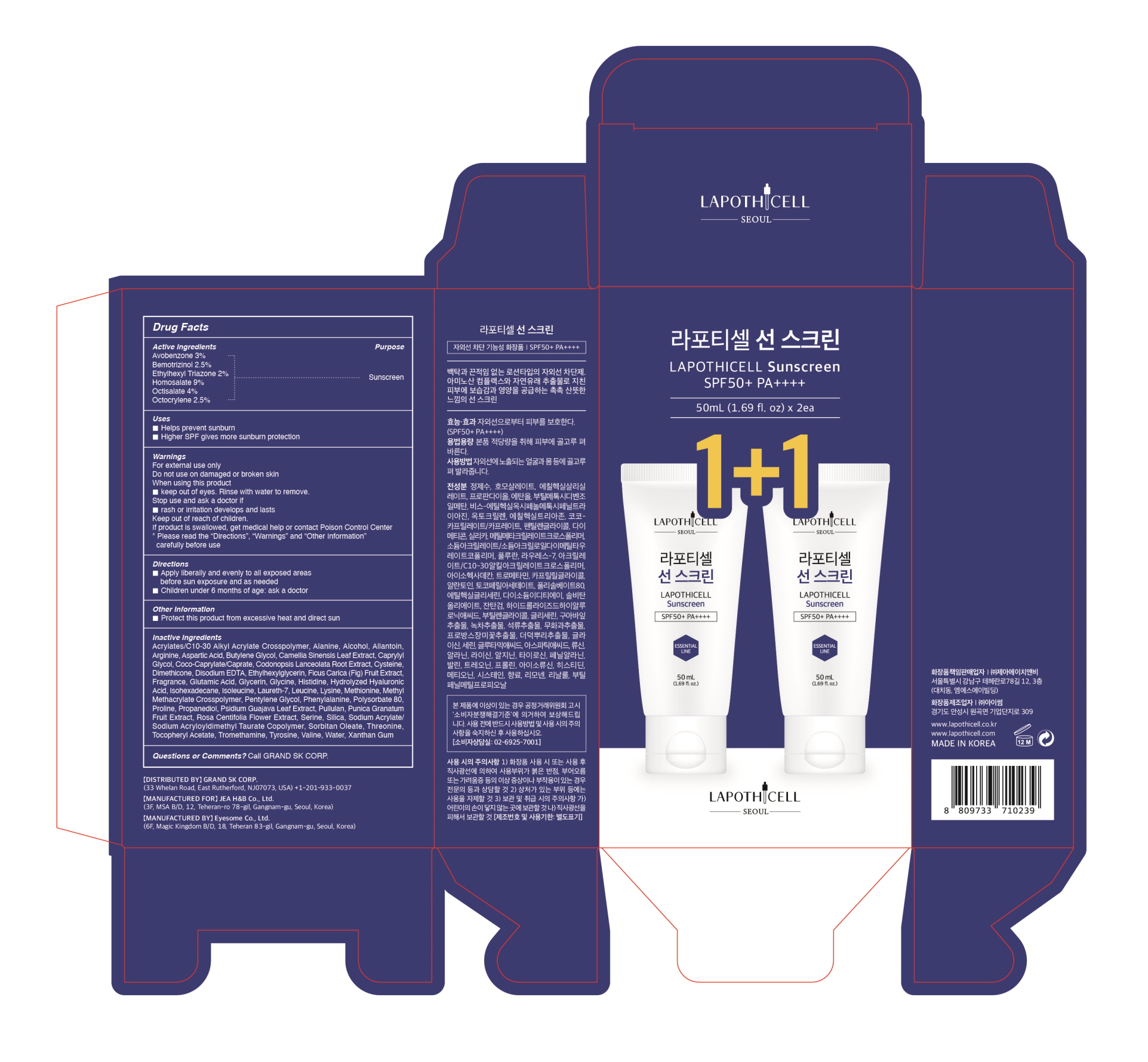
Label: LAPOTHICELL SUNSCREEN- avobenzone, bemotrizinol, ethylhexyl triazone, homosalate, octisalate, octocrylene cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 58832-0005-1, 58832-0005-2 - Packager: EYESOME. Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 25, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- DOSAGE & ADMINISTRATION
-
WARNINGS
For external use only
Do not use on damaged or broken skin
When using this product
*Keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if
*rash or irritation develops and lasts
Keep our of reach of children.
If product is swallowed, get medical help or contact Poison Control Center
*Please read the "Directions", "Warnings", and "Other Information" carefully before use
- Keep out of reach of children.
- INDICATIONS & USAGE
-
INACTIVE INGREDIENT
Water, Propanediol, Alcohol, Coco-Caprylate/Caprate, Pentylene Glycol, Dimethicone, Silica, Methyl Methacrylate Crosspolymer, Sodium Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Pullulan, Laureth-7, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Isohexadecane, Tromethamine, Caprylyl Glycol, Allantoin, Tocopheryl Acetate, Polysorbate 80, Ethylhexylglycerin, Disodium EDTA, Sorbitan Oleate, Xanthan Gum, Hydrolyzed Hyaluronic Acid, Butylene Glycol, Glycerin, Psidium Guajava Leaf Extract, Camellia Sinensis Leaf Extract, Punica Granatum Fruit Extract, Ficus Carica (Fig) Fruit Extract, Rosa Centifolia Flower Extract, Codonopsis Lanceolata Root Extract, Glycine, Serine, Glutamic Acid, Aspartic Acid, Leucine, Alanine, Lysine, Arginine, Tyrosine, Phenylalanine, Valine, Threonine, Proline, Isoleucine, Histidine, Methionine, Cysteine, Fragrance, Limonene, Linalool, Butylphenyl Methylpropional
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LAPOTHICELL SUNSCREEN
avobenzone, bemotrizinol, ethylhexyl triazone, homosalate, octisalate, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58832-0005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 4.5 g in 50 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 2 g in 50 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 1.5 g in 50 mL BEMOTRIZINOL (UNII: PWZ1720CBH) (BEMOTRIZINOL - UNII:PWZ1720CBH) BEMOTRIZINOL 1.75 g in 50 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 1.75 g in 50 mL ETHYLHEXYL TRIAZONE (UNII: XQN8R9SAK4) (ETHYLHEXYL TRIAZONE - UNII:XQN8R9SAK4) ETHYLHEXYL TRIAZONE 1 g in 50 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) DIMETHICONE (UNII: 92RU3N3Y1O) CAPRYLYL GLYCOL (UNII: 00YIU5438U) ALLANTOIN (UNII: 344S277G0Z) POLYSORBATE 80 (UNII: 6OZP39ZG8H) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58832-0005-2 2 in 1 CARTON 08/25/2020 1 NDC:58832-0005-1 50 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/20/2020 Labeler - EYESOME. Co.,Ltd. (557795360) Registrant - EYESOME. Co.,Ltd. (557795360) Establishment Name Address ID/FEI Business Operations EYESOME. Co.,Ltd. 557795360 manufacture(58832-0005)