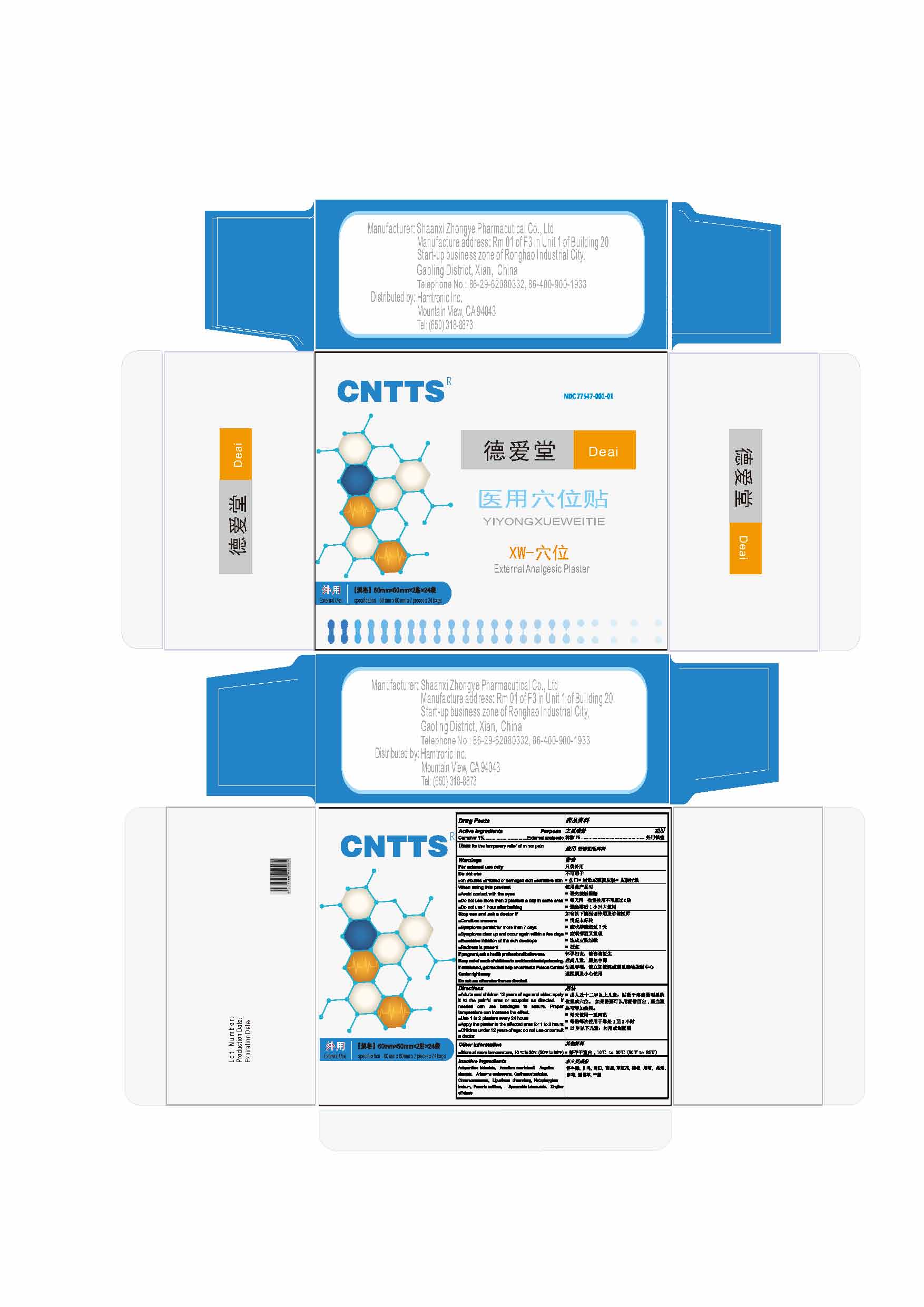
Label: YIYONGXUEWEITIE- camphor plaster
-
Contains inactivated NDC Code(s)
NDC Code(s): 77547-001-01, 77547-001-02 - Packager: Beijing Shenzhou Pharmaceutical
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 18, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- ASK DOCTOR
- Keep out of reach of children
- If pregnant
-
Directions
■Adults and children 12 years of age and older: apply it to the painful area or acupoint as directed. If needed can use bandages to secure. Proper temperature can increase the effect.
■Use 1 to 2 plasters every 24 hours
■Apply the plaster to the affected area for 1 to 2 hours
■Children under 12 years of age: do not use or consult a doctor. - Other information
- Inactive ingredients
- Purpose
- Do not use
- When using this product
- Stop use and ask a doctor if
- Use
- Uses
- Inner Bag
-
INGREDIENTS AND APPEARANCE
YIYONGXUEWEITIE
camphor plasterProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:77547-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (NATURAL) (UNII: N20HL7Q941) (CAMPHOR (NATURAL) - UNII:N20HL7Q941) CAMPHOR (NATURAL) 1 g in 100 g Inactive Ingredients Ingredient Name Strength ACHYRANTHES BIDENTATA ROOT (UNII: 5QIU26R6P1) ACONITUM CARMICHAELII ROOT (UNII: XJ28061U1S) ANGELICA SINENSIS ROOT (UNII: B66F4574UG) ARISAEMA ERUBESCENS WHOLE (UNII: SF6XYE1WTC) CARTHAMUS TINCTORIUS FLOWER BUD (UNII: B86IS274O0) CINNAMOMUM CASSIA TWIG (UNII: 4UD6VQR2UP) LIGUSTICUM SINENSE SUBSP. CHUANXIONG ROOT (UNII: RR83T99U97) NOTOPTERYGIUM INCISUM ROOT (UNII: 5Z2WW4J6RI) PAEONIA LACTIFLORA ROOT (UNII: 3Z3866YW6P) SPERANSKIA TUBERCULATA WHOLE (UNII: WP70OW6J01) ZINGIBER OFFICINALE WHOLE (UNII: IN6Q3S3414) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:77547-001-01 24 in 1 BOX 06/18/2020 1 2 in 1 PACKAGE 1 NDC:77547-001-02 1 g in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 06/18/2020 Labeler - Beijing Shenzhou Pharmaceutical (554506481)