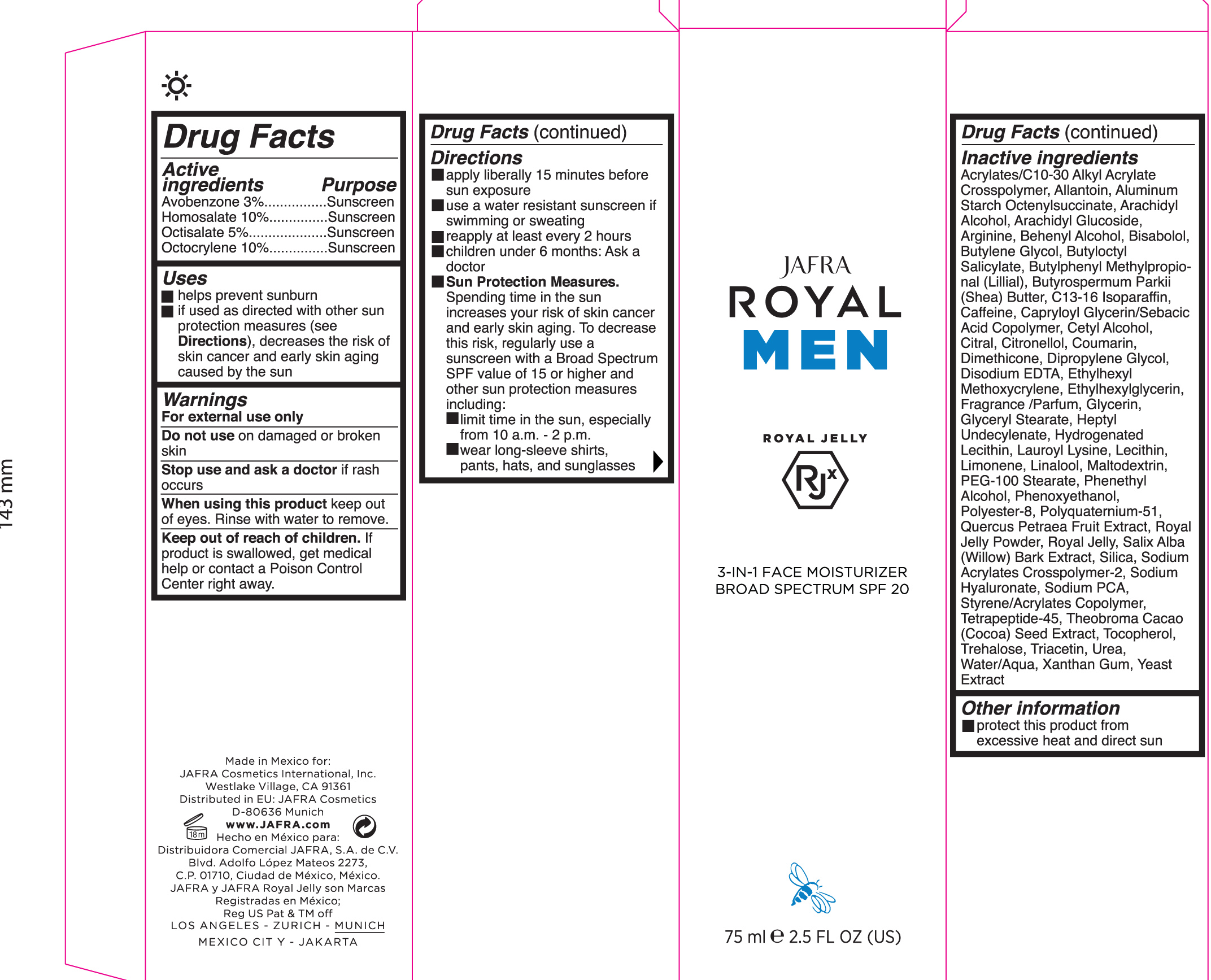
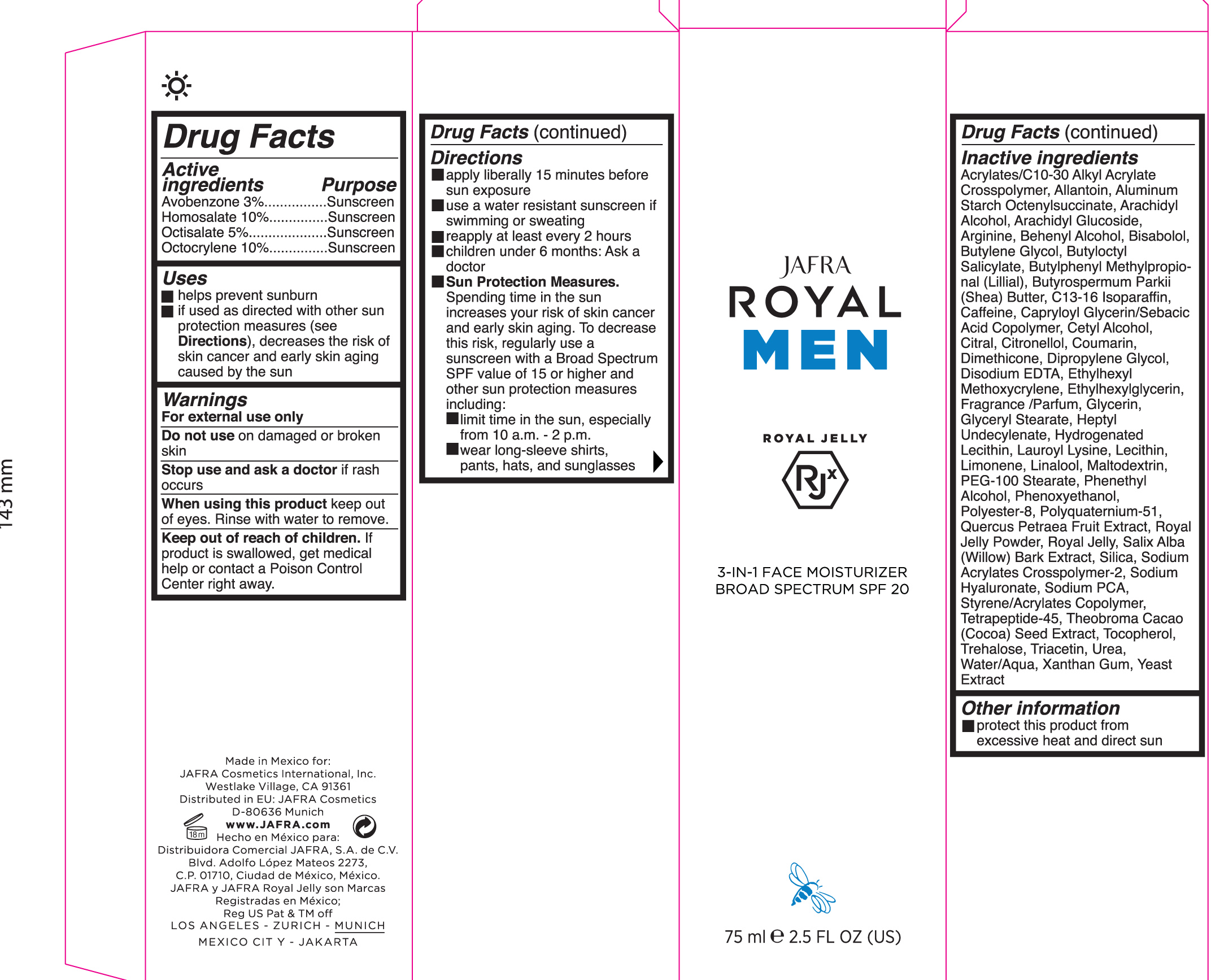
Label: 3-IN-1 FACE MOISTURIZER BROAD SPECTRUM SPF 20- avobenzone, homosalate, octisalate, octocrylene jelly
-
Contains inactivated NDC Code(s)
NDC Code(s): 68828-283-01 - Packager: Jafra cosmetics International
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 27, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- WARNINGS
- INDICATIONS & USAGE
-
DOSAGE & ADMINISTRATION
Directions
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- reapply at leastevery 2 hours.
- Children under 6 months of age: ask a doctor.
