Label: PLEO NIG- aspergillus niger var. niger solution/ drops
-
Contains inactivated NDC Code(s)
NDC Code(s): 60681-2107-1, 60681-2107-2 - Packager: Sanum Kehlbeck GmbH & Co. KG
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 8, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- INDICATIONS
- DIRECTIONS FOR USE
- DOSAGE
- INGREDIENTS
-
WARNING
If symptoms persist more than a few days, contact a licensed practitioner. As with any drug, if you are pregnant or nursing a baby, seek the advice of a health care professional before using this product.
- SPL UNCLASSIFIED SECTION
-
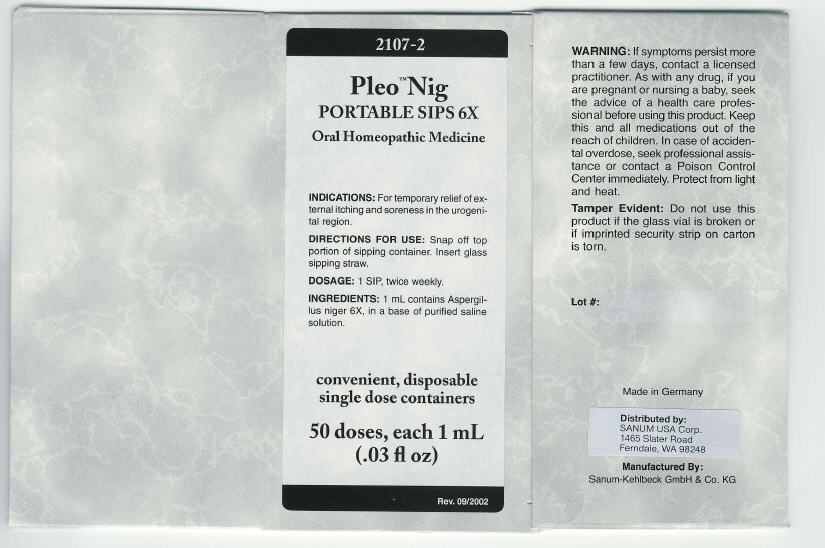
PRINCIPAL DISPLAY PANEL - 1 mL Carton
2107-2
Pleo™ Nig
PORTABLE SIPS 6XOral Homeopathic Medicine
INDICATIONS: For temporary relief of ex-
ternal itching and soreness in the urogeni-
tal region.DIRECTIONS FOR USE: Snap off top
portion of sipping container. Insert glass
sipping straw.DOSAGE: 1 SIP, twice weekly.
INGREDIENTS: 1 mL contains Aspergil-
lus niger 6X, in a base of purified saline
solution.convenient, disposable
single dose containers50 doses, each 1 mL
(.03 fl oz)Rev. 09/2002
-
INGREDIENTS AND APPEARANCE
PLEO NIG
aspergillus niger var. niger solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:60681-2107 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength aspergillus niger var. niger (UNII: 9IOA40ANG6) (aspergillus niger var. niger - UNII:9IOA40ANG6) aspergillus niger var. niger 6 [HP_X] in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) sodium chloride (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60681-2107-1 10 in 1 CARTON 1 1 mL in 1 VIAL, GLASS 2 NDC:60681-2107-2 50 in 1 CARTON 2 1 mL in 1 VIAL, GLASS Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved homeopathic 03/19/1997 Labeler - Sanum Kehlbeck GmbH & Co. KG (318386133)