Label: panretin- alitretinoin gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 64365-501-01 - Packager: Ligand Pharmaceuticals Incorporated
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 6, 2006
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- N/A - Section Title Not Found In Database
-
Description
Panretin® gel 0.1% contains alitretinoin and is intended for topical application only. The chemical name is 9-cis-retinoic acid and the structural formula is as follows:
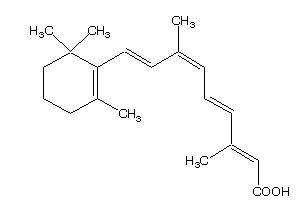
Chemically, alitretinoin is related to vitamin A. It is a yellow powder with a molecular weight of 300.44 and a molecular formula of C20H28O2. It is slightly soluble in ethanol (7.01 mg/g at 25oC) and insoluble in water. Panretin® gel is a clear, yellow gel containing 0.1% (w/w) alitretinoin in a base of dehydrated alcohol USP, polyethylene glycol 400 NF, hydroxypropyl cellulose NF, and butylated hydroxytoluene NF.
-
CLINICAL PHARMACOLOGY
Mechanism of Action
Alitretinoin (9-cis-retinoic acid) is a naturally-occurring endogenous retinoid that binds to and activates all known intracellular retinoid receptor subtypes (RARα, RARβ, RARγ, RXRα, RXRβ and RXRγ). Once activated these receptors function as transcription factors that regulate the expression of genes that control the process of cellular differentiation and proliferation in both normal and neoplastic cells. Alitretinoin inhibits the growth of Kaposi’s sarcoma (KS) cells in vitro.
Pharmacokinetics
No studies have examined plasma 9-cis-retinoic acid concentrations before and after treatment with Panretin® gel. There is, however, indirect evidence that absorption is not extensive. Plasma concentrations of 9-cis-retinoic acid were evaluated during clinical studies in patients with cutaneous lesions of AIDS-related KS after repeated multiple-daily dose application of Panretin® gel for up to 60 weeks. The range of 9-cis-retinoic acid plasma concentrations in these patients was similar to the range of circulating, naturally-occurring 9-cis-retinoic acid plasma concentrations in untreated healthy volunteers.
Although there are no detectable plasma concentrations of 9-cis-retinoic acid metabolites after topical application of Panretin® gel, in vitro studies indicate that the drug is metabolized to 4-hydroxy-9-cis-retinoic acid and 4-oxo-9-cis-retinoic acid by CYP 2C9, 3A4, 1A1, and 1A2 enzymes. In vivo, 4-oxo-9-cis-retinoic acid is the major circulating metabolite following oral administration of 9-cis-retinoic acid.
No formal pharmacokinetic drug interaction studies between Panretin® gel and antiretroviral agents have been conducted.
Clinical Studies
Panretin® gel is not a systemic therapy; it therefore cannot treat visceral Kaposi’s sarcoma (KS) nor prevent the development of new KS lesions where it has not been applied. Visceral KS disease was not monitored in these trials, and the appearance of new KS lesions was not considered part of the response assessment in clinical trials.
Panretin® gel was evaluated in two multicenter, prospective, randomized, double-blind, vehicle-controlled studies in patients with cutaneous lesions of AIDS-related KS. In both studies the primary efficacy endpoint was the patients’ cutaneous KS tumor response rate through 12 weeks of study drug treatment which was assessed by evaluating from 3 to 8 KS index lesions according to the modified AIDS Clinical Trials Group (ACTG) response criteria as applied to topical therapy (i.e., evaluation of height and area reductions of the index lesions only; progressive disease in non-index lesions and new lesions were not considered progressive disease; progressive disease was scored only in the treated index lesions). A global evaluation by physicians was also carried out. It considered all of the patient’s treated lesions (index and other) compared to baseline. In this evaluation, patients with at least a 50% improvement in the KS lesions were considered responders. In addition, photographs of lesions in patients considered responders by the modified ACTG criteria were examined by the FDA for a cosmetically beneficial response, defined as at least a 50% improvement in appearance compared to baseline, considering both the KS lesions and dermal toxicity at the lesion site, in at least 50% of the index lesions and maintained for at least 3 weeks. Patients were also asked about their satisfaction with the treatment.
In Study 1, a total of 268 patients were entered from centers in the U.S. and Canada. Patients were treated topically three to four times a day with either Panretin® gel or a matching vehicle gel for a minimum of 12 weeks, followed by an open-label phase in patients who had not yet progressed on Panretin® gel. Responses during the double-blind phase are shown in Table 1. Responses to Panretin® gel were seen in both previously untreated patients and in patients with prior systemic and/or topical KS treatment. A total of 72 patients responded to Panretin® gel during the randomized or crossover portions of the study. At a median duration of monitoring of 16 weeks, only 15% of the 72 patients had relapsed. Panretin® gel would not be expected to affect development of new lesions in untreated areas and these were seen in about 50% of patients, at similar rates in treated and untreated patients, responders and non-responders. The patients’ assessment of their overall satisfaction with the drug effect on all treated lesions significantly favored Panretin® gel.
