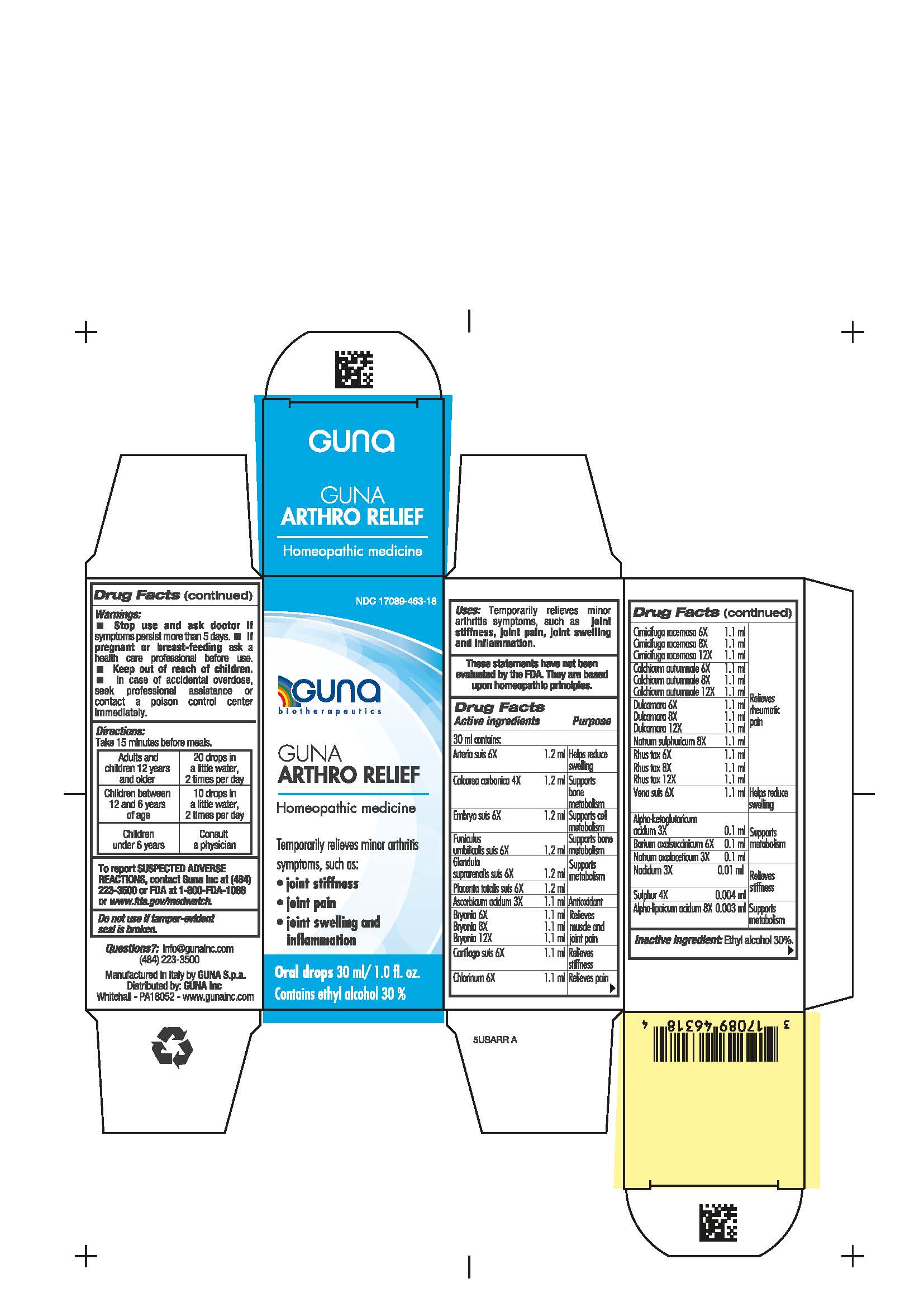
Label: GUNA ARTHRO RELIEF- arteria suis - alpha ketoglutaricum acidum - alpha lipoicum acidum - ascorbicum acidum - barium oxalsuccinicum - bryonia alba - calcarea carbonica - cartilago suis - chlorinum - cimicifuga racemosa - colchicum autumnale - dulcamara - embryo suis - funiculus umbilicalis suis - glandula suprarenalis suis - nadidum - natrum oxalaceticum - natrum sulphuricum - placenta totalis suis - rhus toxicodendron - sulphur- vena suis - solution/ drops
- NDC Code(s): 17089-463-18
- Packager: Guna spa
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 23, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INDICATIONS & USAGE
- INACTIVE INGREDIENT
- QUESTIONS
- WARNINGS
- DIRECTIONS
- PREGNANCY
- WARNINGS
- USES
-
ACTIVE INGREDIENTS/PURPOSE
Arteria suis 6X Helps reduce swelling
Calcarea carbonica 4X Supports bone metabolism
Embryo suis 6X Supports cell metabolism
Funiculus umbilicalis suis 6X Supports bone metabolism
Glandula suprarenalis suis 6X Supports metabolism
Placenta totalis suis 6X Supports metabolism
Ascorbicum acidum 3X Antioxidant
Bryonia alba 6X, 8X, 12X Relieves muscle and joint pain
Cartilago suis 6X Relieves stiffness
Chlorinum 6X Relieves pain
Cimicifuga racemosa 6X, 8X, 12X Relieves rheumatic pain
Colchicum autumnale 6X, 8X, 12X Relieves rheumatic pain
Dulcamara 6X, 8X, 12X Relieves rheumatic pain
Natrum sulphuricum 8X Relieves rheumatic pain
Rhus toxicodendron 6X, 8X, 12X Relieves rheumatic pain
Vena suis 6X Helps reduce swelling
Alpha-ketoglutaricum acidum 3X Supports metabolism
Barium oxalsuccinicum 6X Supports metabolism
Natrum oxalaceticum 3X Supports metabolism
Nadidum 3X Relieves stiffness
Sulphur 4X Relieves stiffness
Alpha-lipoicum acidum 8X Supports metabolism
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GUNA ARTHRO RELIEF
arteria suis - alpha ketoglutaricum acidum - alpha lipoicum acidum - ascorbicum acidum - barium oxalsuccinicum - bryonia alba - calcarea carbonica - cartilago suis - chlorinum - cimicifuga racemosa - colchicum autumnale - dulcamara - embryo suis - funiculus umbilicalis suis - glandula suprarenalis suis - nadidum - natrum oxalaceticum - natrum sulphuricum - placenta totalis suis - rhus toxicodendron - sulphur- vena suis - solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17089-463 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OYSTER SHELL CALCIUM CARBONATE, CRUDE (UNII: 2E32821G6I) (OYSTER SHELL CALCIUM CARBONATE, CRUDE - UNII:2E32821G6I) OYSTER SHELL CALCIUM CARBONATE, CRUDE 4 [hp_X] in 30 mL SODIUM SULFATE (UNII: 0YPR65R21J) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM SULFATE 8 [hp_X] in 30 mL .ALPHA.