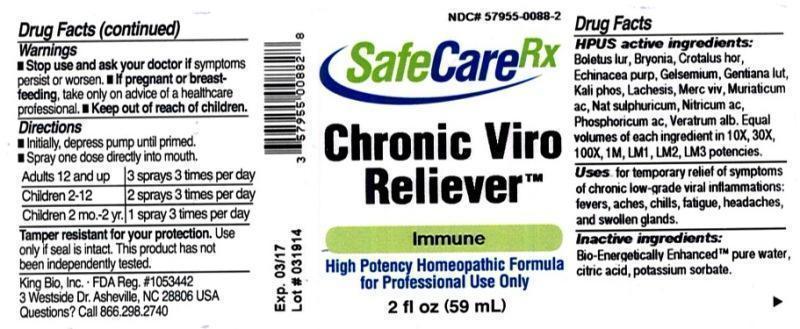
Label: CHRONIC VIRO RELIEVER- boletus luridus, bryonia, crotalus horridus, echinacea purpurea, gelsemium sempervirens, gentiana lutea, kali phosphoricum, lachesis mutus, mercurius vivus, muriaticum acidum, natrum sulphuricum, nitricum acidum, phosphoricum acidum, veratrum album liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 57955-0088-2 - Packager: King Bio Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 31, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
HPUS active ingredients: Boletus luridus, Bryonia, Crotalus horridus, Echinacea purpurea, Gelsemium sempervirens, Gentiana lutea, Kali phosphoricum, Lachesis mutus, Mercurius vivus, Muriaticum acidum, Natrum sulphuricum, Nitricum acidum, Phosphoricum acidum, Veratrum album . Equal volumes of each ingredient in 10X, 30X, 100X, 1M, LM1, LM2, LM3 potencies.
- PURPOSE
- INACTIVE INGREDIENT
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CHRONIC VIRO RELIEVER
boletus luridus, bryonia, crotalus horridus, echinacea purpurea, gelsemium sempervirens, gentiana lutea, kali phosphoricum, lachesis mutus, mercurius vivus, muriaticum acidum, natrum sulphuricum, nitricum acidum, phosphoricum acidum, veratrum album liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57955-0088 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BOLETUS LURIDUS FRUITING BODY (UNII: 9HKN65WT0X) (BOLETUS LURIDUS FRUITING BODY - UNII:9HKN65WT0X) BOLETUS LURIDUS FRUITING BODY 10 [hp_X] in 59 mL BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 10 [hp_X] in 59 mL CROTALUS HORRIDUS HORRIDUS VENOM (UNII: YHA2XLJ956) (CROTALUS HORRIDUS HORRIDUS VENOM - UNII:YHA2XLJ956) CROTALUS HORRIDUS HORRIDUS VENOM 10 [hp_X] in 59 mL ECHINACEA PURPUREA (UNII: QI7G114Y98) (ECHINACEA PURPUREA - UNII:QI7G114Y98) ECHINACEA PURPUREA 10 [hp_X] in 59 mL GELSEMIUM SEMPERVIRENS ROOT (UNII: 639KR60Q1Q) (GELSEMIUM SEMPERVIRENS ROOT - UNII:639KR60Q1Q) GELSEMIUM SEMPERVIRENS ROOT 10 [hp_X] in 59 mL GENTIANA LUTEA ROOT (UNII: S72O3284MS) (GENTIANA LUTEA ROOT - UNII:S72O3284MS) GENTIANA LUTEA ROOT 10 [hp_X] in 59 mL POTASSIUM PHOSPHATE, DIBASIC (UNII: CI71S98N1Z) (PHOSPHATE ION - UNII:NK08V8K8HR) POTASSIUM PHOSPHATE, DIBASIC 10 [hp_X] in 59 mL LACHESIS MUTA VENOM (UNII: VSW71SS07I) (LACHESIS MUTA VENOM - UNII:VSW71SS07I) LACHESIS MUTA VENOM 10 [hp_X] in 59 mL MERCURY (UNII: FXS1BY2PGL) (MERCURY - UNII:FXS1BY2PGL) MERCURY 10 [hp_X] in 59 mL HYDROCHLORIC ACID (UNII: QTT17582CB) (HYDROCHLORIC ACID - UNII:QTT17582CB) HYDROCHLORIC ACID 10 [hp_X] in 59 mL SODIUM SULFATE (UNII: 0YPR65R21J) (SODIUM SULFATE ANHYDROUS - UNII:36KCS0R750) SODIUM SULFATE 10 [hp_X] in 59 mL NITRIC ACID (UNII: 411VRN1TV4) (NITRIC ACID - UNII:411VRN1TV4) NITRIC ACID 10 [hp_X] in 59 mL PHOSPHORIC ACID (UNII: E4GA8884NN) (PHOSPHORIC ACID - UNII:E4GA8884NN) PHOSPHORIC ACID 10 [hp_X] in 59 mL VERATRUM ALBUM ROOT (UNII: QNS6W5US1Z) (VERATRUM ALBUM ROOT - UNII:QNS6W5US1Z) VERATRUM ALBUM ROOT 10 [hp_X] in 59 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57955-0088-2 59 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/28/2014 Labeler - King Bio Inc. (617901350) Registrant - King Bio Inc. (617901350) Establishment Name Address ID/FEI Business Operations King Bio Inc. 617901350 manufacture(57955-0088)