Study 2 was an international study with a planned enrollment of 270 patients. Patients were treated topically twice a day with Panretin® gel or a matching vehicle for 12 weeks. The study was stopped early because of positive interim results in the initial 82 patient data set. Results of the study are shown in Table 1. Responses to Panretin® gel were seen both in previously untreated patients and in patients with prior systemic and/or topical KS treatment.
TABLE 1: Summary of Tumor Responses STUDY 1 STUDY 2 Panretin®
N=134
Gel
Vehicle
N=134
GelPanretin®
N=36
Gel
Vehicle
N=46
Gel
Modified ACTG Response(index lesions)
1% CR
34% PR
p=0.0012
16% PR
36% PR
7% PR
Physician's Global/Subjective Assessment(all treated lesions)
19% PR
p=0.00014
4% PR
47% PR
11% PR
Beneficial Response Photographs(index lesions only)
15%
p=0.0026
4%
19%
2%In the clinical trials, responses were seen as early as two (2) weeks; most patients, however, required four (4) to eight (8) weeks of treatment, and some patients did not experience significant improvement until 14 or more weeks of treatment. The cumulative percentage of patients who achieved a response was less than 1% at 2 weeks, 10% at 4 weeks, and 28% at 8 weeks.
In both studies, responses occurred in patients with a wide range of baseline CD4+ lymphocyte counts, including patients with CD4+ lymphocyte counts less than 50 cells/mm3. Nearly all patients received concomitant combination antiretroviral therapy.
Photographs of patients revealed a substantial erythematous and edematous response in some cases, leading to a cosmetically mixed outcome even in apparent responders. Nonetheless, in Study 1 it appeared that a cosmetically satisfactory result occurred at about the same rate as the Physician’s Global response rate and in both studies such a response was more frequent than in the vehicle control.
-
INDICATIONS AND USAGE
Panretin® gel is indicated for topical treatment of cutaneous lesions in patients with AIDS-related Kaposi’s sarcoma. Panretin® gel is not indicated when systemic anti-KS therapy is required (e.g., more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement). There is no experience to date using Panretin® gel with systemic anti-KS treatment.
- CONTRAINDICATIONS
-
WARNINGS
Pregnancy: Panretin® gel could cause fetal harm if significant absorption were to occur in a pregnant woman. 9-cis-Retinoic acid has been shown to be teratogenic in rabbits and mice. An increased incidence of fused sternebrae and limb and craniofacial defects occurred in rabbits given oral doses of 0.5 mg/kg/day (about five times the estimated daily human topical dose on a mg/m2 basis, assuming complete systemic absorption of 9-cis-retinoic acid, when Panretin® gel is administered as a 60 g tube over 1 month in a 60 kg human) during the period of organogenesis. Limb and craniofacial defects also occurred in mice given a single oral dose of 50 mg/kg on day eleven of gestation (about 127 times the estimated daily human topical dose on a mg/m2 basis). Oral 9-cis-retinoic acid was also embryocidal, as indicated by early resorptions and post-implantation loss when it was given during the period of organogenesis to rabbits at doses of 1.5 mg/kg/day (about 15 times the estimated daily human topical dose on a mg/m2 basis) and to rats at doses of 5 mg/kg/day (about 25 times the estimated daily human topical dose on a mg/m2 basis). Animal reproduction studies with topical 9-cis-retinoic acid have not been conducted. It is not known whether topical Panretin® gel can modulate endogenous 9-cis-retinoic acid levels in a pregnant woman nor whether systemic exposure is increased by application to ulcerated lesions or by duration of treatment. There are no adequate and well-controlled studies in pregnant women. If Panretin® gel is used during pregnancy, or if the patient becomes pregnant while taking it, the patient should be apprised of the potential hazard to the fetus. Women of child-bearing potential should be advised to avoid becoming pregnant.
-
PRECAUTIONS
Panretin® gel is indicated for topical treatment of Kaposi’s sarcoma. Patients with cutaneous T-cell lymphoma were less tolerant of topical Panretin® gel; five of seven patients had 6 episodes of treatment-limiting toxicities—grade 3 dermal irritation—with Panretin® gel (0.01% or 0.05%).
Photosensitivity
Retinoids as a class have been associated with photosensitivity. There were no reports of photosensitivity associated with the use of Panretin® gel in the clinical studies. Nonetheless, because in vitro data indicate that 9-cis-retinoic acid may have a weak photosensitizing effect, patients should be advised to minimize exposure of treated areas to sunlight and sunlamps during the use of Panretin® gel.
Drug Interactions
Patients who are applying Panretin® gel should not concurrently use products that contain DEET (N,N-diethyl-m-toluamide), a common component of insect repellent products. Animal toxicology studies showed increased DEET toxicity when DEET was included as part of the formulation.
