Label: SODIUM SULFACETAMIDE lotion
- NDC Code(s): 68682-010-04
- Packager: Oceanside Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated May 31, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Each mL of Sodium Sulfacetamide Lotion, 10% contains 100 mg of sodium sulfacetamide in a vehicle consisting of edetic acid, hydroxyethyl cellulose, lauramide DEA, methylparaben, PEG-400 monolaurate, propylene glycol, purified water, silicone emulsion, sodium chloride, sodium metabisulfite, and xanthan gum.
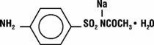
Sodium sulfacetamide is a sulfonamide with antibacterial activity. Chemically, sodium sulfacetamide is N' -[(4-amino phenyl) sulfonyl] - acetamide, monosodium salt, monohydrate. The structural formula is:
-
CLINICAL PHARMACOLOGY
The most widely accepted mechanism of action of sulfonamides is the Woods-Fildes theory, based on sulfonamides acting as a competitive inhibitor of para-aminobenzoic acid (PABA) utilization, an essential component for bacterial growth. While absorption through intact skin in humans has not been determined, in vitro studies with human cadaver skin indicated a percutaneous absorption of about 4%. Sodium sulfacetamide is readily absorbed from the gastrointestinal tract when taken orally and excreted in the urine largely unchanged. The biological half-life has been reported to be between 7 to 13 hours.
The pharmacokinetics of sulfacetamide and its major metabolite sulfaniliamide in Sodium Sulfacetamide Lotion was evaluated in adult subjects (N=14) with acne vulgaris. The subjects applied Sodium Sulfacetamide Lotion to their face, back, chest, and shoulders every 12 hours for 28 days. The percentage of the applied dose of Sodium Sulfacetamide Lotion excreted in the urine as sulfacetamide plus sulfanilamide ranged from 0.08 to 0.33%.
- INDICATIONS
-
CONTRAINDICATIONS
Sodium Sulfacetamide Lotion is contraindicated for use by patients having known hypersensitivity to sulfonamides or any other component of this preparation (see WARNINGS).
-
WARNINGS
Fatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias. Hypersensitivity reactions may occur when a sulfonamide is readministered, irrespective of the route of administration. Sensitivity reactions have been reported in individuals with no prior history of sulfonamide hypersensitivity. At the first sign of hypersensitivity, skin rash, or other reactions, discontinue use of this preparation (see ADVERSE REACTIONS).
Sodium Sulfacetamide Lotion contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people (see CONTRAINDICATIONS).
-
PRECAUTIONS
General
For external use only. Keep away from eyes. If irritation develops, use of the product should be discontinued and appropriate therapy instituted. Patients should be carefully observed for possible local irritation or sensitization during long-term therapy. Hypersensitivity reactions may occur when a sulfonamide is readministered irrespective of the route of administration, and cross-sensitivity between different sulfonamides may occur. Sodium sulfacetamide can cause reddening and scaling of the skin. Particular caution should be employed if areas of involved skin to be treated are denuded or abraded.
Keep out of reach of children.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential.
Animal reproduction studies have not been conducted with Sodium Sulfacetamide Lotion. It is also not known whether Sodium Sulfacetamide Lotion can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Sodium Sulfacetamide Lotion should be given to a pregnant woman only if clearly needed.
Kernicterus may occur in the newborn as a result of treatment of a pregnant woman at term with orally administered sulfonamide. There are no adequate and well-controlled studies of Sodium Sulfacetamide Lotion in pregnant women, and it is not known whether topically applied sulfonamides can cause fetal harm when administered to a pregnant woman.
Nursing Mothers
It is not known whether sodium sulfacetamide is excreted in human milk following topical use of Sodium Sulfacetamide Lotion. Systemically administered sulfonamides are capable of producing kernicterus in the infants of lactating women. Small amounts of orally administered sulfonamides have been reported to be eliminated in human milk. Because many drugs are excreted in human milk, caution should be exercised in prescribing for nursing women.
-
ADVERSE REACTIONS
In controlled clinical trials for the management of acne vulgaris, the occurrence of adverse reactions associated with the use of Sodium Sulfacetamide Lotion was infrequent and restricted to local events. The total incidence of adverse reactions reported in these studies was less than 2%. Only one of 105 patients treated with Sodium Sulfacetamide Lotion had adverse reactions of erythema, itching, and edema. It has been reported that sodium sulfacetamide may cause local irritation, stinging, and burning. While the irritation may be transient, occasionally the use of medication has to be discontinued.
To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 118 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
SODIUM SULFACETAMIDE
sodium sulfacetamide lotionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68682-010 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength sulfacetamide sodium (UNII: 4NRT660KJQ) (sulfacetamide - UNII:4965G3J0F5) sulfacetamide sodium 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength EDETIC ACID (UNII: 9G34HU7RV0) HYDROXYETHYL CELLULOSE (2000 MPA.S AT 1%) (UNII: S38J6RZN16) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) LAURIC DIETHANOLAMIDE (UNII: I29I2VHG38) methylparaben (UNII: A2I8C7HI9T) sodium chloride (UNII: 451W47IQ8X) sodium metabisulfite (UNII: 4VON5FNS3C) water (UNII: 059QF0KO0R) xanthan gum (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68682-010-04 1 in 1 CARTON 12/18/2015 1 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA019931 12/18/2015 Labeler - Oceanside Pharmaceuticals (832011691) Establishment Name Address ID/FEI Business Operations Bausch Health Companies Inc. 245141858 MANUFACTURE(68682-010) , PACK(68682-010) , LABEL(68682-010)