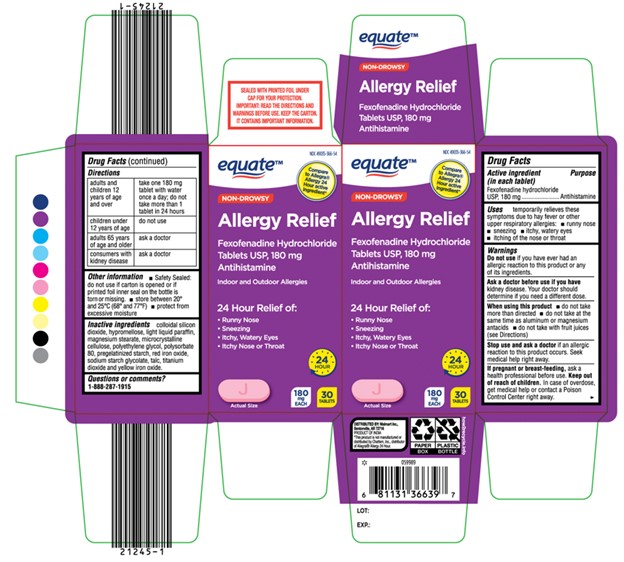
Label: EQUATE ALLERGY RELIEF- fexofenadine hydrochloride tablet
- NDC Code(s): 49035-366-02, 49035-366-54, 49035-366-59
- Packager: Walmart Inc.
- This is a repackaged label.
- Source NDC Code(s): 69230-300
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient(s)
- Purpose
- Use(s)
-
Warnings
Ask a doctor or pharmacist before use if
you have kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
- Questions
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
EQUATE ALLERGY RELIEF
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-366(NDC:69230-300) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) LIGHT MINERAL OIL (UNII: N6K5787QVP) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYSORBATE 80 (UNII: 6OZP39ZG8H) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) Product Characteristics Color pink Score no score Shape CAPSULE Size 18mm Flavor Imprint Code J;44 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-366-54 1 in 1 CARTON 04/14/2011 1 30 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:49035-366-59 2 in 1 CARTON 04/14/2011 2 30 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:49035-366-02 1 in 1 CARTON 04/14/2011 3 120 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204097 04/14/2011 Labeler - Walmart Inc. (051957769) Establishment Name Address ID/FEI Business Operations Legacy Pharmaceutical Packaging, LLC 143213275 repack(49035-366) , relabel(49035-366)