Although there was no clinical evidence in the vehicle-controlled studies of drug interactions with systemic antiretroviral agents, including protease inhibitors, macrolide antibiotics, and azole antifungals, the effect of Panretin® gel on the steady-state concentrations of these drugs is not known. No drug interaction data are available on concomitant administration of Panretin® gel and systemic anti-KS agents.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to assess the carcinogenic potential of 9-cis-retinoic acid have not been conducted. 9-cis-Retinoic acid was not mutagenic in vitro (bacterial assays, Chinese hamster ovary cell HGPRT mutation assay) and was not clastogenic in vitro (chromosome aberration test in human lymphocytes) nor in vivo (mouse micronucleus test).
-
ADVERSE REACTIONS
The safety of Panretin® gel has been assessed in clinical studies of 385 patients with AIDS-related KS. Adverse events associated with the use of Panretin® gel in patients with AIDS-related KS occurred almost exclusively at the site of application. The dermal toxicity begins as erythema; with continued application of Panretin® gel, erythema may increase and edema may develop. Dermal toxicity may become treatment-limiting, with intense erythema, edema, and vesiculation. Usually, however, adverse events are mild to moderate in severity; they led to withdrawal from the study in only 7% of the patients. Severe local (application site) skin adverse events occurred in about 10% of patients in the U.S. study (versus 0% in the vehicle control). Table 2 lists the adverse events that occurred at the application site with an incidence of at least 5% during the double-blind phase in the Panretin® gel-treated group and in the vehicle control group in either of the two controlled studies. Adverse events were reported at other sites but generally were similar in the two groups.
TABLE 2: Adverse Events with an Incidence of at Least 5% at the Application Site in Either Controlled Study in Patients Receiving Panretin® Gel or Vehicle Control Includes Investigator terms:
1Erythema, scaling, irritation, redness, rash, dermatitis
2Burning, pain
3Itching, pruritus
4Flaking, peeling, desquamation, exfoliation
5Excoriation, cracking, scab, crusting, drainage, eschar, fissure or oozing
6Stinging, tingling
7Edema, swelling, inflammation
Adverse Event Term Study 1 Study 2 Panretin® Gel
N=134 Pts.
%
Vehicle Gel
N=134 Pts.
%
Panretin® Gel
N=36 Pts.
%
Vehicle Gel
N=46 Pts.
%
Rash1 77 11 25 4 Pain2 34 7 0 4 Pruritus3 11 4 8 4 Exfoliative dermatitis4 9 2 3 0 Skin disorder5 8 1 0 0 Paresthesia6 3 0 22 7 Edema7 8 3 3 0 -
OVERDOSAGE
There has been no experience with acute overdose of Panretin® gel in humans. Systemic toxicity following acute overdosage with topical application of Panretin® gel is unlikely because of limited systemic plasma levels observed with normal therapeutic doses. There is no specific antidote for overdosage.
-
DOSAGE AND ADMINISTRATION
Panretin® gel should initially be applied two (2) times a day to cutaneous KS lesions. The application frequency can be gradually increased to three (3) or four (4) times a day according to individual lesion tolerance. If application site toxicity occurs, the application frequency can be reduced. Should severe irritation occur, application of drug can be temporarily discontinued for a few days until the symptoms subside.
Sufficient gel should be applied to cover the lesion with a generous coating. The gel should be allowed to dry for three to five minutes before covering with clothing. Because unaffected skin may become irritated, application of the gel to normal skin surrounding the lesions should be avoided. In addition, do not apply the gel on or near mucosal surfaces of the body.
A response of KS lesions may be seen as soon as two weeks after initiation of therapy but most patients require longer application. With continued application, further benefit may be attained. Some patients have required over 14 weeks to respond. In clinical trials, Panretin® gel was applied for up to 96 weeks. Panretin® gel should be continued as long as the patient is deriving benefit.
Occlusive dressings should not be used with Panretin® gel.
-
How Supplied
Panretin® gel is available in tubes containing 60 grams. Store at 25° C (77° F); excursions permitted to 15-30° C (59-86° F) [see USP Controlled Room Temperature].
Manufactured for: Ligand Pharmaceuticals Incorporated
San Diego, CA 92121
by: Bristol-Myers Squibb Company
Princeton, NJ 08543 USANDC 64365-501-01
Ligand Part #3000153 (Rev. 1001)
-
INGREDIENTS AND APPEARANCE
PANRETIN
alitretinoin gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64365-501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength alitretinoin (UNII: 1UA8E65KDZ) (alitretinoin - UNII:1UA8E65KDZ) 1 mg in 1 g Inactive Ingredients Ingredient Name Strength Dehydrated alcohol () Polyethylene Glycol 400 () Hydroxypropyl Cellulose () Butylated Hydroxytoluene () Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64365-501-01 60 g in 1 TUBE Labeler - Ligand Pharmaceuticals Incorporated
