Label: LIDOCAINE 4% CREAM- lidocaine cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 52565-122-07, 52565-122-15, 52565-122-30 - Packager: Teligent Pharma, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 22, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION
- INGREDIENTS
-
INACTIVE:
Butylated Hydroxytoluene, Cetostearyl Alcohol, Citric Acid, Edetate Disodium, Light Mineral Oil, Methylparaben, Polyoxyl 20 Cetostearyl Ether, Propylene Glycol, Propylparaben, Purified Water, Sodium Citrate and White Petrolatum.
The chemical designation of Lidocaine is 2-(diethylamino)-N(2,6-dimethylphenyl), acetamide. The structure of lidocaine is:
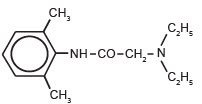
-
MECHANISM OF ACTION
Lidocaine 4% cream applied to intact skin provides dermal analgesia by the release of lidocaine from the cream into the epidermis and dermis. Lidocaine is a local anesthetic agent of the amide type. Local anesthetics reversibly block the initiation and conduction of nerve impulses by interfering with the flux of sodium ions through the neuronal membrane. The onset, depth and duration of dermal analgesia provided depend upon the site and duration of application.
-
INDICATIONS AND USAGE
Lidocaine 4% cream is indicated for use on normal intact skin for temporary relief of pain and itching due to minor cuts, minor scrapes, minor skin irritations, minor burns and insect bites.
Lidocaine 4% cream is not recommended for internal use, in the or near the eyes and in large quantities, particularly over raw surfaces or blistered areas. - CONTRAINDICATIONS
-
WARNINGS
For external use only. Avoid contact with the eyes. Do not use over large areas of the body. Do not use for more than seven days unless directed by a doctor. Keep this and all drugs out of the reach of children. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Inappropriate use of this product, such as on large areas of the body, application on mucous membranes, or on individuals that are allergic to the amide type anesthetics, may result in serious side effects. Consultation with a doctor before using this product is strongly recommended. -
PRECAUTIONS
Repeated doses of lidocaine 4% cream may increase blood levels of lidocaine. Avoid contact with the eyes. If eye contact occurs, immediately wash out the eye with water or saline.
The patient should be aware that dermal analgesia may be accompanied by the block of all sensations in the treated skin. For this reason, the patient should avoid inadvertent trauma to the treated area by scratching, rubbing or exposure to extreme hot or cold temperatures until complete sensation has returned. - DRUG INTERACTIONS
- DIRECTIONS
- SPL UNCLASSIFIED SECTION
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LIDOCAINE 4% CREAM
lidocaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52565-122 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 g Inactive Ingredients Ingredient Name Strength Butylated Hydroxytoluene (UNII: 1P9D0Z171K) Cetostearyl Alcohol (UNII: 2DMT128M1S) Citric Acid Acetate (UNII: DSO12WL7AU) Edetate Disodium Anhydrous (UNII: 8NLQ36F6MM) Light Mineral Oil (UNII: N6K5787QVP) Methylparaben (UNII: A2I8C7HI9T) Polyoxyl 20 Cetostearyl Ether (UNII: YRC528SWUY) Propylparaben (UNII: Z8IX2SC1OH) Water (UNII: 059QF0KO0R) Sodium Citrate, Unspecified Form (UNII: 1Q73Q2JULR) Petrolatum (UNII: 4T6H12BN9U) Propylene Glycol (UNII: 6DC9Q167V3) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52565-122-07 5 in 1 CARTON 01/22/2019 1 5 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:52565-122-15 1 in 1 CARTON 01/22/2019 2 15 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:52565-122-30 1 in 1 CARTON 01/22/2019 3 30 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/22/2019 Labeler - Teligent Pharma, Inc. (011036910) Establishment Name Address ID/FEI Business Operations Teligent, Inc. 011036910 manufacture(52565-122)


