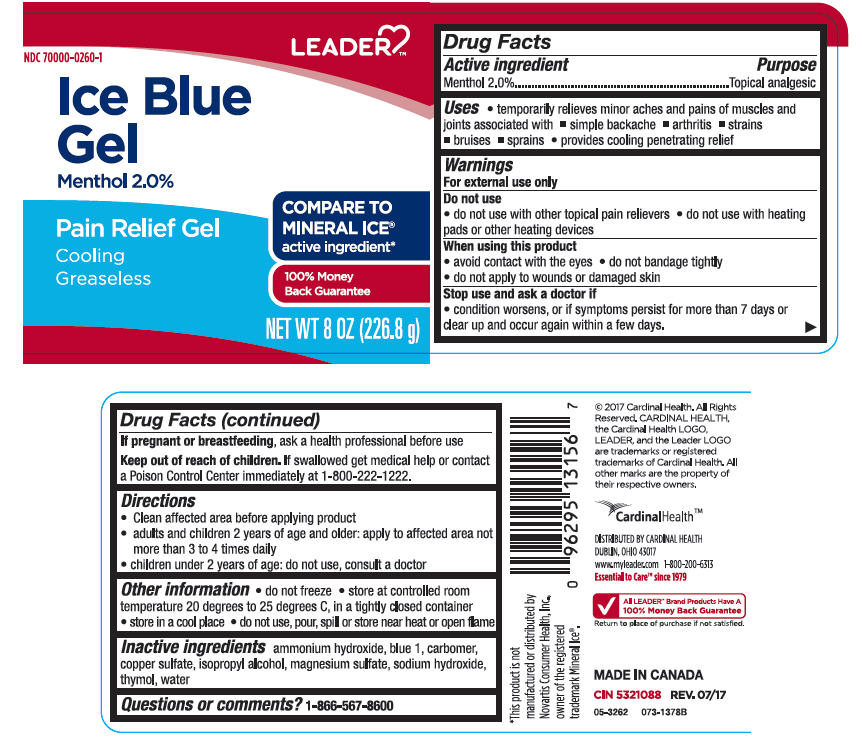
Label: LEADER ICE BLUE ANALGESIC- menthol, unspecified form gel
- NDC Code(s): 70000-0260-1
- Packager: Cardinal Health, 110 dba LEADER
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- ♦
- do not use with other topical pain relievers
- ♦
- do not use with heating pads or other heating devices
When using this product
- ♦
- avoid contact with the eyes
- ♦
- do not bandage tightly
- ♦
- do not apply to wounds or damaged skin
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 226.8 g Jar Label
-
INGREDIENTS AND APPEARANCE
LEADER ICE BLUE ANALGESIC
menthol, unspecified form gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0260 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Menthol, Unspecified Form (UNII: L7T10EIP3A) (Menthol, Unspecified Form - UNII:L7T10EIP3A) Menthol, Unspecified Form 20 mg in 1 g Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) DISODIUM N-ETHYL-N-4-((4-(ETHYL((3-SULFOPHENYL)METHYL)AMINO)PHENYL)(2-SULFOPHENYL)METHYLENE)-2,5-CYCLOHEXADIEN-1-YLIDENE)-3-SULFOBENZENEMETHANAMINIUM (UNII: PPQ093M8HR) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) CUPRIC SULFATE ANHYDROUS (UNII: KUW2Q3U1VV) ISOPROPYL ALCOHOL (UNII: ND2M416302) MAGNESIUM SULFATE ANHYDROUS (UNII: ML30MJ2U7I) SODIUM HYDROXIDE (UNII: 55X04QC32I) THYMOL (UNII: 3J50XA376E) WATER (UNII: 059QF0KO0R) Product Characteristics Color BLUE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0260-1 226.8 g in 1 JAR; Type 0: Not a Combination Product 05/31/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M017 05/31/2017 Labeler - Cardinal Health, 110 dba LEADER (063997360) Registrant - Garcoa, Inc. (036464697) Establishment Name Address ID/FEI Business Operations Garcoa, Inc. 036464697 MANUFACTURE(70000-0260)