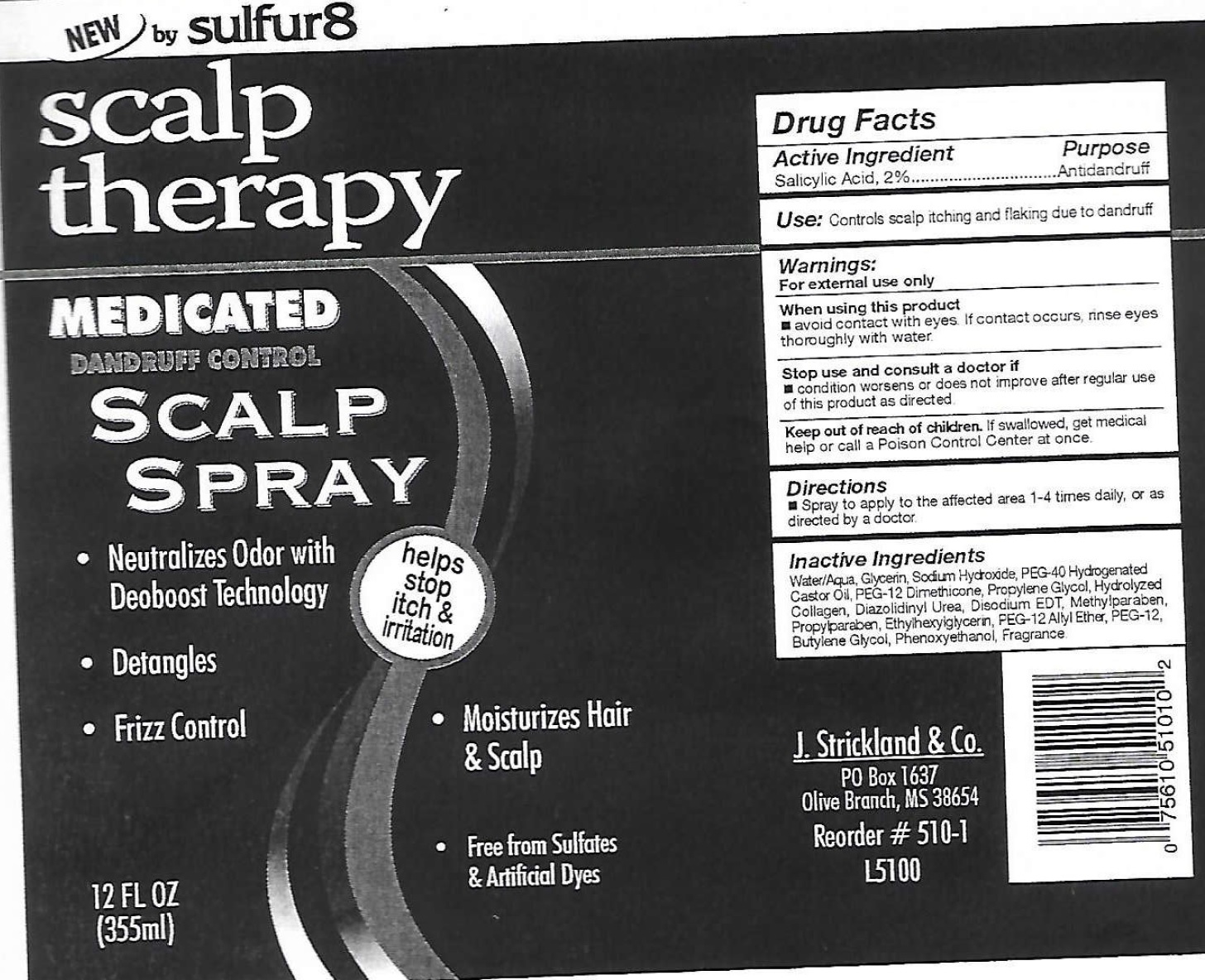
Label: SULFUR 8 SCALP THERAPY MEDICATED DANDRUFF CONTROL- salicylic acid spray
- NDC Code(s): 12022-032-00
- Packager: J. Strickland and Co.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Use:
- Warnings
- Directions
- Inactive Ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
SULFUR 8 SCALP THERAPY MEDICATED DANDRUFF CONTROL
salicylic acid sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:12022-032 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) SODIUM HYDROXIDE (UNII: 55X04QC32I) PEG-12 DIMETHICONE (300 CST) (UNII: ZEL54N6W95) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) POLYETHYLENE GLYCOL 600 (UNII: NL4J9F21N9) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:12022-032-00 355 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 01/01/2020 Labeler - J. Strickland and Co. (007023112)