Label: RITE AID MAXIMUM STRENGTH- benzocaine gel, dentifrice
-
Contains inactivated NDC Code(s)
NDC Code(s): 11822-0317-2, 11822-0317-9 - Packager: Rite Aid
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 9, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
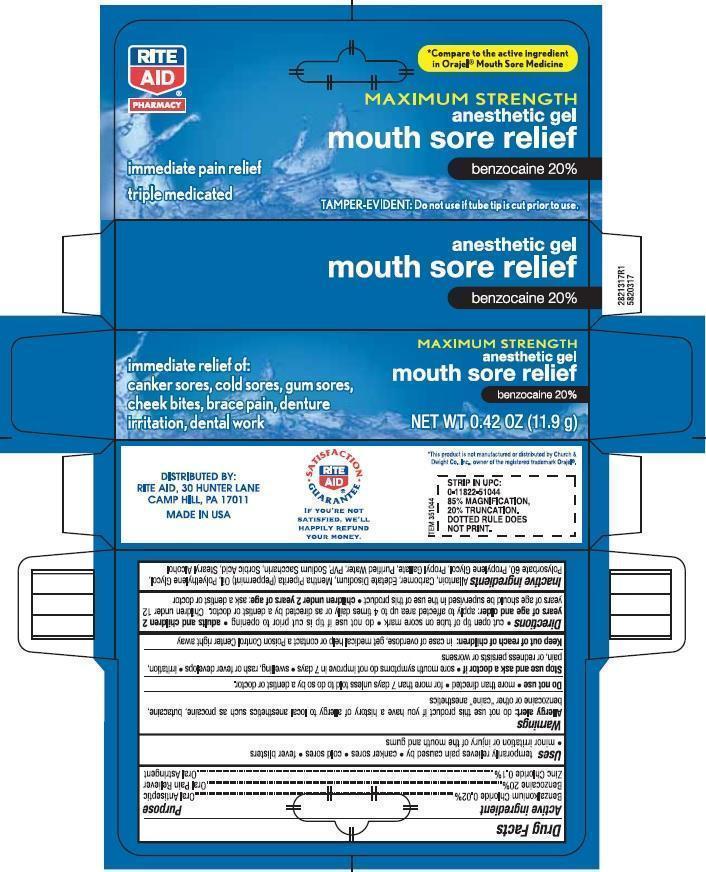
Active ingredient
Benzalkonium Chloride 0.02% .................................................... Oral Antiseptic
Benzocaine 7.5% ...................................................................... Oral Pain Reliever
Zinc Chloride 0.1% ................................................................... Oral Astringent
- Uses
-
WARNINGS
Allergy alert: do not use this product if you have a history of allergy to local anesthetics such as
procaine, butacaine, benzocaine or other “caine” anesthetics- Do not use more than directed for more than 7 days unless told to do so by a dentist or doctor.
- Stop use and ask a doctor if sore mouth symptoms do not get better in 7 days. swelling, rash or fever develops, irritation, pain or redness persists or worsens
-
Directions
cut open tip of tube on score mark. do not use if tip is cut prior to opening
Adults and children 2 years of age and older: apply to affected area up to 4 times daily or as directed by a doctor. Children under 12 years should be supervised in the use of this product.
Childern under 2 years of age: ask a dentist or doctor
- DOSAGE & ADMINISTRATION
- Other Information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RITE AID MAXIMUM STRENGTH
benzocaine gel, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11822-0317 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 20.0 g in 100 g BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.04 g in 100 g ZINC CHLORIDE (UNII: 86Q357L16B) (ZINC CATION - UNII:13S1S8SF37) ZINC 0.1 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SORBIC ACID (UNII: X045WJ989B) POLYSORBATE 60 (UNII: CAL22UVI4M) POVIDONE K90 (UNII: RDH86HJV5Z) CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) EDETATE DISODIUM (UNII: 7FLD91C86K) ALLANTOIN (UNII: 344S277G0Z) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYL GALLATE (UNII: 8D4SNN7V92) PEPPERMINT OIL (UNII: AV092KU4JH) Product Characteristics Color white (White / Clear to slight pinkish orange) Score Shape Size Flavor PEPPERMINT (Peppermint) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11822-0317-9 1 in 1 CARTON 1 NDC:11822-0317-2 11.9 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 01/20/2011 Labeler - Rite Aid (014578892) Registrant - Lornamead (126440440) Establishment Name Address ID/FEI Business Operations Lornamead 126440440 manufacture(11822-0317) , pack(11822-0317)