Label: PAROXETINE- paroxetine hydrochloride hemihydrate tablet, film coated
-
NDC Code(s):
0378-7001-10,
0378-7001-93,
0378-7002-10,
0378-7002-93, view more0378-7003-10, 0378-7003-93, 0378-7004-10, 0378-7004-93
- Packager: Mylan Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 27, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use PAROXETINE TABLETS safely and effectively. See full prescribing information for PAROXETINE TABLETS.
PAROXETINE tablets, for oral use
Initial U.S. Approval: 1992WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
Increased risk of suicidal thoughts and behavior in pediatric and young adult patients taking antidepressants. Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors. Paroxetine tablets are not approved for use in pediatric patients. (5.1, 8.4)
INDICATIONS AND USAGE
Paroxetine tablets are a selective serotonin reuptake inhibitor (SSRI) indicated in adults for the treatment of (1):
- •
- Major Depressive Disorder (MDD)
- •
- Obsessive Compulsive Disorder (OCD)
- •
- Panic Disorder (PD)
- •
- Social Anxiety Disorder (SAD)
- •
- Generalized Anxiety Disorder (GAD)
- •
- Posttraumatic Stress Disorder (PTSD)
DOSAGE AND ADMINISTRATION
- •
- Recommended starting and maximum daily dosage for MDD, OCD, PD, and PTSD: (2.2)
Indication
Starting Daily Dose
Maximum Daily Dose
MDD
20 mg
50 mg
OCD
20 mg
60 mg
PD
10 mg
60 mg
PTSD
20 mg
50 mg
- •
- Recommended starting dosage for SAD and GAD is 20 mg daily. (2.3)
- •
- Elderly patients, patients with severe renal impairment or severe hepatic impairment: Starting dosage is 10 mg daily. Maximum dosage is 40 mg daily. (2.4)
- •
- When discontinuing paroxetine tablets, reduce dosage gradually. (2.6, 5.7)
DOSAGE FORMS AND STRENGTHS
- •
- Tablets: 10 mg, scored; 20 mg, scored; 30 mg; and 40 mg tablets. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- •
- Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents, but also when taken alone. If occurs, discontinue paroxetine tablets and serotonergic agents and initiate supportive measures. (5.2)
- •
- Embryofetal and Neonatal Toxicity: Can cause fetal and neonatal harm. Increased risk of cardiovascular malformations with exposure during the first trimester. Exposure in late pregnancy may lead to an increased risk for persistent pulmonary hypertension of the newborn. (5.4, 8.1)
- •
- Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, other antiplatelet drugs, warfarin, and other anticoagulant drugs may increase risk. (5.5)
- •
- Activation of Mania/Hypomania: Screen patients for bipolar disorder. (5.6)
- •
- Seizures: Use with caution in patients with seizure disorders. (5.8)
- •
- Angle-Closure Glaucoma: Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles treated with antidepressants. (5.9)
- •
- Sexual Dysfunction: Paroxetine tablets may cause symptoms of sexual dysfunction. (5.13)
ADVERSE REACTIONS
Most common adverse reactions (≥ 5% and at least twice placebo) are abnormal ejaculation, asthenia, constipation, decreased appetite, diarrhea, dizziness, dry mouth, female genital disorder, impotence, infection, insomnia, libido decreased, male genital disorder, nausea, nervousness, somnolence, sweating, tremor, yawn. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Drugs Highly Bound to Plasma Protein: Monitor for adverse reactions and reduce dosage of paroxetine tablets or other protein-bound drugs (e.g., warfarin) as warranted. (7)
- •
- Drugs Metabolized by CYP2D6: Reduce dosage of drugs metabolized by CYP2D6 as warranted. (7)
- •
- Concomitant Use with Tamoxifen: Consider use of an alternative antidepressant with little or no CYP2D6 inhibition. (5.11, 7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 2/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Administration Information
2.2 Recommended Dosage for MDD, OCD, PD, and PTSD
2.3 Recommended Dosage for SAD and GAD
2.4 Screen for Bipolar Disorder Prior to Starting Paroxetine Tablets
2.5 Recommended Dosage for Elderly Patients, Patients with Severe Renal Impairment, and Patients with Severe Hepatic Impairment
2.6 Switching Patients to or From a Monoamine Oxidase Inhibitor (MAOI)
2.7 Discontinuation of Treatment With Paroxetine Tablets
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
5.2 Serotonin Syndrome
5.3 Drug Interactions Leading to QT Prolongation
5.4 Embryofetal and Neonatal Toxicity
5.5 Increased Risk of Bleeding
5.6 Activation of Mania or Hypomania
5.7 Discontinuation Syndrome
5.8 Seizures
5.9 Angle-Closure Glaucoma
5.10 Hyponatremia
5.11 Reduction of Efficacy of Tamoxifen
5.12 Bone Fracture
5.13 Sexual Dysfunction
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal and Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Major Depressive Disorder
14.2 Obsessive Compulsive Disorder
14.3 Panic Disorder
14.4 Social Anxiety Disorder
14.5 Generalized Anxiety Disorder
14.6 Posttraumatic Stress Disorder
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]. Paroxetine tablets are not approved for use in pediatric patients [see Use in Specific Populations (8.4)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Administration Information
Administer paroxetine tablets as a single daily dose in the morning, with or without food.
2.2 Recommended Dosage for MDD, OCD, PD, and PTSD
The recommended starting dosages and maximum dosages of paroxetine tablets in patients with MDD, OCD, PD, and PTSD are presented in Table 1.
In patients with an inadequate response, increase dosage in increments of 10 mg per day at intervals of at least 1 week, depending on tolerability.
Table 1: Recommended Daily Dosage of Paroxetine Tablets in Patients with MDD, OCD, PD, and PTSD Indication
Starting Dose
Maximum Dose
MDD
20 mg
50 mg
OCD
20 mg
60 mg
PD
10 mg
60 mg
PTSD
20 mg
50 mg
2.3 Recommended Dosage for SAD and GAD
SAD
The starting and recommended dosage in patients with SAD is 20 mg daily. In clinical trials the effectiveness of paroxetine tablets was demonstrated in patients dosed in a range of 20 mg to 60 mg daily. While the safety of paroxetine tablets has been evaluated in patients with SAD at doses up to 60 mg daily, available information does not suggest any additional benefit for doses above 20 mg daily [see Clinical Studies (14.4)].
GAD
The starting and recommended dosage in patients with GAD is 20 mg daily. In clinical trials the effectiveness of paroxetine tablets in GAD was demonstrated in patients dosed in a range of 20 mg to 50 mg daily. There is not sufficient evidence to suggest a greater benefit to doses higher than 20 mg daily [see Clinical Studies (14.5)].
In patients with an inadequate response, increase dosage in increments of 10 mg per day at intervals of at least 1 week, depending on tolerability.
2.4 Screen for Bipolar Disorder Prior to Starting Paroxetine Tablets
Prior to initiating treatment with paroxetine tablets or another antidepressant, screen patients for a personal or family history of bipolar disorder, mania, or hypomania [see Warnings and Precautions (5.6)].
2.5 Recommended Dosage for Elderly Patients, Patients with Severe Renal Impairment, and Patients with Severe Hepatic Impairment
The recommended initial dosage is 10 mg per day for elderly patients, patients with severe renal impairment, and patients with severe hepatic impairment. Dosage should not exceed 40 mg/day.
2.6 Switching Patients to or From a Monoamine Oxidase Inhibitor (MAOI)
At least 14 days must elapse between discontinuation of a monoamine oxidase inhibitor (MAOI and initiation of paroxetine tablets. In addition, at least 14 days must elapse after stopping paroxetine tablets before starting an MAOI antidepressant [see Contraindications (4), Warnings and Precautions (5.2)].
2.7 Discontinuation of Treatment With Paroxetine Tablets
Adverse reactions may occur upon discontinuation of paroxetine tablets [see Warnings and Precautions (5.7)]. Gradually reduce the dosage rather than stopping paroxetine tablets abruptly whenever possible.
-
3 DOSAGE FORMS AND STRENGTHS
Paroxetine Tablets, USP are available containing paroxetine hydrochloride, USP (hemihydrate) equivalent to 10 mg, 20 mg, 30 mg or 40 mg of paroxetine.
- •
- The 10 mg tablets are blue, film-coated, modified capsule-shaped, scored tablets debossed with M on one side of the tablet and N to the left of the score and 1 to the right of the score on the other side.
- •
- The 20 mg tablets are blue, film-coated, modified capsule-shaped, scored tablets debossed with M on one side of the tablet and N to the left of the score and 2 to the right of the score on the other side.
- •
- The 30 mg tablets are blue, film-coated, round, unscored tablets debossed with M over N3 on one side of the tablet and blank on the other side.
- •
- The 40 mg tablets are blue, film-coated, round, unscored tablets debossed with M over N4 on one side of the tablet and blank on the other side.
-
4 CONTRAINDICATIONS
Paroxetine tablets are contraindicated in patients:
- •
- Taking, or within 14 days of stopping, MAOIs (including the MAOIs linezolid and intravenous methylene blue) because of an increased risk of serotonin syndrome [see Warnings and Precautions (5.2), Drug Interactions (7)].
- •
- Taking thioridazine because of risk of QT prolongation [see Warnings and Precautions (5.3) and Drug Interactions (7)].
- •
- Taking pimozide because of risk of QT prolongation [see Warnings and Precautions (5.3), Drug Interactions (7)].
- •
- With known hypersensitivity (e.g., anaphylaxis, angioedema, Stevens-Johnson syndrome) to paroxetine or any of the inactive ingredients in paroxetine tablets [see Adverse Reactions (6.1), (6.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
Table 2: Risk Differences of the Number of Patients with Suicidal Thoughts and Behaviors in the Pooled Placebo-Controlled Trials of Antidepressants in Pediatric and Adult Patients Age Range
Drug-Placebo Difference in Number of Patients with Suicidal Thoughts and Behaviors per 1,000 Patients Treated
Increases Compared to Placebo
< 18 years old
14 additional cases
18-24 years old
5 additional cases
Decreases Compared to Placebo
25-64 years old
1 fewer case
≥ 65 years old
6 fewer cases
Paroxetine tablets are not approved for use in pediatric patients.
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing paroxetine tablets, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
5.2 Serotonin Syndrome
SSRIs, including paroxetine tablets, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, meperidine, methadone, tryptophan, buspirone, amphetamines and St. John’s Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [see Contraindications (4), Drug Interactions (7.1)]. Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of paroxetine tablets with MAOIs is contraindicated. In addition, do not initiate paroxetine tablets in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection) or at lower doses. If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking paroxetine tablets discontinue paroxetine tablets before initiating treatment with the MAOI [see Contraindications (4), Drug Interactions (7)].
Monitor all patients taking paroxetine tablets for the emergence of serotonin syndrome. Discontinue treatment with paroxetine tablets and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of paroxetine tablets with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
5.3 Drug Interactions Leading to QT Prolongation
The CYP2D6 inhibitory properties of paroxetine can elevate plasma levels of thioridazine and pimozide. Since thioridazine and pimozide given alone produce prolongation of the QTc interval and increase the risk of serious ventricular arrhythmias, the use of paroxetine tablets is contraindicated in combination with thioridazine and pimozide [see Contraindications (4), Drug Interactions (7),Clinical Pharmacology (12.3)].
5.4 Embryofetal and Neonatal Toxicity
Paroxetine tablets can cause fetal harm when administered to a pregnant woman. Epidemiological studies have shown that infants exposed to paroxetine in the first trimester of pregnancy have an increased risk of cardiovascular malformations. Exposure to paroxetine in late pregnancy may lead to an increased risk for persistent pulmonary hypertension of the newborn (PPNH) and/or neonatal complications requiring prolonged hospitalization, respiratory support, and tube feeding.
