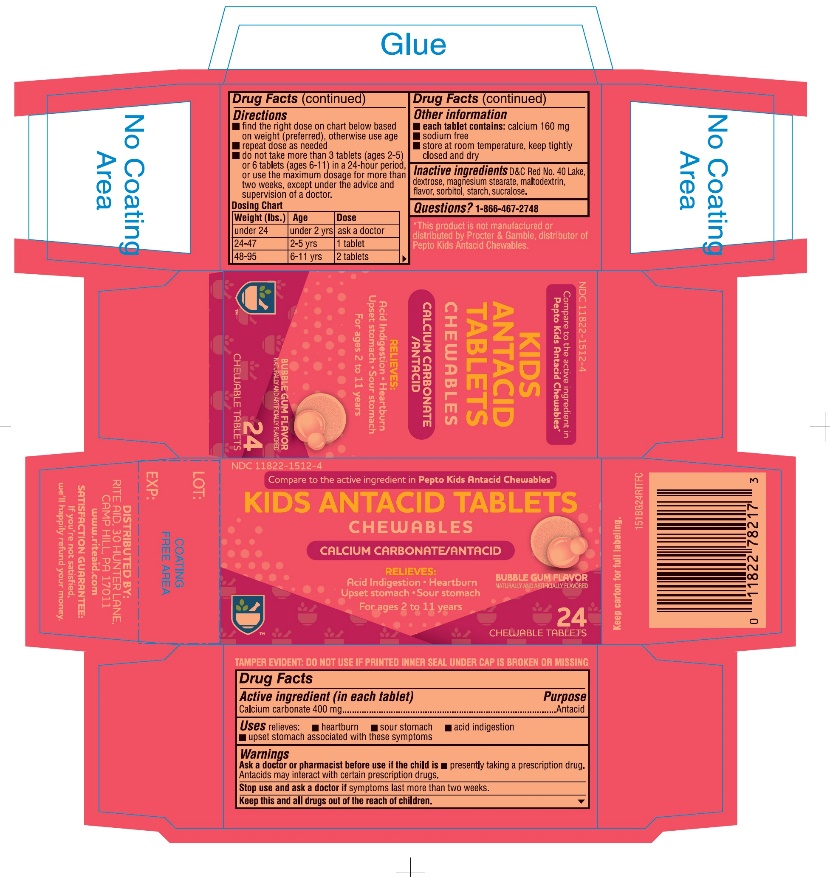
Label: KIDS ANTACID- calcium carbonate tablet, chewable
- NDC Code(s): 11822-1512-4
- Packager: RITE AID CORPORATION
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each tablet)
- Purpose
- Uses
- Warnings
-
Directions
- ▪
- find the right dose on chart below based on weight (preferred), otherwise use age
- ▪
- repeat dose as needed
- ▪
- do not take more than 3 tablets (ages 2-5) or 6 tablets (ages 6-11) in a 24-hour period, or use the maximum dosage for more than two weeks, except under the advice and supervision of a doctor.
Dosing Chart
Weight (lbs.) Age Dose under 24
under 2 yrs
ask a doctor
24-47
2-5 yrs
1 tablet
48-95
6-11 yrs
2 tablets
- Other information
- Inactive ingredients
- Questions?
- Package Label Principal Display Panel - 24 Tablet Carton
-
INGREDIENTS AND APPEARANCE
KIDS ANTACID
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11822-1512 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 400 mg Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) SORBITOL (UNII: 506T60A25R) STARCH, CORN (UNII: O8232NY3SJ) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color PINK Score no score Shape ROUND Size 16mm Flavor BUBBLE GUM Imprint Code RP151 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11822-1512-4 1 in 1 CARTON 11/04/2019 1 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 11/04/2019 Labeler - RITE AID CORPORATION (014578892)