Label: ANTI-ITCH MEDICATED MAXIMUM STRENGTH- benzocaine and resorcinol cream
- NDC Code(s): 69842-588-28
- Packager: CVS Health
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 25, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
- Warnings
- Stop use and ask doctor if
- Keep out of reach of children.
- Directions
- Other Information:
-
Inactive ingredients
Water, Mineral Oil, Cetyl Alcohol, Propylene Glycol, Glyceryl Stearate, PEG-100 Stearate, Isopropyl Palmitate, Aloe Barbadensis Leaf Extract, Tocopheryl Acetate, Retinyl Palmitate, Zea Mays (Corn) Oil, Cholecalciferol, Lanolin Alcohol, Fragrance, Methylparaben, Carbomer, Isopropyl Myristate, Isopropyl Stearate, Sodium Sulfite, Triethanolamine, Trisodium EDTA, Maltodextrin
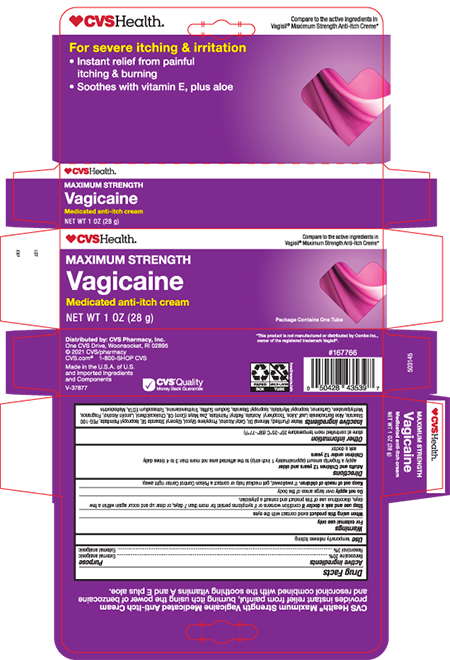
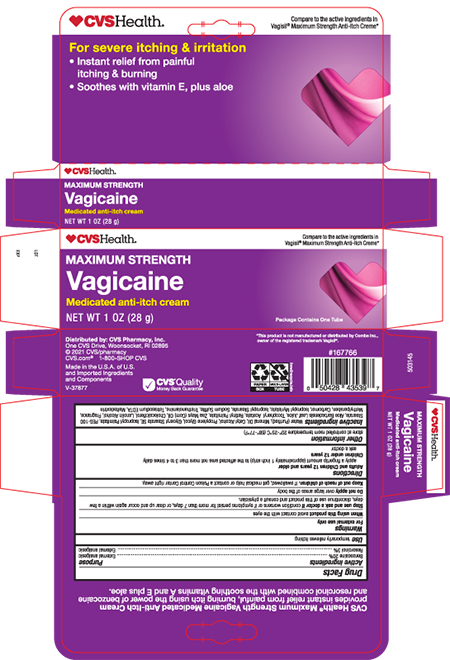
- Principal Display Panel- Tube
- Principal Display Panel- Carton
-
INGREDIENTS AND APPEARANCE
ANTI-ITCH MEDICATED MAXIMUM STRENGTH
benzocaine and resorcinol creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-588 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg in 1 g RESORCINOL (UNII: YUL4LO94HK) (RESORCINOL - UNII:YUL4LO94HK) RESORCINOL 30 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) MINERAL OIL (UNII: T5L8T28FGP) CETYL ALCOHOL (UNII: 936JST6JCN) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) PEG-100 STEARATE (UNII: YD01N1999R) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) ALOE VERA LEAF (UNII: ZY81Z83H0X) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) CORN OIL (UNII: 8470G57WFM) CHOLECALCIFEROL (UNII: 1C6V77QF41) LANOLIN ALCOHOLS (UNII: 884C3FA9HE) METHYLPARABEN (UNII: A2I8C7HI9T) CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) ISOPROPYL STEARATE (UNII: 43253ZW1MZ) SODIUM SULFITE (UNII: VTK01UQK3G) TROLAMINE (UNII: 9O3K93S3TK) TRISODIUM HEDTA (UNII: K3E0U7O8KI) MALTODEXTRIN (UNII: 7CVR7L4A2D) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-588-28 1 in 1 CARTON 09/30/2021 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part348 09/30/2021 Labeler - CVS Health (062312574) Establishment Name Address ID/FEI Business Operations Sheffield Pharmaceuticals LLC 151177797 manufacture(69842-588)