If paroxetine tablets are used during pregnancy, or if the patient becomes pregnant while taking paroxetine tablets, the patient should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)].
5.5 Increased Risk of Bleeding
Drugs that interfere with serotonin reuptake inhibition, including paroxetine tablets, increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDS), other antiplatelet drugs, warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Based on data from the published observational studies, exposure to SSRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Use in Specific Populations (8.1)]. Bleeding events related to drugs that interfere with serotonin reuptake have ranged from ecchymoses, hematomas, epistaxis, and petechiae to life-threatening hemorrhages.
Inform patients about the increased risk of bleeding associated with the concomitant use of paroxetine tablets and antiplatelet agents or anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio.
5.6 Activation of Mania or Hypomania
In patients with bipolar disorder, treating a depressive episode with paroxetine tablets or another antidepressant may precipitate a mixed/manic episode. During controlled clinical trials of paroxetine tablets, hypomania or mania occurred in approximately 1% of paroxetine tablets-treated unipolar patients compared to 1.1% of active-control and 0.3% of placebo-treated unipolar patients. Prior to initiating treatment with paroxetine tablets, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
5.7 Discontinuation Syndrome
Adverse reactions after discontinuation of serotonergic antidepressants, particularly after abrupt discontinuation, include: nausea, sweating, dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), tremor, anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. A gradual reduction in dosage rather than abrupt cessation is recommended whenever possible [see Dosage and Administration (2.7)].
During clinical trials of GAD and PTSD, gradual decreases in the daily dose by 10 mg/day at weekly intervals followed by 1 week at 20 mg/day was used before treatment was discontinued. The following adverse reactions were reported at an incidence of 2% or greater for paroxetine tablets and were at least twice that reported for placebo: Abnormal dreams, paresthesia, and dizziness. Adverse reactions have been reported upon discontinuation of treatment with paroxetine tablets in pediatric patients. The safety and effectiveness of paroxetine tablets in pediatric patients have not been established [see Boxed Warning, Warnings and Precautions (5.1), Use in Specific Populations (8.4)].
5.8 Seizures
Paroxetine tablets and oral suspension have not been systematically evaluated in patients with seizure disorders. Patients with history of seizures were excluded from clinical studies. During clinical studies, seizures occurred in 0.1% of patients treated with paroxetine tablets. Paroxetine tablets should be prescribed with caution in patients with a seizure disorder. Discontinue paroxetine tablets in any patient who develops seizures.
5.9 Angle-Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs including paroxetine tablets may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy. Cases of angle-closure glaucoma associated with use of paroxetine tablets have been reported. Avoid use of antidepressants, including paroxetine tablets in patients with untreated anatomically narrow angles.
5.10 Hyponatremia
Hyponatremia may occur as a result of treatment with SSRIs, including paroxetine tablets. Cases with serum sodium lower than 110 mmol/L have been reported. Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH).
In patients with symptomatic hyponatremia, discontinue paroxetine tablets and institute appropriate medical intervention. Elderly patients, patients taking diuretics, and those who are volume-depleted may be at greater risk of developing hyponatremia with SSRIs [see Use in Specific Populations (8.5)].
5.11 Reduction of Efficacy of Tamoxifen
Some studies have shown that the efficacy of tamoxifen, as measured by the risk of breast cancer relapse/mortality, may be reduced with concomitant use of paroxetine tablets as a result of paroxetine’s irreversible inhibition of CYP2D6 and lower blood levels of tamoxifen [see Drug Interactions (7)]. One study suggests that the risk may increase with longer duration of coadministration. However, other studies have failed to demonstrate such a risk. When tamoxifen is used for the treatment or prevention of breast cancer, prescribers should consider using an alternative antidepressant with little or no CYP2D6 inhibition.
5.12 Bone Fracture
Epidemiological studies on bone fracture risk during exposure to some antidepressants, including SSRIs, have reported an association between antidepressant treatment and fractures. There are multiple possible causes for this observation, and it is unknown to what extent fracture risk is directly attributable to SSRI treatment.
5.13 Sexual Dysfunction
Use of SSRIs, including paroxetine tablets, may cause symptoms of sexual dysfunction [see Adverse Reactions (6.1)]. In male patients, SSRI use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, SSRI use may result in decreased libido and delayed or absent orgasm.
It is important for prescribers to inquire about sexual function prior to initiation of paroxetine tablets and to inquire specifically about changes in sexual function during treatment, because sexual function may not be spontaneously reported. When evaluating changes in sexual function, obtaining a detailed history (including timing of symptom onset) is important because sexual symptoms may have other causes, including the underlying psychiatric disorder. Discuss potential management strategies to support patients in making informed decisions about treatment.
-
6 ADVERSE REACTIONS
The following adverse reactions are included in more detail in other sections of the prescribing information:
- •
- Hypersensitivity reactions to paroxetine [see Contraindications (4)]
- •
- Suicidal Thoughts and Behaviors [see Warnings and Precautions (5.1)]
- •
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- •
- Embryofetal and Neonatal Toxicity [see Warnings and Precautions (5.4)]
- •
- Increased Risk of Bleeding [see Warnings and Precautions (5.5)]
- •
- Activation of Mania/Hypomania [see Warnings and Precautions (5.6)]
- •
- Discontinuation Syndrome [see Warnings and Precautions (5.7)]
- •
- Seizures [see Warnings and Precautions (5.8)]
- •
- Angle-closure Glaucoma [see Warnings and Precautions (5.9)]
- •
- Hyponatremia [see Warnings and Precautions (5.10)]
- •
- Bone Fracture [see Warnings and Precautions (5.12)]
- •
- Sexual Dysfunction [see Warnings and Precautions (5.13)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data for paroxetine tablets are from:
- •
- 6-week clinical trials in MDD patients who received paroxetine tablets 20 mg to 50 mg once daily
- •
- 12-week clinical trials in OCD patients who received paroxetine tablets 20 mg to 60 mg once daily
- •
- 10- to 12-week clinical trials in PD patients who received paroxetine tablets 10 mg to 60 mg once daily
- •
- 12-week clinical trials in SAD patients who received paroxetine tablets 20 mg to 50 mg once daily
- •
- 8-week clinical trials in GAD patients who received paroxetine tablets 10 mg to 50 mg once daily
- •
- 12-week clinical trials in PTSD patients who received paroxetine tablets 20 mg to 50 mg once daily
Adverse Reactions Leading to Discontinuation
Twenty percent (1,199/6,145) of patients treated with paroxetine tablets in clinical trials in MDD and 16.1% (84/522), 11.8% (64/542), 9.4% (44/469), 10.7% (79/735), and 11.7% (79/676) of patients treated with paroxetine tablets in clinical trials in SAD, OCD, PD, GAD, and PTSD, respectively, discontinued treatment due to an adverse reaction. The most common adverse reactions (≥ 1%) associated with discontinuation (i.e., those adverse reactions associated with dropout at a rate approximately twice or greater for paroxetine tablets compared to placebo) are presented in Table 3:
Table 3: Adverse Reactions Reported as Leading to Discontinuation (≥ 1% of Paroxetine Tablets Treated Patients and Greater than Placebo) in MDD, OCD, PD, SAD, GAD, and PTSD Trials Where numbers are not provided the incidence of the adverse reactions in patients treated with paroxetine tablets was not > 1% or was not greater than or equal to 2 times the incidence of placebo. - *
- Incidence corrected for gender.
MDD
OCD
PD
SAD
GAD
PTSD
Paroxetine
Tablets
%
Placebo
%
Paroxetine
Tablets
%
Placebo
%
Paroxetine
Tablets
%
Placebo
%
Paroxetine
Tablets
%
Placebo
%
Paroxetine
Tablets
%
Placebo
%
Paroxetine
Tablets
%
Placebo
%
CNS
Somnolence
2.3
0.7
-
1.9
0.3
3.4
0.3
2.0
0.2
2.8
0.6
Insomnia
-
-
1.7
0
1.3
0.3
3.1
0
-
-
Agitation
1.1
0.5
-
-
-
Tremor
1.1
0.3
-
1.7
0
1.0
0.2
Anxiety
-
-
-
1.1
0
-
-
Dizziness
-
-
1.5
0
1.9
0
1.0
0.2
-
-
Gastrointestinal
Constipation
-
1.1
0
-
-
Nausea
3.2
1.1
1.9
0
3.2
1.2
4.0
0.3
2.0
0.2
2.2
0.6
Diarrhea
1.0
0.3
-
Dry mouth
1.0
0.3
-
-
-
Vomiting
1.0
0.3
-
1.0
0
-
-
Flatulence
1.0
0.3
-
-
Other
Asthenia
1.6
0.4
1.9
0.4
2.5
0.6
1.8
0.2
1.6
0.2
Abnormal
Ejaculation*
1.6
0
2.1
0
4.9
0.6
2.5
0.5
-
-
Sweating
1.0
0.3
-
1.1
0
1.1
0.2
-
-
Impotence*
-
1.5
0
-
-
Libido
Decreased
1.0
0
-
-
Most Common Adverse Reactions
The most commonly observed adverse reactions associated with the use of paroxetine tablets (incidence of 5% or greater and at least twice that for placebo) were:
MDD
Asthenia, sweating, nausea, decreased appetite, somnolence, dizziness, insomnia, tremor, nervousness, ejaculatory disturbance, and other male genital disorders.
OCD
Nausea, dry mouth, decreased appetite, constipation, dizziness, somnolence, tremor, sweating, impotence, and abnormal ejaculation.
PD
Asthenia, sweating, decreased appetite, libido decreased, tremor, abnormal ejaculation, female genital disorders, and impotence.
SAD
Sweating, nausea, dry mouth, constipation, decreased appetite, somnolence, tremor, libido decreased, yawn, abnormal ejaculation, female genital disorders, and impotence.
Adverse Reactions in Patients with MDD
Table 4 presents the adverse reactions that occurred at an incidence of 1% or more and greater than placebo in clinical trials of paroxetine tablets-treated patients with MDD.
Table 4: Adverse Reactions (≥ 1% of Paroxetine Tablets-Treated Patients and Greater than Placebo) in 6-Week Clinical Trials for MDD - *
- Includes mostly “lump in throat” and “tightness in throat.”
- †
- Percentage corrected for gender.
- ‡
- Mostly “ejaculatory delay.”
- §
- Includes “anorgasmia,” “erectile difficulties,” “delayed ejaculation/orgasm,” and “sexual dysfunction,” and “impotence.”
- ¶
- Includes mostly “difficulty with micturition” and “urinary hesitancy.”
- #
- Includes mostly “anorgasmia” and “difficulty reaching climax/orgasm.”
Body System/
Adverse Reaction
Paroxetine Tablets
(n = 421)
%
Placebo
(n = 421)
%
Body as a Whole
Headache
18
17
Asthenia
15
6
Cardiovascular
Palpitation
3
1
Vasodilation
3
1
Dermatologic
Sweating
11
2
Rash
2
1
Gastrointestinal
Nausea
26
9
Dry Mouth
18
12
Constipation
14
9
Diarrhea
12
8
Decreased Appetite
6
2
Flatulence
4
2
Oropharynx Disorder*
2
0
Dyspepsia
2
1
Musculoskeletal
Myopathy
2
1
Myalgia
2
1
Myasthenia
1
0
Nervous System
Somnolence
23
9
Dizziness
13
6
Insomnia
13
6
Tremor
8
2
Nervousness
5
3
Anxiety
5
3
Paresthesia
4
2
Libido Decreased
3
0
Drugged Feeling
2
1
Confusion
1
0
Respiration
Yawn
4
0
Special Senses
Blurred Vision
4
1
Taste Perversion
2
0
Urogenital System
13
0
10
0
Urinary Frequency
3
1
Urination Disorder¶
3
0
2
0
Adverse Reactions in Patients with OCD, PD, and SAD
Table 5 presents adverse reactions that occurred at a frequency of 2% or more in clinical trial patients with OCD, PD, and SAD.