• Sun Protection Measures Spending time in the sun increases your risk of
skin cancer and early skin aging. To decrease this risk, regularly use a
sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun
protection measures including:- limit your time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
-
INACTIVE INGREDIENT
Inactive ingredients: Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Allantoin, Aluminum Starch Octenylsuccinate, Arachidyl
Alcohol, Arachidyl Glucoside, Arginine, Behenyl Alcohol, Blsabolol, Butylene Glycol, Butyloctyl Salicylate, Butylphenyl Methylpropional (LIIIIal), Butyrospanmum Parkll (Shea) Butter, C13-16 Isoparaffin, Caffeine, Capryloyl Glycerin/Sabacic Acid Copolymer, Cetyl Alcohol, citral, citronellol, Coumarin, Dimethicone, Dipropylene Glycol, Disodium EDTA, Ethylhexyl Methoxycrylene, Ethylhexylglycerin, Fragrance/Parfum, Glycerin, Glyceryl Stearate, Heptyl Undecylenate, Hydrogenated Lecithin, Lauroyl Lysine, Lecithin, Limonene, Linalool, Maltodextrin, PEG-100 Stearate, Phenethyl Alcohol, Phenoxyethanol, Polyester-8, Polyquatemlum-51, Quercus Petraea Fruit Extract, Royal Jelly Powder, Royal Jelly, Salix Alba (Willow) Bark Extract, Silica, Sodium Acrylates Crosspolymer-2, Sodium Hyaluronate, Sodium PCA, Styrene/Acrylates Copolymer, Tetrapeptide-45, Theobroma Cacao (Cocoa) Seed Extract, Tocopherol, Trehalose, Triacetin, Urea, Water/Aqua, Xanthan Gum, Yeast Extract - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
3-IN-1 FACE MOISTURIZER BROAD SPECTRUM SPF 20
avobenzone, homosalate, octisalate, octocrylene jellyProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68828-283 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 10 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength ROYAL JELLY (UNII: L497I37F0C) SALIX ALBA BARK (UNII: 205MXS71H7) UREA (UNII: 8W8T17847W) DOCOSANOL (UNII: 9G1OE216XY) LEVOMENOL (UNII: 24WE03BX2T) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) SHEA BUTTER (UNII: K49155WL9Y) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) LIMONENE, (+)- (UNII: GFD7C86Q1W) MALTODEXTRIN (UNII: 7CVR7L4A2D) PHENOXYETHANOL (UNII: HIE492ZZ3T) TRIACETIN (UNII: XHX3C3X673) CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 71DD5V995L) ALLANTOIN, (+)- (UNII: XDK458E1J9) ARACHIDYL ALCOHOL (UNII: 1QR1QRA9BU) C13-16 ISOPARAFFIN (UNII: LED42LZG6O) CAPRYLOYL GLYCERIN/SEBACIC ACID COPOLYMER (2000 MPA.S) (UNII: N7YC58165T) CETYL ALCOHOL (UNII: 936JST6JCN) DIMETHICONE (UNII: 92RU3N3Y1O) DIPROPYLENE GLYCOL (UNII: E107L85C40) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) LAUROYL LYSINE (UNII: 113171Q70B) PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) POLYESTER-8 (1400 MW, CYANODIPHENYLPROPENOYL CAPPED) (UNII: T9296U138P) POLYQUATERNIUM-51 (2-METHACRYLOYLOXYETHYL PHOSPHORYLCHOLINE/N-BUTYL METHACRYLATE; 3:7) (UNII: EL9825H96J) WATER (UNII: 059QF0KO0R) XANTHAN GUM (UNII: TTV12P4NEE) YEAST, UNSPECIFIED (UNII: 3NY3SM6B8U) CAFFEINE (UNII: 3G6A5W338E) CITRAL (UNII: T7EU0O9VPP) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) COCOA (UNII: D9108TZ9KG) TOCOPHEROL (UNII: R0ZB2556P8) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) BUTYLPHENYL METHYLPROPIONAL (UNII: T7540GJV69) HEPTYL UNDECYLENATE (UNII: W77QUB6GXO) PEG-100 STEARATE (UNII: YD01N1999R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) COUMARIN (UNII: A4VZ22K1WT) ETHYLHEXYL METHOXYCRYLENE (UNII: S3KFG6Q5X8) LINALOOL, (+/-)- (UNII: D81QY6I88E) SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) ARACHIDYL GLUCOSIDE (UNII: 6JVW35JOOJ) ARGININE (UNII: 94ZLA3W45F) HYALURONATE SODIUM (UNII: YSE9PPT4TH) TREHALOSE (UNII: B8WCK70T7I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68828-283-01 75 mL in 1 CARTON; Type 0: Not a Combination Product 03/27/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 03/27/2019 Labeler - Jafra cosmetics International (041676479) Establishment Name Address ID/FEI Business Operations Jafra Manufacturing, S.A. de C.V. 814732061 manufacture(68828-283)