-KETOGLUTARIC ACID (UNII: 8ID597Z82X) (.ALPHA.-KETOGLUTARIC ACID - UNII:8ID597Z82X) .ALPHA.-KETOGLUTARIC ACID 3 [hp_X] in 30 mL .ALPHA.-LIPOIC ACID (UNII: 73Y7P0K73Y) (.ALPHA.-LIPOIC ACID - UNII:73Y7P0K73Y) .ALPHA.-LIPOIC ACID 8 [hp_X] in 30 mL SUS SCROFA ARTERY (UNII: 63O327782Q) (SUS SCROFA ARTERY - UNII:63O327782Q) SUS SCROFA ARTERY 6 [hp_X] in 30 mL ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 3 [hp_X] in 30 mL BARIUM OXALOSUCCINATE (UNII: L7A49804ZQ) (BARIUM CATION - UNII:V645272HLN) BARIUM OXALOSUCCINATE 6 [hp_X] in 30 mL BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 6 [hp_X] in 30 mL SUS SCROFA CARTILAGE (UNII: 73ECW5WG2F) (SUS SCROFA CARTILAGE - UNII:73ECW5WG2F) SUS SCROFA CARTILAGE 6 [hp_X] in 30 mL CHLORINE (UNII: 4R7X1O2820) (CHLORINE - UNII:4R7X1O2820) CHLORINE 6 [hp_X] in 30 mL BLACK COHOSH (UNII: K73E24S6X9) (BLACK COHOSH - UNII:K73E24S6X9) BLACK COHOSH 6 [hp_X] in 30 mL COLCHICUM AUTUMNALE BULB (UNII: 993QHL78E6) (COLCHICUM AUTUMNALE BULB - UNII:993QHL78E6) COLCHICUM AUTUMNALE BULB 6 [hp_X] in 30 mL SUS SCROFA EMBRYO (UNII: 9928MC12VO) (SUS SCROFA EMBRYO - UNII:9928MC12VO) SUS SCROFA EMBRYO 6 [hp_X] in 30 mL SOLANUM DULCAMARA FLOWER (UNII: W6J1279A6K) (SOLANUM DULCAMARA FLOWER - UNII:W6J1279A6K) SOLANUM DULCAMARA FLOWER 6 [hp_X] in 30 mL SUS SCROFA UMBILICAL CORD (UNII: 118OYG6W3H) (SUS SCROFA UMBILICAL CORD - UNII:118OYG6W3H) SUS SCROFA UMBILICAL CORD 6 [hp_X] in 30 mL SUS SCROFA ADRENAL GLAND (UNII: 398IYQ16YV) (SUS SCROFA ADRENAL GLAND - UNII:398IYQ16YV) SUS SCROFA ADRENAL GLAND 6 [hp_X] in 30 mL NADIDE (UNII: 0U46U6E8UK) (NADIDE - UNII:0U46U6E8UK) NADIDE 3 [hp_X] in 30 mL SODIUM DIETHYL OXALACETATE (UNII: 6CA025Y4FG) (DIETHYL OXALACETATE - UNII:15S56468G7) SODIUM DIETHYL OXALACETATE 3 [hp_X] in 30 mL SUS SCROFA PLACENTA (UNII: C8CV8867O8) (SUS SCROFA PLACENTA - UNII:C8CV8867O8) SUS SCROFA PLACENTA 6 [hp_X] in 30 mL TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 6 [hp_X] in 30 mL SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 4 [hp_X] in 30 mL SUS SCROFA VEIN (UNII: 2510RH3I89) (SUS SCROFA VEIN - UNII:2510RH3I89) SUS SCROFA VEIN 6 [hp_X] in 30 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 9 mL in 30 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17089-463-18 1 in 1 BOX 09/24/2020 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/24/2020 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-463)