Table 5. Adverse Reactions (≥ 2% of Paroxetine Tablets-Treated Patients and Greater than Placebo) in 10 to 12-Week Clinical Trials for OCD, PD, and SAD - *
- Percentage corrected for gender.
Body System/Preferred Term
Obsessive Compulsive Disorder
Panic Disorder
Social Anxiety Disorder
Paroxetine
Tablets
(n = 542)
%
Placebo
(n = 265)
%
Paroxetine Tablets
(n = 469)
%
Placebo
(n = 324)
%
Paroxetine Tablets
(n = 425)
%
Placebo
(n = 339)
%
Body as a Whole
Asthenia
22
14
14
5
22
14
Abdominal Pain
-
-
4
3
-
-
Chest Pain
3
2
-
-
-
-
Back Pain
-
-
3
2
-
-
Chills
2
1
2
1
-
-
Trauma
-
-
-
-
3
1
Cardiovascular
Vasodilation
4
1
-
-
-
-
Palpitation
2
0
-
-
-
-
Dermatologic
Sweating
9
3
14
6
9
2
Rash
3
2
-
-
-
-
Gastrointestinal
Nausea
23
10
23
17
25
7
Dry Mouth
18
9
18
11
9
3
Constipation
16
6
8
5
5
2
Diarrhea
10
10
12
7
9
6
Decreased Appetite
9
3
7
3
8
2
Dyspepsia
-
-
-
-
4
2
Flatulence
-
-
-
-
4
2
Increased Appetite
4
3
2
1
-
-
Vomiting
-
-
-
-
2
1
Musculoskeletal
Myalgia
-
-
-
-
4
3
Nervous System
Insomnia
24
13
18
10
21
16
Somnolence
24
7
19
11
22
5
Dizziness
12
6
14
10
11
7
Tremor
11
1
9
1
9
1
Nervousness
9
8
-
-
8
7
Libido Decreased
7
4
9
1
12
1
Agitation
-
-
5
4
3
1
Anxiety
-
-
5
4
5
4
Abnormal Dreams
4
1
-
-
-
-
Concentration Impaired
3
2
-
-
4
1
Depersonalization
3
0
-
-
-
-
Myoclonus
3
0
3
2
2
1
Amnesia
2
1
-
-
-
-
Respiratory System
Rhinitis
-
-
3
0
-
-
Pharyngitis
-
-
-
-
4
2
Yawn
-
-
-
-
5
1
Special Senses
Abnormal Vision
4
2
-
-
4
1
Taste Perversion
2
0
-
-
-
-
Urogenital System
Abnormal Ejaculation*
23
1
21
1
28
1
Dysmenorrhea
-
-
-
-
5
4
Female Genital Disorder*
3
0
9
1
9
1
Impotence*
8
1
5
0
5
1
Urinary Frequency
3
1
2
0
-
-
Urination Impaired
3
0
-
-
-
-
Urinary Tract Infection
2
1
2
1
-
-
Adverse Reactions in Patients with GAD and PTSD
Table 6 presents adverse reactions that occurred at a frequency of 2% or more in clinical trials in patients with GAD and PTSD.
Table 6. Adverse Reactions (≥ 2% of Paroxetine Tablets-Treated Patients and Greater than Placebo) in 8- to 12-Week Clinical Trials for GAD and PTSDa - *
- Percentage corrected for gender.
Body System/Preferred Term
Generalized Anxiety Disorder
Posttraumatic Stress Disorder
Paroxetine
Tablets
(n = 735)
%
Placebo
(n = 529)
%
Paroxetine
Tablets
(n = 676)
%
Placebo
(n = 504)
%
Body as a Whole
Asthenia
14
6
12
4
Headache
7
14
-
-
Infection
6
3
5
4
Abdominal Pain
4
3
Trauma
6
5
Cardiovascular
Vasodilation
3
1
2
1
Dermatologic
Sweating
6
2
5
1
Gastrointestinal
Nausea
20
5
19
8
Dry Mouth
11
5
10
5
Constipation
10
2
5
3
Diarrhea
9
7
11
5
Decreased Appetite
5
1
6
3
Vomiting
3
2
3
2
Dyspepsia
-
-
5
3
Nervous System
Insomnia
11
8
12
11
Somnolence
15
5
16
5
Dizziness
6
5
6
5
Tremor
5
1
4
1
Nervousness
4
3
-
-
Libido Decreased
9
2
5
2
Abnormal Dreams
3
Respiratory System
Respiratory Disorder
7
5
-
-
Sinusitis
4
3
-
-
Yawn
4
-
2
<1
Special Senses
Abnormal Vision
2
1
3
1
Urogenital System
Abnormal Ejaculation*
25
2
13
2
Female Genital Disorder*
4
1
5
1
Impotence*
4
3
9
1
Dose Dependent Adverse Reactions
MDD
A comparison of adverse reaction rates in a fixed-dose study comparing paroxetine tablets 10 mg, 20 mg, 30 mg, and 40 mg once daily with placebo in the treatment of MDD revealed dose dependent adverse reactions, as shown in Table 7:
Table 7. Adverse Reactions (≥ 5% of Paroxetine Tablets-Treated Patients and ≥ Twice the Rate of Placebo) (in a Dose-Comparison Trial in the Treatment of MDD Placebo
Paroxetine Tablets
Body System/Preferred Term
n = 51
%
10 mg
n = 102
%
20 mg
n = 104
%
30 mg
n = 101
%
40 mg
n = 102
%
Body as a Whole
Asthenia
0.0
2.9
10.6
13.9
12.7
Dermatology
Sweating
2.0
1.0
6.7
8.9
11.8
Gastrointestinal
Constipation
5.9
4.9
7.7
9.9
12.7
Decreased Appetite
2.0
2.0
5.8
4.0
4.9
Diarrhea
7.8
9.8
19.2
7.9
14.7
Dry Mouth
2.0
10.8
18.3
15.8
20.6
Nausea
13.7
14.7
26.9
34.7
36.3
Nervous System
Anxiety
0.0
2.0
5.8
5.9
5.9
Dizziness
3.9
6.9
6.7
8.9
12.7
Nervousness
0.0
5.9
5.8
4.0
2.9
Paresthesia
0.0
2.9
1.0
5.0
5.9
Somnolence
7.8
12.7
18.3
20.8
21.6
Tremor
0.0
0.0
7.7
7.9
14.7
Special Senses
Blurred Vision
2.0
2.9
2.9
2.0
7.8
Urogenital System
Abnormal Ejaculation
0.0
5.8
6.5
10.6
13.0
Impotence
0.0
1.9
4.3
6.4
1.9
Male Genital Disorders
0.0
3.8
8.7
6.4
3.7
OCD
In a fixed-dose study comparing placebo and paroxetine tablets 20 mg, 40 mg, and 60 mg in the treatment of OCD, there was no clear relationship between adverse reactions and the dose of paroxetine tablets to which patients were assigned.
PD
In a fixed-dose study comparing placebo and paroxetine tablets 10 mg, 20 mg, and 40 mg in the treatment of PD, the following adverse reactions were shown to be dose-dependent: asthenia, dry mouth, anxiety, libido decreased, tremor, and abnormal ejaculation.
SAD
In a fixed-dose study comparing placebo and paroxetine tablets 20 mg, 40 mg and 60 mg in the treatment of SAD, for most of the adverse reactions, there was no clear relationship between adverse reactions and the dose of paroxetine tablets to which patients were assigned.
Male and Female Sexual Dysfunction
Although changes in sexual desire, sexual performance, and sexual satisfaction often occur as manifestations of a psychiatric disorder, they may also be a consequence of SSRI treatment. However, reliable estimates of the incidence and severity of untoward experiences involving sexual desire, performance, and satisfaction are difficult to obtain, however, in part because patients and healthcare providers may be reluctant to discuss them. Accordingly, estimates of the incidence of untoward sexual experience and performance cited in labeling may underestimate their actual incidence.
The percentage of patients reporting symptoms of sexual dysfunction in males and females with MDD, OCD, PD, SAD, GAD, and PTSD are displayed in Table 8.
Table 8. Adverse Reactions Related to Sexual Dysfunction in Patients Treated with Paroxetine Tablets in Clinical Trials of MDD, OCD, PD, SAD, GAD, and PTSD Paroxetine Tablets
Placebo
n (males)
1446
%
1042
%
Decreased Libido
6 to 15
0 to 5
Ejaculatory Disturbance
13 to 28
0 to 2
Impotence
2 to 9
0 to 3
n (females)
1822
%
1340
%
Decreased Libido
0 to 9
0 to 2
Orgasmic Disturbance
2 to 9
0 to 1
Paroxetine tablets treatment has been associated with several cases of priapism. In those cases with a known outcome, patients recovered without sequelae.
Hallucinations
In pooled clinical trials of paroxetine tablets, hallucinations were observed in 0.2% of paroxetine tablets-treated patients compared to 0.1% of patients receiving placebo.
Less Common Adverse Reactions
The following adverse reactions occurred during the clinical studies of paroxetine tablets and are not included elsewhere in the labeling.
Adverse reactions are categorized by body system and listed in order of decreasing frequency according to the following definitions: Frequent adverse reactions are those occurring on 1 or more occasions in at least 1/100 patients; infrequent adverse reactions are those occurring in 1/100 to 1/1,000 patients; rare adverse reactions are those occurring in fewer than 1/1,000 patients.
Body as a Whole: Infrequent: Allergic reaction, chills, face edema, malaise, neck pain; rare: Adrenergic syndrome, cellulitis, moniliasis, neck rigidity, pelvic pain, peritonitis, sepsis, ulcer.
Cardiovascular System: Frequent: Hypertension, tachycardia; infrequent: Bradycardia, hematoma, hypotension, migraine, postural hypotension, syncope; rare: Angina pectoris, arrhythmia nodal, atrial fibrillation, bundle branch block, cerebral ischemia, cerebrovascular accident, congestive heart failure, heart block, low cardiac output, myocardial infarct, myocardial ischemia, pallor, phlebitis, pulmonary embolus, supraventricular extrasystoles, thrombophlebitis, thrombosis, varicose vein, vascular headache, ventricular extrasystoles.
Digestive System: Infrequent: Bruxism, colitis, dysphagia, eructation, gastritis, gastroenteritis, gingivitis, glossitis, increased salivation, abnormal liver function tests, rectal hemorrhage, ulcerative stomatitis; rare: Aphthous stomatitis, bloody diarrhea, bulimia, cardiospasm, cholelithiasis, duodenitis, enteritis, esophagitis, fecal impactions, fecal incontinence, gum hemorrhage, hematemesis, hepatitis, ileitis, ileus, intestinal obstruction, jaundice, melena, mouth ulceration, peptic ulcer, salivary gland enlargement, sialadenitis, stomach ulcer, stomatitis, tongue discoloration, tongue edema, tooth caries.
Endocrine System: Rare: Diabetes mellitus, goiter, hyperthyroidism, hypothyroidism, thyroiditis.
Hemic and Lymphatic Systems: Infrequent: Anemia, leukopenia, lymphadenopathy, purpura; rare: Abnormal erythrocytes, basophilia, bleeding time increased, eosinophilia, hypochromic anemia, iron deficiency anemia, leukocytosis, lymphedema, abnormal lymphocytes, lymphocytosis, microcytic anemia, monocytosis, normocytic anemia, thrombocythemia, thrombocytopenia.
