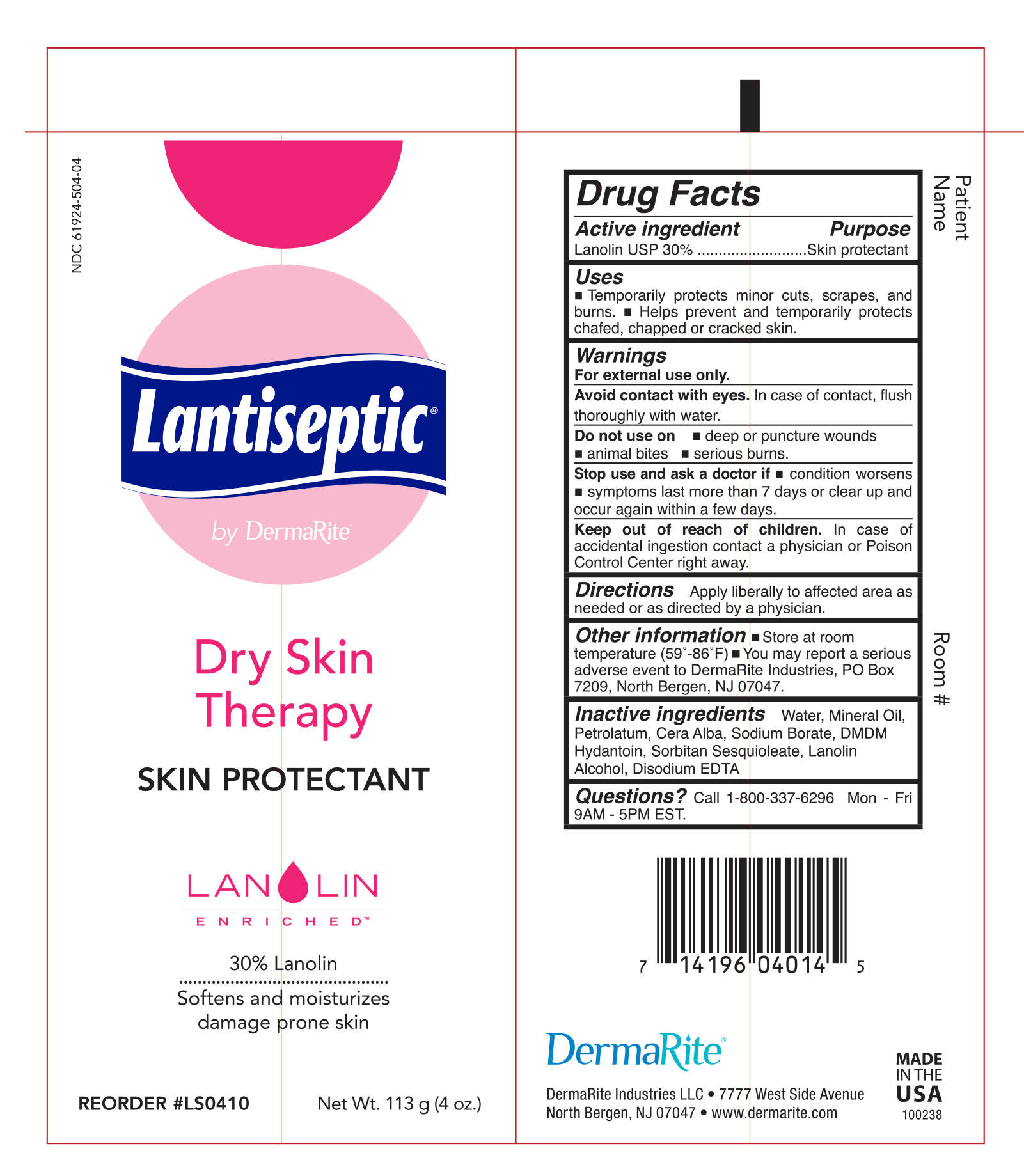
Label: LANTISEPTIC BY DERMARITE DRY SKIN THERAPY- lanolin cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 61924-504-04, 61924-504-05, 61924-504-14 - Packager: Dermarite Industries LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 17, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Uses
-
Warnings
• For external use only.
• Avoid contact with eyes. In case of contact, flush thoroughly with water
- Do not use on
- Deep or puncture wounds
- animal bites
- serious burns
Stop use and ask a doctor if
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Keep our of reach of children. In case of accidental ingestion contact a physician or Poision Control Center right away.
- Directions
- Other Information
- Inactive Ingredients
- Questions or Comments?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
LANTISEPTIC BY DERMARITE DRY SKIN THERAPY
lanolin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61924-504 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LANOLIN (UNII: 7EV65EAW6H) (LANOLIN - UNII:7EV65EAW6H) LANOLIN 300 mg in 1 g Inactive Ingredients Ingredient Name Strength PHENOXYETHANOL (UNII: HIE492ZZ3T) YELLOW WAX (UNII: 2ZA36H0S2V) CAPRYLYL GLYCOL (UNII: 00YIU5438U) EDETATE DISODIUM (UNII: 7FLD91C86K) LANOLIN ALCOHOLS (UNII: 884C3FA9HE) MINERAL OIL (UNII: T5L8T28FGP) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) SODIUM BORATE (UNII: 91MBZ8H3QO) SORBITAN SESQUIOLEATE (UNII: 0W8RRI5W5A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61924-504-14 14.2 g in 1 PACKET; Type 0: Not a Combination Product 12/25/2018 2 NDC:61924-504-04 113 g in 1 TUBE; Type 0: Not a Combination Product 12/25/2018 3 NDC:61924-504-05 5 g in 1 PACKET; Type 0: Not a Combination Product 12/25/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 12/25/2018 Labeler - Dermarite Industries LLC (883925562) Registrant - Dermarite Industries LLC (883925562)