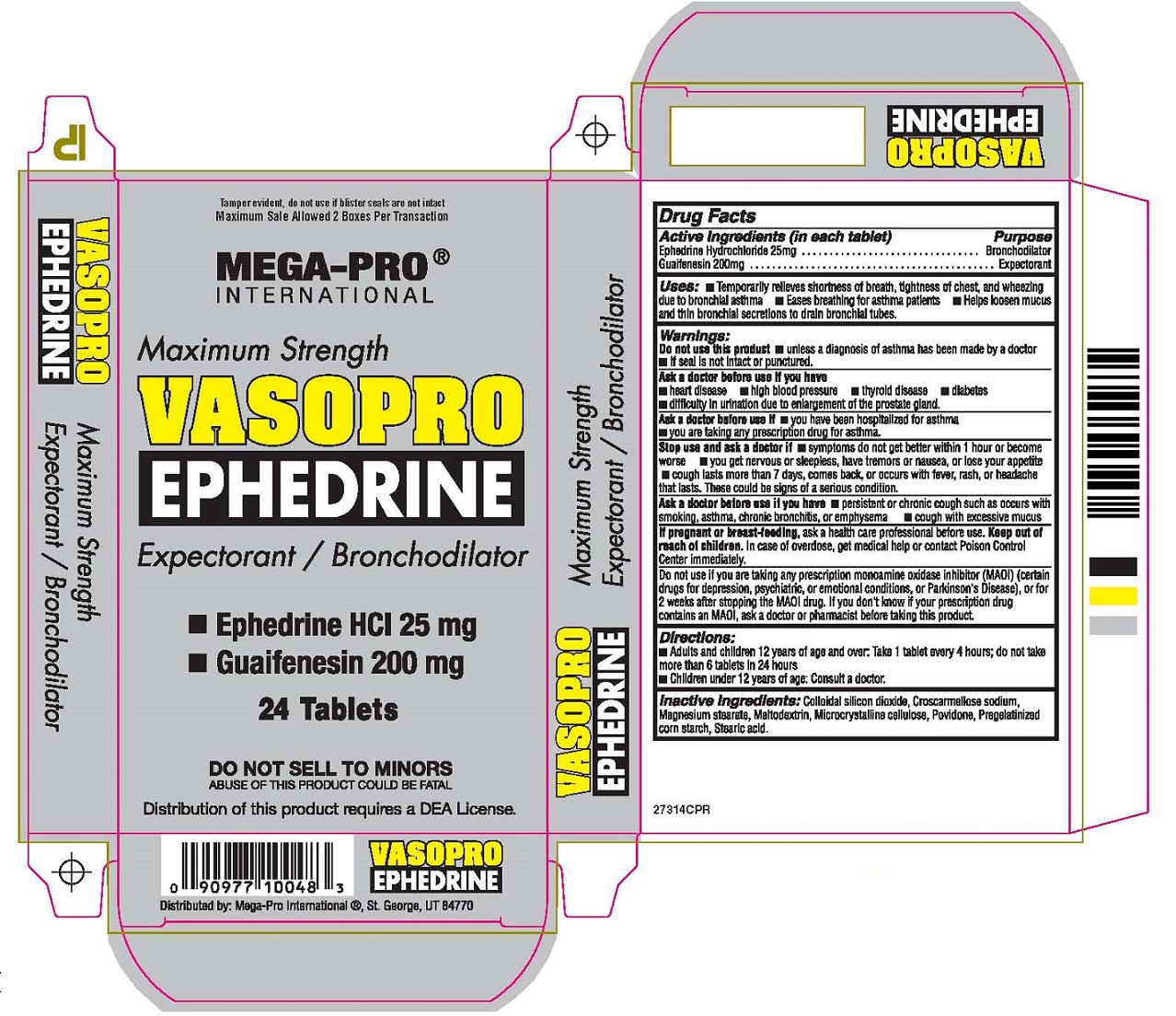
Label: VASOPRO EPHEDRINE- ephedrine hydrochloride, guaifenesin tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 70258-320-24 - Packager: Optimal Research Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 23, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients (in each tablet)
- Purpose
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
Warnings:
Do not use this product • unless a diagnosis of asthma has been made by a doctor
• If seal is not intact or punctured.Ask a doctor before use if you have
• heart disease • high blood pressure • thyroid disease • diabetes
• difficulty in urination due to enlargement of the prostate gland.Ask a doctor before use if • you have been hospitalized for asthma
• you are taking any prescription drug for asthma.
Stop use and ask a doctor if • symptoms do not get better within 1 hour or become worse
• you get nervous or sleepless, have tremors or nausea, or lose your appetite
• cough lasts more than 7 days, comes back, or occurs with fever, rash, or headache that lasts.
These could be signs of a serious condition.
Ask a doctor before use if you have • persistent or chronic cough such as occurs with smoking, asthma,
chronic bronchitis, or emphysema • cough with excessive mucus
If pregnant or breast-feeding, ask a health care professional before use.
Do not use if you are taking any prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression,
psychiatric, or emotional conditions, or Parkinson’s Disease), or for 2 weeks after stopping the MAOI drug.
If you don’t know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- Directions:
- INACTIVE INGREDIENT
-
SPL UNCLASSIFIED SECTION
Tamper evident, do not use if blister seals are not intact
Maximum Sale Allowed 2 Boxes Per TransactionMEGA-PRO®
INTERNATIONALMaximum Strength
VASOPRO
EPHEDRINE
Expectorant / Bronchodilator
24 Tablets
DO NOT SELL TO MINORS
ABUSE OF THIS PRODUCT COULD BE FATALDistribution of this product requires a DEA License.
Distributed by: Mega-Pro International®, St. George, UT 84770
- Packaging
-
INGREDIENTS AND APPEARANCE
VASOPRO EPHEDRINE
ephedrine hydrochloride, guaifenesin tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70258-320 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EPHEDRINE HYDROCHLORIDE (UNII: NLJ6390P1Z) (EPHEDRINE - UNII:GN83C131XS) EPHEDRINE HYDROCHLORIDE 25 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg Inactive Ingredients Ingredient Name Strength COLLOIDAL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POVIDONE (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score 4 pieces Shape ROUND Size 9mm Flavor Imprint Code MP101 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70258-320-24 1 in 1 BOX 11/23/2015 1 24 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 11/23/2015 Labeler - Optimal Research Inc (612492678) Establishment Name Address ID/FEI Business Operations Pharmatech Llc 968082094 manufacture(70258-320)