Metabolic and Nutritional: Frequent: Weight gain; infrequent: Edema, peripheral edema, SGOT increased, SGPT increased, thirst, weight loss; rare: Alkaline phosphatase increased, bilirubinemia, BUN increased, creatinine phosphokinase increased, dehydration, gamma globulins increased, gout, hypercalcemia, hypercholesteremia, hyperglycemia, hyperkalemia, hyperphosphatemia, hypocalcemia, hypoglycemia, hypokalemia, hyponatremia, ketosis, lactic dehydrogenase increased, non-protein nitrogen (NPN) increased.
Musculoskeletal System: Frequent: Arthralgia; infrequent: Arthritis, arthrosis; rare: Bursitis, myositis, osteoporosis, generalized spasm, tenosynovitis, tetany.
Nervous System: Frequent: Emotional lability, vertigo; infrequent: Abnormal thinking, alcohol abuse, ataxia, dystonia, dyskinesia, euphoria, hostility, hypertonia, hypesthesia, hypokinesia, incoordination, lack of emotion, libido increased, manic reaction, neurosis, paralysis, paranoid reaction; rare: Abnormal gait, akinesia, antisocial reaction, aphasia, choreoathetosis, circumoral paresthesias, convulsion, delirium, delusions, diplopia, drug dependence, dysarthria, extrapyramidal syndrome, fasciculations, grand mal convulsion, hyperalgesia, hysteria, manic-depressive reaction, meningitis, myelitis, neuralgia, neuropathy, nystagmus, peripheral neuritis, psychotic depression, psychosis, reflexes decreased, reflexes increased, stupor, torticollis, trismus, withdrawal syndrome.
Respiratory System: Infrequent: Asthma, bronchitis, dyspnea, epistaxis, hyperventilation, pneumonia, respiratory flu; rare: Emphysema, hemoptysis, hiccups, lung fibrosis, pulmonary edema, sputum increased, stridor, voice alteration.
Skin and Appendages: Frequent: Pruritus; infrequent: Acne, alopecia, contact dermatitis, dry skin, ecchymosis, eczema, herpes simplex, photosensitivity, urticaria; rare: Angioedema, erythema nodosum, erythema multiforme, exfoliative dermatitis, fungal dermatitis, furunculosis; herpes zoster, hirsutism, maculopapular rash, seborrhea, skin discoloration, skin hypertrophy, skin ulcer, sweating decreased, vesiculobullous rash.
Special Senses: Frequent: Tinnitus; infrequent: Abnormality of accommodation, conjunctivitis, ear pain, eye pain, keratoconjunctivitis, mydriasis, otitis media; rare: Amblyopia, anisocoria, blepharitis, cataract, conjunctival edema, corneal ulcer, deafness, exophthalmos, eye hemorrhage, glaucoma, hyperacusis, night blindness, otitis externa, parosmia, photophobia, ptosis, retinal hemorrhage, taste loss, visual field defect.
Urogenital System: Infrequent: Amenorrhea, breast pain, cystitis, dysuria, hematuria, menorrhagia, nocturia, polyuria, pyuria, urinary incontinence, urinary retention, urinary urgency, vaginitis; rare: Abortion, breast atrophy, breast enlargement, endometrial disorder, epididymitis, female lactation, fibrocystic breast, kidney calculus, kidney pain, leukorrhea, mastitis, metrorrhagia, nephritis, oliguria, salpingitis, urethritis, urinary casts, uterine spasm, urolith, vaginal hemorrhage, vaginal moniliasis.
6.2 Postmarketing Experience
The following reactions have been identified during post approval use of paroxetine tablets. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Acute pancreatitis, elevated liver function tests (the most severe cases were deaths due to liver necrosis, and grossly elevated transaminases associated with severe liver dysfunction), Guillain-Barré syndrome, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS), syndrome of inappropriate ADH secretion, prolactinemia and galactorrhea; extrapyramidal symptoms which have included akathisia, bradykinesia, cogwheel rigidity, oculogyric crisis which has been associated with concomitant use of pimozide; status epilepticus, acute renal failure, pulmonary hypertension, allergic alveolitis, anosmia, hyposmia, anaphylaxis, eclampsia, laryngismus, optic neuritis, porphyria, restless legs syndrome (RLS), ventricular fibrillation, ventricular tachycardia (including torsade de pointes), hemolytic anemia, events related to impaired hematopoiesis (including aplastic anemia, pancytopenia, bone marrow aplasia, and agranulocytosis), vasculitic syndromes (such as Henoch-Schönlein purpura), and premature births in pregnant women. There has been a case report of severe hypotension when paroxetine tablets were added to chronic metoprolol treatment.
-
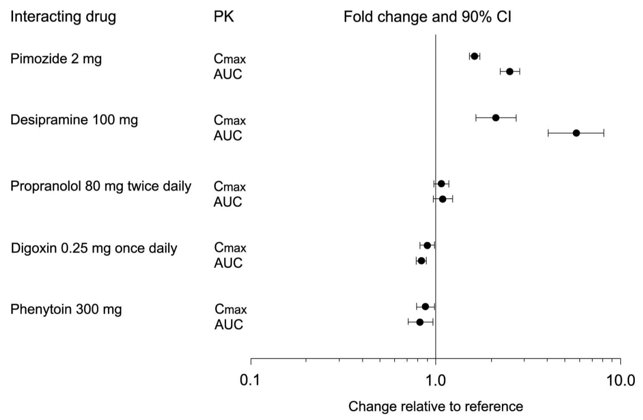
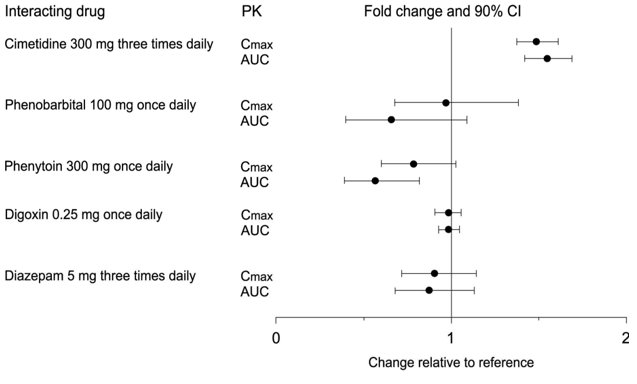
7 DRUG INTERACTIONS
Table 9 presents clinically significant drug interactions with paroxetine tablets.
Table 9: Clinically Significant Drug Interactions with Paroxetine Tablets Monoamine Oxidase Inhibitors (MAOIs)
Clinical Impact
The concomitant use of SSRIs, including paroxetine tablets, and MAOIs increases the risk of serotonin syndrome.
Intervention
Paroxetine tablets are contraindicated in patients taking MAOIs, including MAOIs such as linezolid or intravenous methylene blue [see Dosage and Administration (2.5), Contraindications (4), Warnings and Precautions (5.2)].
Examples
selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue
Pimozide and Thioridazine
Clinical Impact
Increased plasma concentrations of pimozide and thioridazine, drugs with a narrow therapeutic index, may increase the risk of QTc prolongation and ventricular arrhythmias.
Intervention
Paroxetine tablets are contraindicated in patients taking pimozide or thioridazine [see Contraindications (4)].
Other Serotonergic Drugs
Clinical Impact
The concomitant use of serotonergic drugs with paroxetine tablets increases the risk of serotonin syndrome.
Intervention
Monitor patients for signs and symptoms of serotonin syndrome, particularly during treatment initiation and dosage increases. If serotonin syndrome occurs, consider discontinuation of paroxetine tablets and/or concomitant serotonergic drugs [see Warnings and Precautions (5.2)].
Examples
other SSRIs, SNRIs, triptans, tricyclic antidepressants, opioids, lithium, tryptophan, buspirone, amphetamines, and St. John’s Wort
Drugs that Interfere with Hemostasis (antiplatelet agents and anticoagulants)
Clinical Impact
The concurrent use of an antiplatelet agent or anticoagulant with paroxetine tablets may potentiate the risk of bleeding.
Intervention
Inform patients of the increased risk of bleeding associated with the concomitant use of paroxetine tablets and antiplatelet agents and anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio [see Warnings and Precautions (5.5)].
Examples
aspirin, clopidogrel, heparin, warfarin
Drugs Highly Bound to Plasma Protein
Clinical Impact
Paroxetine tablets are highly bound to plasma protein. The concomitant use of paroxetine tablets with another drug that is highly bound to plasma protein may increase free concentrations of paroxetine tablets or other tightly-bound drugs in plasma.
Intervention
Monitor for adverse reactions and reduce dosage of paroxetine tablets or other protein-bound drugs as warranted.
Examples
warfarin
Drugs Metabolized by CYP2D6
Clinical Impact
Paroxetine tablets are a CYP2D6 inhibitor [see Clinical Pharmacology (12.3)]. The concomitant use of paroxetine tablets with a CYP2D6 substrate may increase the exposure of the CYP2D6 substrate.
Intervention
Decrease the dosage of a CYP2D6 substrate if needed with concomitant paroxetine tablets use. Conversely, an increase in dosage of a CYP2D6 substrate may be needed if paroxetine tablets are discontinued.
Examples
propafenone, flecainide, atomoxetine, desipramine, dextromethorphan, metoprolol, nebivolol, perphenazine, tolterodine, venlafaxine, risperidone.
Tamoxifen
Clinical Impact
Concomitant use of tamoxifen with paroxetine tablets may lead to reduced plasma concentrations of the active metabolite (endoxifen) and reduced efficacy of tamoxifen
Intervention
Consider use of an alternative antidepressant with little or no CYP2D6 inhibition [see Warnings and Precautions (5.11)].
Fosamprenavir/Ritonavir
Clinical Impact
Co-administration of fosamprenavir/ritonavir with paroxetine tablets significantly decreased plasma levels of paroxetine.
Intervention
Any dose adjustment should be guided by clinical effect (tolerability and efficacy).
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on data from published observational studies, exposure to SSRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Warnings and Precautions (5.5) and Clinical Considerations].
Epidemiological studies have shown that infants exposed to paroxetine in the first trimester of pregnancy have an increased risk of congenital malformations, particularly cardiovascular malformations. If paroxetine is used during pregnancy, or if the patient becomes pregnant while taking paroxetine, advise the patient of the potential hazard to the fetus.
Clinical Considerations
Unless the benefits of paroxetine to the mother justify continuing treatment, consideration should be given to either discontinuing paroxetine therapy or switching to another antidepressant [see Warnings and Precautions (5.7)]. For
- •
- A study based on Swedish national registry data demonstrated that infants exposed to paroxetine during pregnancy (n = 815) had an increased risk of cardiovascular malformations (2% risk in paroxetine-exposed infants) compared to the entire registry population (1% risk), for an odds ratio (OR) of 1.8 (95% confidence interval 1.1 to 2.8). No increase in the risk of overall congenital malformations was seen in the paroxetine-exposed infants. The cardiac malformations in the paroxetine-exposed infants were primarily ventricular septal defects (VSDs) and atrial septal defects (ASDs). Septal defects range in severity from those that resolve spontaneously to those which require surgery.
- •
- A separate retrospective cohort study from the United States (United Healthcare data) evaluated 5,956 infants of mothers dispensed antidepressants during the first trimester (n = 815 for paroxetine). This study showed a trend towards an increased risk for cardiovascular malformations for paroxetine (risk of 1.5%) compared to other antidepressants (risk of 1%), for an OR of 1.5 (95% confidence interval 0.8 to 2.9). Of the 12 paroxetine-exposed infants with cardiovascular malformations, 9 had VSDs. This study also suggested an increased risk of overall major congenital malformations including cardiovascular defects for paroxetine (4% risk) compared to other (2% risk) antidepressants (OR 1.8; 95% confidence interval 1.2 to 2.8).
