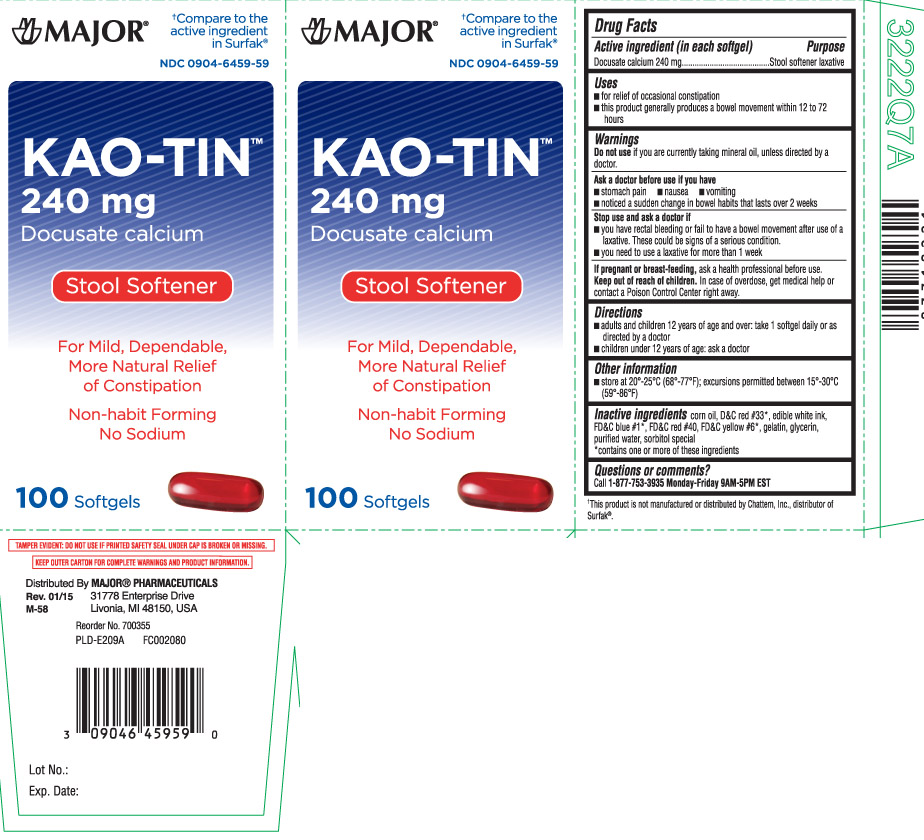
Label: KAO TIN- docusate calcium capsule, liquid filled
- NDC Code(s): 0904-6459-59
- Packager: Major Pharmaceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 27, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each softgel)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
†Compare to the active ingredient in Surfak®
Kao-Tin
240 mg
Docusate calcium
Stool Softener
For mild, dependable, more natural relief of constipation
Non-habit forming
No sodium
Softgels
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY DEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
Distributed by: MAJOR PHARMACEUTICALS
31778 Enterorise Drive
Livonia, MI 48150 USA
†This product is not manufactured or distributed by Chattem, Inc., distributor of Surfak®
- Product Label
-
INGREDIENTS AND APPEARANCE
KAO TIN
docusate calcium capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0904-6459 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE CALCIUM (UNII: 6K7YS503HC) (DOCUSATE - UNII:M7P27195AG) DOCUSATE CALCIUM 240 mg Inactive Ingredients Ingredient Name Strength CORN OIL (UNII: 8470G57WFM) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SORBITAN (UNII: 6O92ICV9RU) Product Characteristics Color red Score no score Shape CAPSULE Size 18mm Flavor Imprint Code P58;SCU Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0904-6459-59 1 in 1 BOX 02/28/2015 02/28/2025 1 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 02/28/2015 02/28/2025 Labeler - Major Pharmaceuticals (191427277)