Label: CLEARASIL RAPIDLY CLEAR ACNE SOLUTIONS- salicylic acid kit
- NDC Code(s): 63824-431-90, 63824-432-01, 63824-449-10
- Packager: RB Health (US) LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
For external use only
When using this product
- avoid contact with the eyes. If product gets into the eyes, rinse thoroughly with water.
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- limit use to the face and neck
-
Directions
- wet face
- dispense product into hands, lather and massage gently onto face and neck, avoiding the delicate eye area
- rinse thoroughly with warm water and pat dry
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
- Other information
-
Inactive ingredients
water, sodium cocoyl isethionate, cetearyl alcohol, glycerin, sodium laureth sulfate, sodium cocoamphoacetate, cocamidopropyl betaine, sodium hydroxide, acrylates/C10-30 alkyl acrylate crosspolymer, fragrance, disodium EDTA, lavandula stoechas extract, helichrysum italicum extract, cistus monspeliensis extract
- Questions?
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
For external use only
When using this product
- avoid contact with the eyes. If product gets into the eyes, rinse thoroughly with water.
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- limit use to the face and neck
-
Directions
- clean the skin thoroughly before applying this product
- wipe a pad over face and neck to cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
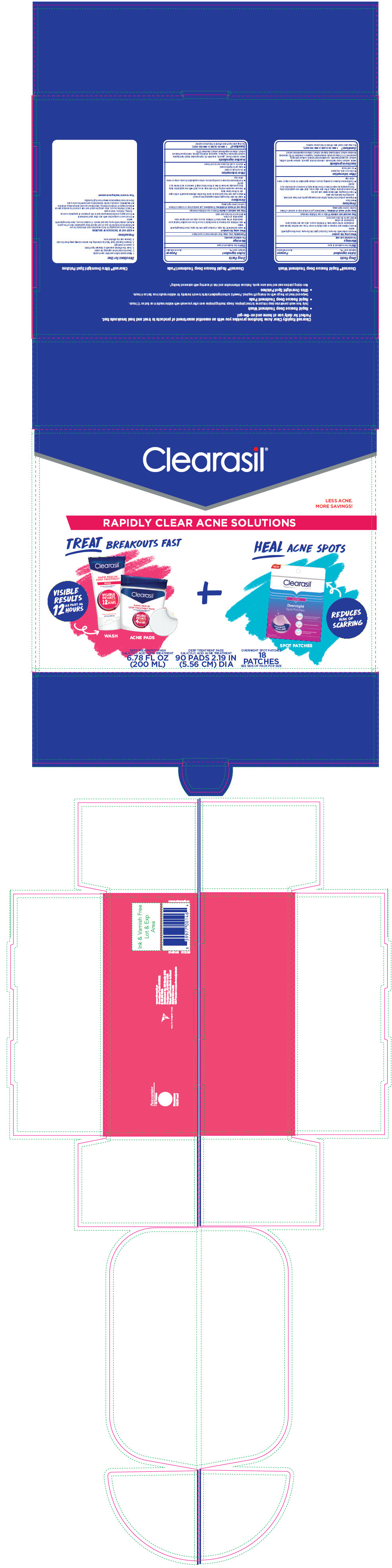
PRINCIPAL DISPLAY PANEL - Kit Carton
Clearasil ®
LESS ACNE.