- •
- Two large case-control studies using separate databases, each with > 9,000 birth defect cases and > 4,000 controls, found that maternal use of paroxetine during the first trimester of pregnancy was associated with a 2- to 3-fold increased risk of right ventricular outflow tract obstructions. In one study the OR was 2.5 (95% confidence interval, 1.0 to 6.0, 7 exposed infants) and in the other study the OR was 3.3 (95% confidence interval, 1.3 to 8.8, 6 exposed infants).
Other studies have found varying results as to whether there was an increased risk of overall, cardiovascular, or specific congenital malformations. A meta-analysis of epidemiological data over a 16-year period (1992 to 2008) on first trimester paroxetine use in pregnancy and congenital malformations included the above-noted studies in addition to others (n = 17 studies that included overall malformations and n = 14 studies that included cardiovascular malformations; n = 20 distinct studies). While subject to limitations, this meta-analysis suggested an increased occurrence of cardiovascular malformations (prevalence odds ratio [POR] 1.5; 95% confidence interval 1.2 to 1.9) and overall malformations (POR 1.2; 95% confidence interval 1.1 to 1.4) with paroxetine use during the first trimester. It was not possible in this meta-analysis to determine the extent to which the observed prevalence of cardiovascular malformations might have contributed to that of overall malformations, nor was it possible to determine whether any specific types of cardiovascular malformations might have contributed to the observed prevalence of all cardiovascular malformations.
Unless the benefits of paroxetine to the mother justify continuing treatment, consideration should be given to either discontinuing paroxetine therapy or switching to another antidepressant [see Warnings and Precautions (5.7)]. For women who intend to become pregnant or are in their first trimester of pregnancy, paroxetine should only be initiated after consideration of the other available treatment options [see Warnings and Precautions (5.4)].
Treatment of Pregnant Women During Their Third Trimester
Neonates exposed to SSRIs or serotonin and norepinephrine reuptake inhibitors (SNRIs), including paroxetine tablets, late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding. Such complications can arise immediately upon delivery. Reported clinical findings have included respiratory distress, cyanosis, apnea, seizures, temperature instability, feeding difficulty, vomiting, hypoglycemia, hypotonia, hypertonia, hyperreflexia, tremor, jitteriness, irritability, and constant crying. These features are consistent with either a direct toxic effect of SSRIs and SNRIs or, possibly, a drug discontinuation syndrome. It should be noted that, in some cases, the clinical picture is consistent with serotonin syndrome [see Warnings and Precautions (5.2)].
Exposure to SSRIs in late pregnancy may have an increased risk for persistent pulmonary hypertension of the newborn (PPHN). PPHN occurs in 1 – 2 per 1,000 live births in the general population and is associated with substantial neonatal morbidity and mortality. In a retrospective case-control study of 377 women whose infants were born with PPHN and 836 women whose infants were born healthy, the risk for developing PPHN was approximately six-fold higher for infants exposed to SSRIs after the 20th week of gestation compared to infants who had not been exposed to antidepressants during pregnancy.
There have also been postmarketing reports of premature births in pregnant women exposed to paroxetine or other SSRIs.
When treating a pregnant woman with paroxetine during the third trimester, the physician should carefully consider both the potential risks and benefits of treatment. A prospective longitudinal study of 201 women with a history of major depression who were euthymic at the beginning of pregnancy. The women who discontinued antidepressant medication during pregnancy were more likely to experience a relapse of major depression than women who continued antidepressant medication.
Maternal Adverse Reactions
Use of paroxetine tablets in the month before delivery may be associated with an increased risk of postpartum hemorrhage [see Warnings and Precautions (5.5)].
Animal Findings
Reproduction studies were performed at doses up to 50 mg/kg/day in rats and 6 mg/kg/day in rabbits administered during organogenesis. These doses are approximately 6 (rat) and less than 2 (rabbit) times the maximum recommended human dose (MRHD – 75 mg) on an mg/m2 basis. These studies have revealed no evidence of developmental effects. However, in rats, there was an increase in pup deaths during the first 4 days of lactation when dosing occurred during the last trimester of gestation and continued throughout lactation. This effect occurred at a dose of 1 mg/kg/day which is than the MRHD on an mg/m2 basis. The no-effect dose for rat pup mortality was not determined. The cause of these deaths is not known.
8.3 Nursing Mothers
Like many other drugs, paroxetine is secreted in human milk. Because of the potential for serious adverse reactions in nursing infants from paroxetine tablets, a decision should be made whether to discontinue nursing infants or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness of paroxetine tablets in pediatric patients have not been established [see Box Warning]. Effectiveness was not demonstrated in three placebo-controlled trials in 752 paroxetine tablets-treated pediatric patients with MDD.
Antidepressants increase the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning, Warnings and Precautions (5.1)]. Decreased appetite and weight loss have been observed in association with the use of SSRIs.
In placebo-controlled clinical trials conducted with pediatric patients, the following adverse reactions were reported in at least 2% of pediatric patients treated with paroxetine tablets and occurred at a rate at least twice that for pediatric patients receiving placebo: emotional lability (including self-harm, suicidal thoughts, attempted suicide, crying, and mood fluctuations), hostility, decreased appetite, tremor, sweating, hyperkinesia, and agitation.
Adverse reactions upon discontinuation of treatment with paroxetine tablets in the pediatric clinical trials that included a taper phase regimen, which occurred in at least 2% of patients and at a rate at least twice that of placebo, were: emotional lability (including suicidal ideation, suicide attempt, mood changes, and tearfulness), nervousness, dizziness, nausea, and abdominal pain.
8.5 Geriatric Use
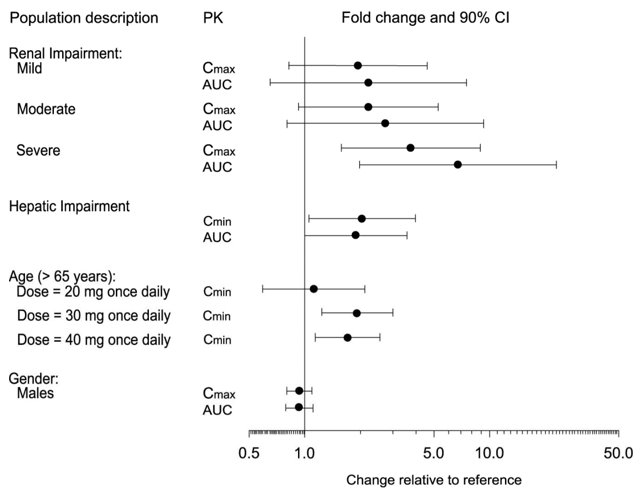
In premarketing clinical trials with paroxetine tablets, 17% of patients treated with paroxetine tablets (approximately 700) were 65 years of age or older. Pharmacokinetic studies revealed a decreased clearance in the elderly, and a lower starting dose is recommended;, however, no overall differences in safety or effectiveness were observed between elderly and younger patients [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
SSRIs including paroxetine tablets, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse reaction [see Warnings and Precautions (5.7)].
8.6 Renal and Hepatic Impairment
Increased plasma concentrations of paroxetine occur in patients with renal and hepatic impairment. The initial dosage of paroxetine tablets should be reduced in patients with severe renal impairment and in patients with severe hepatic impairment [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
The following have been reported with paroxetine tablet overdosage:
- •
- Seizures, which may be delayed, and altered mental status including coma.
- •
- Cardiovascular toxicity, which may be delayed, including QRS and QTc interval prolongation. Hypertension most commonly seen, but rarely can see hypotension alone or with co-ingestants including alcohol.
- •
- Serotonin syndrome (patients with a multiple drug overdosage with other proserotonergic drugs may have a higher risk).
Gastrointestinal decontamination with activated charcoal should be considered in patients who present early after a paroxetine overdose.
Consider contacting a Poison Center (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
-
11 DESCRIPTION
Paroxetine tablets contain paroxetine hydrochloride, an SSRI. It is the hydrochloride salt of a phenylpiperidine compound identified chemically as (3S-trans)-3-[(1,3-Benzodioxol-5-yloxy)methyl]-4-(4-fluorophenyl)-piperidine hydrochloride hemihydrate and has the molecular formula of C19H20FNO3•HCl•1/2H2O. The molecular weight is 374.8 (329.4 as free base). The structural formula of paroxetine hydrochloride is:
Paroxetine hydrochloride, USP (hemihydrate) is an odorless, white or almost white crystalline powder, having a melting point range of 129° to 131°C and a solubility of 5.4 mg/mL in water.
Paroxetine Tablets: Paroxetine tablets are for oral administration. Each film-coated tablet contains 10 mg, 20 mg, 30 mg or 40 mg paroxetine equivalent to 11.38 mg, 22.76 mg, 34.14 mg or 45.52 mg of paroxetine hydrochloride hemihydrate, respectively.
Inactive ingredients consist of dibasic calcium phosphate dihydrate, FD&C Blue No. 1 Aluminum Lake, hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, sodium lauryl sulfate, sodium starch glycolate (potato), titanium dioxide and triacetin.
Paroxetine Hydrochloride Meets USP Organic Impurities Test 1.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of paroxetine tablets in the treatment of MDD, SAD, OCD, PD, GAD, and PTSD is unknown, but is presumed to be linked to potentiation of serotonergic activity in the central nervous system resulting from inhibition of neuronal reuptake of serotonin (5-hydroxy-tryptamine, 5-HT).
12.2 Pharmacodynamics
Studies at clinically relevant doses in humans have demonstrated that paroxetine blocks the uptake of serotonin into human platelets. In vitro studies in animals also suggest that paroxetine is a potent and highly selective inhibitor of neuronal serotonin reuptake (SSRI) and has only very weak effects on norepinephrine and dopamine neuronal reuptake.
12.3 Pharmacokinetics
Nonlinearity in pharmacokinetics is observed with increasing doses of paroxetine tablets.
In a meta-analysis of paroxetine from 4 studies done in healthy volunteers following multiple dosing of 20 mg/day to 40 mg/day, males did not exhibit a significantly lower Cmax or AUC than females.
Absorption
Paroxetine hydrochloride is completely absorbed after oral dosing of a solution of the hydrochloride salt. In a study in which normal male subjects (n = 15) received 30 mg tablets daily for 30 days, steady-state paroxetine concentrations were achieved by approximately 10 days for most subjects, although it may take substantially longer in an occasional patient. At steady state, mean values of Cmax, Tmax, Cmin, and T½ were 61.7 ng/mL (CV 45%), 5.2 hr. (CV 10%), 30.7 ng/mL (CV 67%), and 21.0 hours (CV 32%), respectively. The steady-state Cmax and Cmin values were about 6 and 14 times what would be predicted from single-dose studies. Steady-state drug exposure based on AUC0-24 was about 8 times greater than would have been predicted from single-dose data in these subjects. The excess accumulation is a consequence of the fact that 1 of the enzymes that metabolizes paroxetine is readily saturable.
Paroxetine is equally bioavailable from the oral suspension and tablet.
Effect of Food
The effects of food on the bioavailability of paroxetine were studied in subjects administered a single dose with and without food. AUC was only slightly increased (6%) when drug was administered with food but the Cmax was 29% greater, while the time to reach peak plasma concentration decreased from 6.4 hours post-dosing to 4.9 hours.
Distribution
Paroxetine distributes throughout the body, including the CNS, with only 1% remaining in the plasma.
Approximately 95% and 93% of paroxetine is bound to plasma protein at 100 ng/mL and 400 ng/mL, respectively. Under clinical conditions, paroxetine concentrations would normally be less than 400 ng/mL. Paroxetine does not alter the in vitro protein binding of phenytoin or warfarin.
Elimination
Metabolism
The mean elimination half-life is approximately 21 hours (CV 32%) after oral dosing of 30 mg tablets daily for 30 days of paroxetine tablets.
In steady-state dose proportionality studies involving elderly and nonelderly patients, at doses of 20 mg to 40 mg daily for the elderly and 20 mg to 50 mg daily for the nonelderly, some nonlinearity was observed in both populations, again reflecting a saturable metabolic pathway. In comparison to Cmin values after 20 mg daily, values after 40 mg daily were only about 2 to 3 times greater than doubled.
Paroxetine is extensively metabolized after oral administration. The principal metabolites are polar and conjugated products of oxidation and methylation, which are readily cleared. Conjugates with glucuronic acid and sulfate predominate, and major metabolites have been isolated and identified. Data indicate that the metabolites have no more than 1/50 the potency of the parent compound at inhibiting serotonin uptake. The metabolism of paroxetine is accomplished in part by CYP2D6. Saturation of this enzyme at clinical doses appears to account for the nonlinearity of paroxetine kinetics with increasing dose and increasing duration of treatment. The role of this enzyme in paroxetine metabolism also suggests potential drug-drug interactions [see Drug Interactions (7)]. Pharmacokinetic behavior of paroxetine has not been evaluated in subjects who are deficient in CYP2D6 (poor metabolizers).
Excretion
Approximately 64% of a 30-mg oral solution dose of paroxetine was excreted in the urine with 2% as the parent compound and 62% as metabolites over a 10-day post-dosing period. About 36% was excreted in the feces (probably via the bile), mostly as metabolites and less than 1% as the parent compound over the 10-day post-dosing period.
Drug Interaction Studies
There are clinically significant, known drug interactions between paroxetine and other drugs [see Drug Interactions (7)].
Theophylline
Reports of elevated theophylline levels associated with paroxetine tablets treatment have been reported. While this interaction has not been formally studied, it is recommended that theophylline levels be monitored when these drugs are concurrently administered.
Drugs Metabolized by Cytochrome CYP3A4
An in vivo interaction study involving the coadministration under steady-state conditions of paroxetine and terfenadine, a substrate for CYP3A4, revealed no effect of paroxetine on terfenadine pharmacokinetics. In addition, in vitro studies have shown ketoconazole, a potent inhibitor of CYP3A4 activity, to be at least 100 times more potent than paroxetine as an inhibitor of the metabolism of several substrates for this enzyme, including terfenadine, astemizole, cisapride, triazolam, and cyclosporine. Paroxetine’s extent of inhibition of CYP3A4 activity is not expected to be of clinical significance.
Specific Populations
The impact of specific populations on the pharmacokinetics of paroxetine are shown in Figure 3.
The recommended starting dosage and maximum dosage of paroxetine tablets are reduced in elderly patients, patients with severe renal impairment, and patients with severe hepatic impairment [see Dosage and Administration (2.4)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Two-year carcinogenicity studies were conducted in rodents given paroxetine in the diet at 1, 5, and 25 mg/kg/day (mice) and 1, 5, and 20 mg/kg/day (rats). These doses are up to 2.0 (mouse) and 3.2 (rat) times the MRHD of 75 mg on a mg/m2 basis. There was a significantly greater number of male rats in the high-dose group with reticulum cell sarcomas (1/100, 0/50, 0/50, and 4/50 for control, low-, middle-, and high-dose groups, respectively) and a significantly increased linear trend across dose groups for the occurrence of lymphoreticular tumors in male rats. Female rats were not affected. Although there was a dose-related increase in the number of tumors in mice, there was no drug-related increase in the number of mice with tumors. The relevance of these findings to humans is unknown.
Mutagenesis
Paroxetine produced no genotoxic effects in a battery of 5 in vitro and 2 in vivo assays that included the following: Bacterial mutation assay, mouse lymphoma mutation assay, unscheduled DNA synthesis assay, and tests for cytogenetic aberrations in vivo in mouse bone marrow and in vitro in human lymphocytes and in a dominant lethal test in rats.
Impairment of Fertility
Some clinical studies have shown that SSRIs (including paroxetine) may affect sperm quality during SSRI treatment, which may affect fertility in some men.
A reduced pregnancy rate was found in reproduction studies in rats at a dose of paroxetine of 15 mg/kg/day, which is 2.4 times the MRHD of 75 mg on a mg/m2 basis. Irreversible lesions occurred in the reproductive tract of male rats after dosing in toxicity studies for 2 to 52 weeks. These lesions consisted of vacuolation of epididymal tubular epithelium at 50 mg/kg/day and atrophic changes in the seminiferous tubules of the testes with arrested spermatogenesis at 25 mg/kg/day (8.2 and 4.1 times the MRHD of 75 mg on a mg/m2 basis).
-
14 CLINICAL STUDIES
14.1 Major Depressive Disorder
The efficacy of paroxetine tablets as a treatment for major depressive disorder (MDD) has been established in 6 placebo-controlled studies of patients with MDD (aged 18 to 73). In these studies, paroxetine tablets were shown to be statistically significantly more effective than placebo in treating MDD by at least 2 of the following measures: Hamilton Depression Rating Scale (HDRS), the Hamilton depressed mood item, and the Clinical Global Impression (CGI)-Severity of Illness. Paroxetine tablets were statistically significantly better than placebo in improvement of the HDRS sub-factor scores, including the depressed mood item, sleep disturbance factor, and anxiety factor.
Long-term efficacy of paroxetine tablets for treatment of MDD in outpatients was demonstrated in a randomized withdrawal study. Patients who responded to paroxetine tablets (HDRS total score < 8) during an initial 8-week open-label treatment phase were then randomized to continue paroxetine tablets or placebo, for up to 1 year. Patients treated with paroxetine tablets demonstrated a statistically significant lower relapse rate during the withdrawal phase (15%) compared to those on placebo (39%). Effectiveness was similar for male and female patients.
14.2 Obsessive Compulsive Disorder
The effectiveness of paroxetine tablets in the treatment of obsessive compulsive disorder (OCD) was demonstrated in two 12-week multicenter placebo-controlled studies of adult outpatients (Studies 1 and 2). Patients had moderate to severe OCD (DSM-IIIR) with mean baseline ratings on the Yale Brown Obsessive Compulsive Scale (YBOCS) total score ranging from 23 to 26. In study 1, a dose-range finding study, patients received fixed daily doses of paroxetine tablets 20 mg, 40 mg, or 60 mg. Study 1 demonstrated that daily doses of paroxetine tablets 40 mg and 60 mg are effective in the treatment of OCD. Patients receiving doses of paroxetine tablets 40 mg and 60 mg experienced a mean reduction of approximately 6 and 7 points, respectively, on the YBOCS total score which was statistically significantly greater than the approximate 4-point reduction at 20 mg and a 3-point reduction in the placebo-treated patients. Study 2 was a flexible-dose study comparing paroxetine tablets 20 mg to 60 mg daily with clomipramine 25 mg to 250 mg daily or placebo). In this study, patients receiving paroxetine tablets experienced a mean reduction of approximately 7 points on the YBOCS total score, which was statistically significantly greater than the mean reduction of approximately 4 points in placebo-treated patients.
The following table provides the outcome classification by treatment group on Global Improvement items of the Clinical Global Impression (CGI) scale for Study 1.
Table 10: Outcome Classification (%) on CGI-Global Improvement Item for Completers in Study 1 in Patients with OCD Outcome Classification
Placebo
(n = 74)
%
Paroxetine
Tablets 20 mg
(n = 75)
%
Paroxetine
Tablets 40 mg
(n = 66)
%
Paroxetine
Tablets 60 mg
(n = 66)
%
Worse
14
7
7
3
No Change
44
35
22
19
Minimally Improved
24
33
29
34
Much Improved
11
18
22
24
Very Much Improved
7
7
20
20
Subgroup analyses did not indicate that there were any differences in treatment outcomes as a function of age or gender.
The long-term efficacy of paroxetine tablets for the treatment of OCD was established in a long-term extension to Study 1. Patients who responded to paroxetine tablets during the 3-month double-blind phase and a 6-month extension on open-label paroxetine tablets 20 mg to 60 mg daily were randomized to either paroxetine tablets or placebo in a 6-month double-blind relapse prevention phase. Patients randomized to paroxetine tablets were statistically significantly less likely to relapse than placebo-treated patients.
14.3 Panic Disorder
The effectiveness of paroxetine tablets in the treatment of panic disorder (PD) was demonstrated in three 10- to 12-week multicenter, placebo-controlled studies of adult outpatients (Studies 1, 2, and 3). Patients had PD (DSM-IIIR), with or without agoraphobia. In these studies, paroxetine tablets were shown to be statistically significantly more effective than placebo in treating PD by at least 2 out of 3 measures of panic attack frequency and on the Clinical Global Impression Severity of Illness score.
Study 1 was a 10-week dose-range finding study; patients received fixed doses of paroxetine tablets 10 mg, 20 mg, or 40 mg daily or placebo. A statistically significant difference from placebo was observed only for the paroxetine tablets 40 mg daily group. At endpoint, 76% of patients receiving paroxetine tablets 40 mg daily were free of panic attacks, compared to 44% of placebo-treated patients.
Study 2 was a 12-week flexible-dose study comparing paroxetine tablets 10 mg to 60 mg daily and placebo. At endpoint, 51% of paroxetine tablets-treated patients were free of panic attacks compared to 32% of placebo-treated patients.
Study 3 was a 12-week flexible-dose study comparing paroxetine tablets 10 mg to 60 mg daily to placebo in patients concurrently receiving standardized cognitive behavioral therapy. At endpoint, 33% of the paroxetine tablets-treated patients showed a reduction to 0 or 1 panic attacks compared to 14% of placebo-treated patients.
In Studies 2 and 3, the mean paroxetine tablets dose for completers at endpoint was approximately 40 mg daily.
Long-term efficacy of paroxetine tablets in PD was demonstrated in an extension to Study 1. Patients who responded to paroxetine tablets during the 10-week double-blind phase and during a 3-month double-blind extension phase were randomized to either paroxetine tablets 10 mg, 20 mg, or 40 mg daily or placebo in a 3-month double-blind relapse prevention phase. Patients randomized to paroxetine tablets were statistically significantly less likely to relapse than placebo-treated patients.
Subgroup analyses did not indicate that there were any differences in treatment outcomes as a function of age or gender.
14.4 Social Anxiety Disorder
The effectiveness of paroxetine tablets in the treatment of social anxiety disorder (SAD) was demonstrated in three 12-week, multicenter, placebo-controlled studies (Studies 1, 2, and 3) of adult outpatients with SAD (DSM-IV). In these studies, the effectiveness of paroxetine tablets compared to placebo was evaluated on the basis of (1) the proportion of responders, as defined by a Clinical Global Impression (CGI) Improvement score of 1 (very much improved) or 2 (much improved), and (2) change from baseline in the Liebowitz Social Anxiety Scale (LSAS).
Studies 1 and 2 were flexible-dose studies comparing paroxetine tablets 20 mg to 50 mg daily and placebo. Paroxetine tablets demonstrated statistically significant superiority over placebo on both the CGI Improvement responder criterion and the Liebowitz Social Anxiety Scale (LSAS). In Study 1, for patients who completed to week 12, 69% of paroxetine tablets-treated patients compared to 29% of placebo-treated patients were CGI Improvement responders. In Study 2, CGI Improvement responders were 77% and 42% for the paroxetine tablets- and placebo-treated patients, respectively.