MORE SAVINGS!RAPIDLY CLEAR ACNE SOLUTIONS
TREAT BREAKOUTS FAST
VISIBILE
RESULTS
AS FAST AS
12
HOURSWASH
ACNE PADS+
HEAL ACNE SPOTS
REDUCES
RISK OF
SCARRINGSPOT PATCHES
DEEP TREATMENT WASH
SALICYLIC ACID ACNE TREATMENT
6.78 FL OZ
(200 ML)DEEP TREATMENT PADS
SALICYLIC ACID ACNE TREATMENT
90 PADS 2.19 IN
(5.56 CM) DIAOVERNIGHT SPOT PATCHES
18
PATCHES
SEE SIDE OF PACK FOR SIZE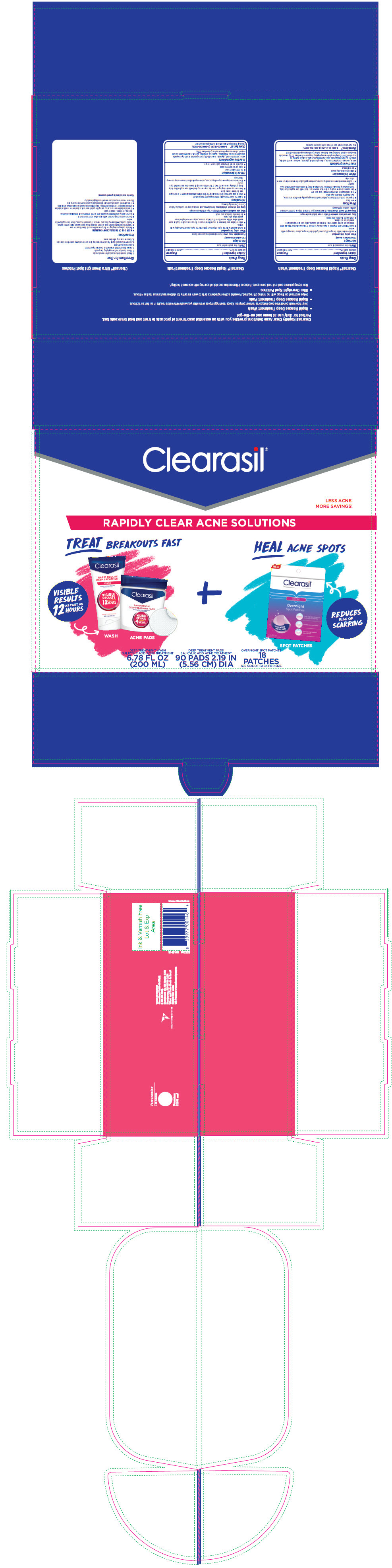
-
INGREDIENTS AND APPEARANCE
CLEARASIL RAPIDLY CLEAR ACNE SOLUTIONS
salicylic acid kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63824-449 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63824-449-10 1 in 1 CARTON 09/01/2020 12/31/2024 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 200 mL Part 2 1 JAR 90 Part 3 1 POUCH 18 Part 1 of 3 CLEARASIL RAPID RESCUE DEEP TREATMENT WASH
salicylic acid lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 2 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM COCOYL ISETHIONATE (UNII: 518XTE8493) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERIN (UNII: PDC6A3C0OX) SODIUM LAURETH-3 SULFATE (UNII: BPV390UAP0) SODIUM COCOAMPHOACETATE (UNII: W7Q5E87674) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SODIUM HYDROXIDE (UNII: 55X04QC32I) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) HELICHRYSUM ITALICUM FLOWER (UNII: P62Y550X24) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63824-432-01 200 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 12/01/2017 Part 2 of 3 CLEARASIL RAPID RESCUE DEEP TREATMENT PADS
salicylic acid clothProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.02 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) GLYCERIN (UNII: PDC6A3C0OX) ISOCETETH-20 (UNII: O020065R7Z) HYDROGENATED STARCH HYDROLYSATE (UNII: 27F77DSJ5V) SODIUM HYDROXIDE (UNII: 55X04QC32I) HELICHRYSUM ITALICUM FLOWER (UNII: P62Y550X24) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) Product Characteristics Color Score Shape ROUND Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63824-431-90 90 in 1 JAR; Type 4: Device Coated/Impregnated/Otherwise Combined with Drug Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 12/05/2017 Part 3 of 3 CLEARASIL ULTRA OVERNIGHT SPOT
cleansing (cold creams, cleansing lotions, liquids, and pads) patchProduct Information Route of Administration TOPICAL Product Characteristics shape ROUND C48348 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 18 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 09/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 09/01/2020 12/31/2024 Labeler - RB Health (US) LLC (081049410)