Study 3 was a 12-week study comparing fixed doses of paroxetine tablets 20 mg, 40 mg, or 60 mg daily with placebo. Paroxetine tablets 20 mg was statistically significantly superior to placebo on both the LSAS Total Score and the CGI Improvement responder criterion; there were trends for superiority over placebo for the paroxetine tablets 40 mg and 60 mg daily dose groups. There was no indication in this study of any additional benefit for doses higher than 20 mg daily.
Subgroup analyses generally did not indicate differences in treatment outcomes as a function of age, race, or gender.
14.5 Generalized Anxiety Disorder
The effectiveness of paroxetine tablets in the treatment of generalized anxiety disorder (GAD) was demonstrated in two 8-week, multicenter, placebo-controlled studies (Studies 1 and 2) of adult outpatients with GAD (DSM-IV).
Study 1 was an 8-week study comparing fixed doses of paroxetine tablets 20 mg or 40 mg daily with placebo. Doses of paroxetine tablets 20 mg or 40 mg were both demonstrated to be statistically significantly superior to placebo on the Hamilton Rating Scale for Anxiety (HAM-A) total score.
There was not sufficient evidence in this study to suggest a greater benefit for the paroxetine tablets 40 mg daily dose compared to the 20 mg daily dose.
Study 2 was a flexible-dose study comparing paroxetine tablets 20 mg to 50 mg daily and placebo. Paroxetine tablets demonstrated statistically significant superiority over placebo on the Hamilton Rating Scale for Anxiety (HAM-A) total score.
A third study, a flexible-dose study comparing paroxetine tablets 20 mg to 50 mg daily to placebo, did not demonstrate statistically significant superiority of paroxetine tablets over placebo on the Hamilton Rating Scale for Anxiety (HAM-A) total score, the primary outcome.
Subgroup analyses did not indicate differences in treatment outcomes as a function of race or gender. There were insufficient elderly patients to conduct subgroup analyses on the basis of age.
In a long-term trial, 566 patients meeting DSM-IV criteria for GAD, who had responded during a single-blind, 8-week acute treatment phase with paroxetine tablets 20 mg to 50 mg daily, were randomized to continuation of paroxetine tablets at their same dose, or to placebo, for up to 24 weeks of observation for relapse. Response during the single-blind phase was defined by having a decrease of ≥ 2 points compared to baseline on the CGI-Severity of Illness scale, to a score of ≤ 3. Relapse during the double-blind phase was defined as an increase of ≥ 2 points compared to baseline on the CGI-Severity of Illness scale to a score of ≥ 4, or withdrawal due to lack of efficacy. Patients continuing to receive paroxetine tablets experienced a statistically significantly lower relapse rate over the subsequent 24 weeks compared to those receiving placebo.
14.6 Posttraumatic Stress Disorder
The effectiveness of paroxetine tablets in the treatment of Posttraumatic Stress Disorder (PTSD) was demonstrated in two 12-week, multicenter, placebo-controlled studies (Studies 1 and 2) of adult outpatients who met DSM-IV criteria for PTSD. The mean duration of PTSD symptoms for the 2 studies combined was 13 years (ranging from 0.1 year to 57 years). The percentage of patients with secondary MDD or non-PTSD anxiety disorders in the combined 2 studies was 41% (356 out of 858 patients) and 40% (345 out of 858 patients), respectively. Study outcome was assessed by (1) the Clinician-Administered PTSD Scale Part 2 (CAPS-2) score and (2) the Clinical Global Impression-Global Improvement Scale (CGI-I). The CAPS-2 is a multi-item instrument that measures 3 aspects of PTSD with the following symptom clusters: Reexperiencing/intrusion, avoidance/numbing and hyperarousal. The 2 primary outcomes for each trial were (1) change from baseline to endpoint on the CAPS-2 total score (17 items), and (2) proportion of responders on the CGI-I, where responders were defined as patients having a score of 1 (very much improved) or 2 (much improved).
Study 1 was a 12-week study comparing fixed doses of paroxetine tablets 20 mg or 40 mg daily to placebo. Doses of paroxetine tablets 20 mg and 40 mg were demonstrated to be statistically significantly superior to placebo on change from baseline for the CAPS-2 total score and on proportion of responders on the CGI-I. There was not sufficient evidence in this study to suggest a greater benefit for the 40 mg daily dose compared to the 20 mg daily dose.
Study 2 was a 12-week flexible-dose study comparing paroxetine tablets 20 mg to 50 mg daily to placebo. Paroxetine tablets were demonstrated to be significantly superior to placebo on change from baseline for the CAPS-2 total score and on proportion of responders on the CGI-I.
A third study, a flexible-dose study comparing paroxetine tablets 20 mg to 50 mg daily to placebo, demonstrated paroxetine tablets to be statistically significantly superior to placebo on change from baseline for CAPS-2 total score, but not on proportion of responders on the CGI-I.
The majority of patients in these trials were women (68% women: 377 out of 551 subjects in Study 1 and 66% women: 202 out of 303 subjects in Study 2). Subgroup analyses did not indicate differences in treatment outcomes as a function of gender. There were an insufficient number of patients who were 65 years and older or were non-Caucasian to conduct subgroup analyses on the basis of age or race, respectively.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Paroxetine Tablets, USP are available containing paroxetine hydrochloride, USP (hemihydrate) equivalent to 10 mg, 20 mg, 30 mg or 40 mg of paroxetine.
The 10 mg tablets are blue, film-coated, modified capsule-shaped, scored tablets debossed with M on one side of the tablet and N to the left of the score and 1 to the right of the score on the other side. They are available as follows:
NDC 0378-7001-93
bottles of 30 tabletsNDC 0378-7001-10
bottles of 1000 tabletsThe 20 mg tablets are blue, film-coated, modified capsule-shaped, scored tablets debossed with M on one side of the tablet and N to the left of the score and 2 to the right of the score on the other side. They are available as follows:
NDC 0378-7002-93
bottles of 30 tabletsNDC 0378-7002-10
bottles of 1000 tabletsThe 30 mg tablets are blue, film-coated, round, unscored tablets debossed with M over N3 on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-7003-93
bottles of 30 tabletsNDC 0378-7003-10
bottles of 1000 tabletsThe 40 mg tablets are blue, film-coated, round, unscored tablets debossed with M over N4 on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-7004-93
bottles of 30 tabletsNDC 0378-7004-10
bottles of 1000 tabletsStore at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Sexual Dysfunction: Advise patients that use of paroxetine tablets may cause symptoms of sexual dysfunction in both male and female patients. Inform patients that they should discuss any changes in sexual function and potential management strategies with their healthcare provider [see Warnings and Precautions (5.13)].
Suicidal Thoughts and Behaviors: Advise patients and caregivers to look for the emergence of suicidality, especially early during treatment and when the dosage is adjusted up or down, and instruct them to report such symptoms to the healthcare provider [see Boxed Warning and Warnings and Precautions (5.1)].
Serotonin Syndrome: Caution patients about the risk of serotonin syndrome, particularly with the concomitant use of paroxetine tablets with other serotonergic drugs including triptans, tricyclic antidepressants, opioids, lithium, tryptophan, buspirone, amphetamines, St. John’s Wort, and with drugs that impair metabolism of serotonin (in particular, MAOIs, both those intended to treat psychiatric disorders and also others, such as linezolid). Instruct patients to contact their health care provider or report to the emergency room if they experience signs or symptoms of serotonin syndrome [see Warnings and Precautions (5.2), Drug Interactions (7)].
Concomitant Medications: Advise patients to inform their physician if they are taking, or plan to take, any prescription or over-the-counter drugs, since there is a potential for drug-drug interactions [see Warning and Precautions (5.3), Drug Interactions (7)].
Increased Risk of Bleeding: Inform patients about the concomitant use of paroxetine tablets with aspirin, NSAIDs, other antiplatelet drugs, warfarin, or other anticoagulants because the combined use has been associated with an increased risk of bleeding. Advise patients to inform their health care providers if they are taking or planning to take any prescription or over-the counter medications that increase the risk of bleeding [see Warnings and Precautions (5.5)].
Activation of Mania/Hypomania: Advise patients and their caregivers to observe for signs of activation of mania/hypomania and instruct them to report such symptoms to the healthcare provider [see Warnings and Precautions (5.6)].
Discontinuation Syndrome: Advise patients not to abruptly discontinue paroxetine tablets and to discuss any tapering regimen with their healthcare provider. Inform patients that adverse reactions can occur when paroxetine tablets are discontinued [see Warnings and Precautions (5.7)].
Allergic Reactions: Advise patients to notify their healthcare provider if they develop an allergic reaction such as rash, hives, swelling, or difficulty breathing [see Adverse Reactions (6.1, 6.2)].
Embryo-Fetal Toxicity: Advise women of the potential risk to the fetus [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)]. Advise patients to notify their healthcare provider if they become pregnant or intend to become pregnant during therapy because of the risk to the fetus.
Nursing: Advise women to notify their healthcare provider if they are breastfeeding an infant [see Use in Specific Populations (8.3)].
-
Medication Guide
Paroxetine Tablets, USP
(pa roxʹ e teen)What is the most important information I should know about paroxetine tablets?
Paroxetine tablets can cause serious side effects, including:
- •
-
Increased risk of suicidal thoughts or actions. Paroxetine tablets and other antidepressant medicines may increase suicidal thoughts and actions in some people 24 years of age and younger, especially within the first few months of treatment or when the dose is changed.Paroxetine tablets are not for use in children.
- •
- Depression or other mental illnesses are the most important causes of suicidal thoughts and actions.
- How can I watch for and try to prevent suicidal thoughts and actions?
- •
- Pay close attention to any changes, especially sudden changes in mood, behavior, thoughts or feelings or if you develop suicidal thoughts or actions. This is very important when an antidepressant medicine is started or when the dose is changed.
- •
- Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts or feelings or if you develop suicidal thoughts or actions.
- •
- Keep all follow-up visits with your healthcare provider as scheduled. Call your healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call your healthcare provider or get emergency medical help right away if you have any of the following symptoms, especially if they are new, worse, or worry you:
- •
- attempts to commit suicide
- •
- acting aggressive or violent
- •
- new or worse depression
- •
- feeling agitated, restless, angry, or irritable
- •
- an increase in activity and talking more than what is normal for you
- •
- acting on dangerous impulses
- •
- thoughts about suicide or dying
- •
- new or worse anxiety or panic attacks
- •
- trouble sleeping
- •
- other unusual changes in behavior or mood
What are paroxetine tablets?
Paroxetine tablets are a prescription medicine used in adults to treat:
- •
- A certain type of depression called Major Depressive Disorder (MDD)
- •
- Obsessive Compulsive Disorder (OCD)
- •
- Panic Disorder (PD)
- •
- Social Anxiety Disorder (SAD)
- •
- Generalized Anxiety Disorder (GAD)
- •
- Posttraumatic Stress Disorder (PTSD)
Do not take paroxetine tablets if you:
- •
- take a monoamine oxidase inhibitor (MAOI)
- •
- have stopped taking an MAOI in the last 14 days
- •
- are being treated with the antibiotic linezolid or the intravenous methylene blue
- •
- are taking pimozide
- •
- are taking thioridazine
- •
- are allergic to paroxetine or any of the ingredients in paroxetine tablets. See the end of this Medication Guide for a complete list of ingredients in paroxetine tablets.
Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI or one of these medicines, including the antibiotic linezolid or intravenous methylene blue.
Do not start taking an MAOI for at least 14 days after you stop treatment with paroxetine tablets.
Before taking paroxetine tablets, tell your healthcare provider about all your medical conditions, including if you:
- •
- have heart problems
- •
- have or had bleeding problems
- •
- have, or have a family history of, bipolar disorder, mania or hypomania
- •
- have or had seizures or convulsions
- •
- have glaucoma (high pressure in the eye)
- •
- have low sodium levels in your blood
- •
- have bone problems
- •
- have kidney or liver problems
- •
- are pregnant or plan to become pregnant. Paroxetine tablets may harm your unborn baby. Talk to your healthcare provider about the risks to your unborn baby if you take paroxetine tablets during pregnancy. Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with paroxetine tablets.
- •
- are breastfeeding or plan to breastfeed. Paroxetine passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with paroxetine tablets.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Paroxetine tablets and some other medicines may affect each other causing possible serious side effects. Paroxetine tablets may affect the way other medicines work and other medicines may affect the way paroxetine tablets work.
Especially tell your healthcare provider if you take:
- •
- medicines used to treat migraine headaches called triptans
- •
- tricyclic antidepressants
- •
- lithium
- •
- tramadol, fentanyl, meperidine, methadone, or other opioids
- •
- tryptophan
- •
- buspirone
- •
- amphetamines
- •
- St. John’s Wort
- •
- medicines that can affect blood clotting such as aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), warfarin
- •
- diuretics
- •
- tamoxifen
- •
- medicines used to treat mood, anxiety, psychotic, or thought disorders, including selective serotonin reuptake (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs)
Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take paroxetine tablets with your other medicines.
Do not start or stop any other medicines during treatment with paroxetine tablets without talking to your healthcare provider first. Stopping paroxetine tablets suddenly may cause you to have serious side effects. See, “What are the possible side effects of paroxetine tablets?”
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take paroxetine tablets?
- •
- Take paroxetine tablets exactly as prescribed. Your healthcare provider may need to change the dose of paroxetine tablets until it is the right dose for you.
- •
- Take paroxetine tablets 1 time each day in the morning.
- •
- Paroxetine tablets may be taken with or without food.
- •
- If you take too many paroxetine tablets, call your poison control center at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What are possible side effects of paroxetine tablets?
Paroxetine tablets can cause serious side effects, including:
- •
- See, “What is the most important information I should know about paroxetine tablets?”
- •
- Serotonin syndrome. A potentially life-threatening problem called serotonin syndrome can happen when you take paroxetine tablets with certain other medicines. See, “Who should not take paroxetine tablets?” Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of the following signs and symptoms of serotonin syndrome:
- •
- agitation
- •
- seeing or hearing things that are not real (hallucinations)
- •
- confusion
- •
- coma
- •
- fast heart beat
- •
- changes in blood pressure
- •
- dizziness
- •
- sweating
- •
- flushing
- •
- high body temperature (hyperthermia)
- •
- shaking (tremors), stiff muscles, or muscle twitching
- •
- loss of coordination
- •
- seizures
- •
- nausea, vomiting, diarrhea
- •
- Eye problems (angle-closure glaucoma). Paroxetine tablets may cause a type of eye problem called angle-closure glaucoma in people with certain other eye conditions. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are. Call your healthcare provider if you have eye pain, changes in your vision, or swelling or redness in or around the eye.
- •
- Medicine interactions. Taking paroxetine tablets with certain other medicines including thioridazine and pimozide may increase the risk of developing a serious heart problem called QT prolongation.
- •
- Seizures (convulsions).
- •
- Manic episodes. Manic episodes may happen in people with bipolar disorder who take paroxetine tablets. Symptoms may include:
- •
- greatly increased energy
- •
- racing thoughts
- •
- unusually grand ideas
- •
- talking more or faster than usual
- •
- severe problems sleeping
- •
- reckless behavior
- •
- excessive happiness or irritability
- •
- Discontinuation syndrome. Suddenly stopping paroxetine tablets may cause you to have serious side effects. Your healthcare provider may want to decrease your dose slowly. Symptoms may include:
- •
- nausea
- •
- sweating
- •
- changes in your mood
- •
- irritability and agitation
- •
- dizziness
- •
- electric shock feeling (paresthesia)
- •
- tremor
- •
- anxiety
- •
- confusion
- •
- headache
- •
- tiredness
- •
- problems sleeping
- •
- hypomania
- •
- ringing in your ears (tinnitus)
- •
- seizures
- •
-
Low sodium levels in your blood (hyponatremia). Low sodium levels in your blood that may be serious and may cause death, can happen during treatment with paroxetine tablets. Elderly people and people who take certain medicines may be at a greater risk for developing low sodium levels in your blood. Signs and symptoms may include:
- •
- headache
- •
- difficulty concentrating
- •
- memory changes
- •
- confusion
- •
- weakness and unsteadiness on your feet which can lead to falls
In more severe or more sudden cases, signs and symptoms include:
-
- •
- seeing or hearing things that are not real (hallucinations)
- •
- fainting
- •
- seizures
- •
- coma
- •
- stopping breathing (respiratory arrest)
- •
- Abnormal bleeding. Taking paroxetine tablets with aspirin, NSAIDs, or blood thinners may increase this risk. Tell your healthcare provider about any unusual bleeding or bruising.
- •
- Bone fractures.
- •
- Sexual problems (dysfunction). Taking selective serotonin reuptake inhibitors (SSRIs), including paroxetine tablets, may cause sexual problems.
- Symptoms in males may include:
- •
- Delayed ejaculation or inability to have an ejaculation
- •
- Decreased sex drive
- •
- Problems getting or keeping an erection
- Symptoms in females may include:
- •
- Decreased sex drive
- •
- Delayed orgasm or inability to have an orgasm
- Talk to your healthcare provider if you develop any changes in your sexual function or if you have any questions or concerns about sexual problems during treatment with paroxetine tablets. There may be treatments your healthcare provider can suggest.
The most common side effects of paroxetine tablets include:
- •
- male and female sexual function problems
- •
- constipation
- •
- diarrhea
- •
- dry mouth
- •
- problems sleeping
- •
- nervousness
- •
- sweating
- •
- yawning
- •
- weakness (asthenia)
- •
- decreased appetite
- •
- dizziness
- •
- infection
- •
- nausea
- •
- sleepiness
- •
- shaking (tremor)
These are not all the possible side effects of paroxetine tablets.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store paroxetine tablets?
- •
- Store paroxetine tablets between 20° to 25°C (68° to 77°F).
Keep paroxetine tablets and all medicines out of the reach of children.
General information about the safe and effective use of paroxetine tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take paroxetine tablets for a condition for which they were not prescribed. Do not give paroxetine tablets to other people, even if they have the same symptoms that you have. They may harm them. You may ask your healthcare provider or pharmacist for information about paroxetine tablets that is written for healthcare professionals.
What are the ingredients in paroxetine tablets?
Active ingredient: paroxetine hydrochloride
Inactive ingredients:
Tablets: dibasic calcium phosphate dihydrate, FD&C Blue No. 1 Aluminum Lake, hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, sodium lauryl sulfate, sodium starch glycolate (potato), titanium dioxide and triacetin.
Manufactured for: Mylan Pharmaceuticals Inc., Morgantown, WV 26505 U.S.A.
Manufactured by: ALPHAPHARM PTY LTD, 15 Garnet Street, Carole Park QLD 4300 Australia
The brands listed are trademarks of their respective owners.
For more information, call Mylan at 1-877-446-3679 (1-877-4-INFO-RX).
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Manufactured by:
ALPHAPHARM PTY LTD
15 Garnet Street
Carole Park QLD 4300
AustraliaRevised: 2/2024
ALP:PXTT:R15mh/ALP:MG:PXTT:R7mh
(2848/8)
-
PRINCIPAL DISPLAY PANEL – 10 mg
NDC 0378-7001-93
Paroxetine
Tablets, USP
10 mgPHARMACIST: Dispense the accompanying
Medication Guide to each patient.Rx only 30 Tablets
Each film-coated tablet contains
paroxetine hydrochloride, USP
(hemihydrate) equivalent to 10 mg
of paroxetine.Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Usual Dosage: See accompanying
prescribing information.Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Made in Australia
Mylan.com
RALP7001H3
2833/2
-
PRINCIPAL DISPLAY PANEL – 20 mg
NDC 0378-7002-93
Paroxetine
Tablets, USP
20 mgPHARMACIST: Dispense the accompanying
Medication Guide to each patient.Rx only 30 Tablets
Each film-coated tablet contains
paroxetine hydrochloride, USP
(hemihydrate) equivalent to 20 mg
of paroxetine.Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Usual Dosage: See accompanying
prescribing information.Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Made in Australia
Mylan.com
RALP7002H3
2835/2
-
PRINCIPAL DISPLAY PANEL – 30 mg
NDC 0378-7003-93
Paroxetine
Tablets, USP
30 mgPHARMACIST: Dispense the accompanying
Medication Guide to each patient.Rx only 30 Tablets
Each film-coated tablet contains
paroxetine hydrochloride, USP
(hemihydrate) equivalent to 30 mg
of paroxetine.Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Usual Dosage: See accompanying
prescribing information.Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Made in Australia
Mylan.com
RALP7003H3
2837/2
-
PRINCIPAL DISPLAY PANEL – 40 mg
NDC 0378-7004-93
Paroxetine
Tablets, USP
40 mgPHARMACIST: Dispense the accompanying
Medication Guide to each patient.Rx only 30 Tablets
Each film-coated tablet contains
paroxetine hydrochloride, USP
(hemihydrate) equivalent to 40 mg
of paroxetine.Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Usual Dosage: See accompanying
prescribing information.Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Made in Australia
Mylan.com
RALP7004H3
2839/2
-
INGREDIENTS AND APPEARANCE
PAROXETINE
paroxetine hydrochloride hemihydrate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0378-7001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PAROXETINE HYDROCHLORIDE (UNII: X2ELS050D8) (PAROXETINE - UNII:41VRH5220H) PAROXETINE 10 mg Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE 102 (UNII: PNR0YF693Y) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score 2 pieces Shape OVAL (modified capsule-shaped) Size 9mm Flavor Imprint Code M;N;1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0378-7001-93 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 2 NDC:0378-7001-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078902 03/24/2008 PAROXETINE
paroxetine hydrochloride hemihydrate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0378-7002 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PAROXETINE HYDROCHLORIDE (UNII: X2ELS050D8) (PAROXETINE - UNII:41VRH5220H) PAROXETINE 20 mg Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE 102 (UNII: PNR0YF693Y) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score 2 pieces Shape OVAL (modified capsule-shaped) Size 11mm Flavor Imprint Code M;N;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0378-7002-93 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 2 NDC:0378-7002-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078902 03/24/2008 PAROXETINE
paroxetine hydrochloride hemihydrate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0378-7003 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PAROXETINE HYDROCHLORIDE (UNII: X2ELS050D8) (PAROXETINE - UNII:41VRH5220H) PAROXETINE 30 mg Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE 102 (UNII: PNR0YF693Y) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score no score Shape ROUND Size 9mm Flavor Imprint Code M;N3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0378-7003-93 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 2 NDC:0378-7003-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078902 03/24/2008 PAROXETINE
paroxetine hydrochloride hemihydrate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0378-7004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PAROXETINE HYDROCHLORIDE (UNII: X2ELS050D8) (PAROXETINE - UNII:41VRH5220H) PAROXETINE 40 mg Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE 102 (UNII: PNR0YF693Y) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score no score Shape ROUND Size 10mm Flavor Imprint Code M;N4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0378-7004-93 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 2 NDC:0378-7004-10 1000 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/24/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078902 03/24/2008 Labeler - Mylan Pharmaceuticals Inc. (059295980)